Abstract
Background
Smokers increasingly use e-cigarettes for many reasons, including attempts to quit combustible cigarettes and to use nicotine where smoking is prohibited. We aimed to assess the association between e-cigarette use and cigarette smoking cessation among adult cigarette smokers, irrespective of their motivation for using e-cigarettes.Methods
PubMed and Web of Science were searched between April 27, 2015, and June 17, 2015. Data extracted included study location, design, population, definition and prevalence of e-cigarette use, comparison group (if applicable), cigarette consumption, level of nicotine dependence, other confounders, definition of quitting smoking, and odds of quitting smoking. The primary endpoint was cigarette smoking cessation. Odds of smoking cessation among smokers using e-cigarettes compared with smokers not using e-cigarettes were assessed using a random effects meta-analysis. A modification of the ACROBAT-NRSI tool and the Cochrane Risk of Bias Tool were used to assess bias. This meta-analysis is registered with PROSPERO (number CRD42015020382).Findings
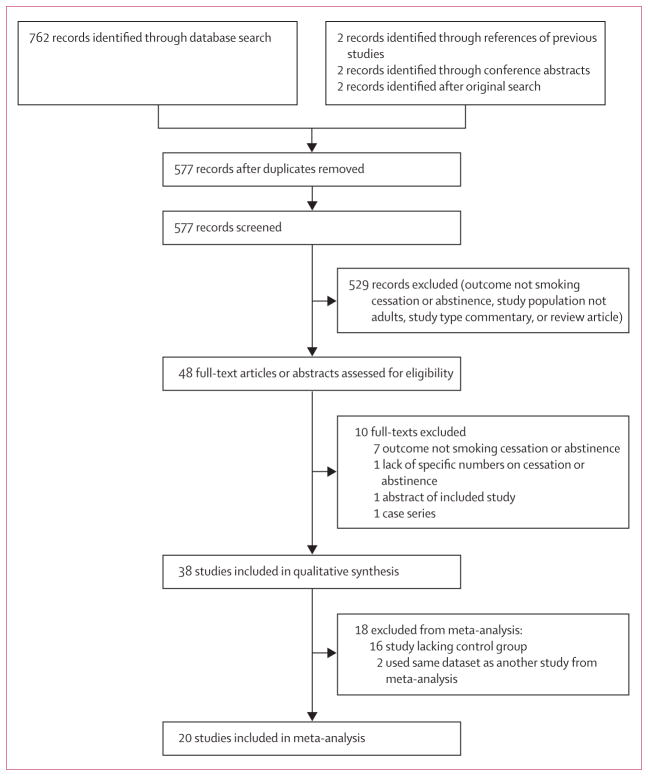
38 studies (of 577 studies identified) were included in the systematic review; all 20 studies with control groups (15 cohort studies, three cross-sectional studies, and two clinical trials) were included in random effects meta-analysis and sensitivity analyses. Odds of quitting cigarettes were 28% lower in those who used e-cigarettes compared with those who did not use e-cigarettes (odds ratio [OR] 0·72, 95% CI 0·57-0·91). Association of e-cigarette use with quitting did not significantly differ among studies of all smokers using e-cigarettes (irrespective of interest in quitting cigarettes) compared with studies of only smokers interested in cigarette cessation (OR 0·63, 95% CI 0·45-0·86 vs 0·86, 0·60-1·23; p=0·94). Other study characteristics (design, population, comparison group, control variables, time of exposure assessment, biochemical verification of abstinence, and definition of e-cigarette use) were also not associated with the overall effect size (p≥0·77 in all cases).Interpretation
As currently being used, e-cigarettes are associated with significantly less quitting among smokers.Funding
National Institutes of Health, National Cancer Institute, FDA Center for Tobacco Products.Free full text

E-cigarettes and smoking cessation in real-world and clinical settings: a systematic review and meta-analysis
Summary
Background
Smokers increasingly use e-cigarettes for many reasons, including attempts to quit combustible cigarettes and to use nicotine where smoking is prohibited. We aimed to assess the association between e-cigarette use and cigarette smoking cessation among adult cigarette smokers, irrespective of their motivation for using e-cigarettes.
Methods
PubMed and Web of Science were searched between April 27, 2015, and June 17, 2015. Data extracted included study location, design, population, definition and prevalence of e-cigarette use, comparison group (if applicable), cigarette consumption, level of nicotine dependence, other confounders, definition of quitting smoking, and odds of quitting smoking. The primary endpoint was cigarette smoking cessation. Odds of smoking cessation among smokers using e-cigarettes compared with smokers not using e-cigarettes were assessed using a random effects meta-analysis. A modification of the ACROBAT-NRSI tool and the Cochrane Risk of Bias Tool were used to assess bias. This meta-analysis is registered with PROSPERO (number CRD42015020382).
Findings
38 studies (of 577 studies identified) were included in the systematic review; all 20 studies with control groups (15 cohort studies, three cross-sectional studies, and two clinical trials) were included in random effects meta-analysis and sensitivity analyses. Odds of quitting cigarettes were 28% lower in those who used e-cigarettes compared with those who did not use e-cigarettes (odds ratio [OR] 0·72, 95% CI 0·57–0·91). Association of e-cigarette use with quitting did not significantly differ among studies of all smokers using e-cigarettes (irrespective of interest in quitting cigarettes) compared with studies of only smokers interested in cigarette cessation (OR 0·63, 95% CI 0·45–0·86 vs 0·86, 0·60–1·23; p=0·94). Other study characteristics (design, population, comparison group, control variables, time of exposure assessment, biochemical verification of abstinence, and definition of e-cigarette use) were also not associated with the overall effect size (p≥0·77 in all cases).
Interpretation
As currently being used, e-cigarettes are associated with significantly less quitting among smokers.
Introduction
E-cigarettes (also known as electronic cigarettes, electronic nicotine delivery systems, vapour pens, and many other terms) are battery-powered devices that heat a solution of humectants (usually propylene glycol or glycerol), nicotine (in most cases), and flavourings (in many cases), to deliver an aerosol that is inhaled by the user. E-cigarette use is increasing in many countries.1,2 Adults report various motivations for e-cigarette use, including to help them quit cigarettes and allowing them to continue to use nicotine in areas where smoking is prohibited,3–7 which are common themes in e-cigarette marketing and promotion.8–10
In 2015, the US Preventive Services Task Force concluded that evidence was insufficient to recommend e-cigarettes for tobacco cessation in adults because of conflicting and limited evidence available at the time the recommendation was prepared.11 Two meta-analyses of combined results from clinical trials have assessed whether e-cigarette use is associated with smoking cessation.12,13 The first,12 based on two randomised trials,14,15 concluded that participants using nicotine e-cigarettes were more likely to have abstained from smoking cigarettes for 6 months (relative risk 2·29, 95% CI 1·05–4·96) than were participants using no-nicotine e-cigarettes, although the authors had little confidence in the results because of the small number of trials and small sample sizes. The second,13 based on six studies (the same two randomised trials,14,15 two cohort studies,16,17 and two cross-sectional studies18,19) found the proportion of individuals using nicotine-containing e-cigarettes who quit cigarettes to be 20% (95% CI 11–28). These meta-analyses did not compare e-cigarette users to a control group not using e-cigarettes. A third meta-analysis20 of five population-level studies (four longitudinal21–24 and one cross-sectional25) found that e-cigarette use was associated with a significant depression in smoking cessation (odds ratio [OR] 0·61, 95% CI 0·50–0·75).
The different results of the meta-analyses of clinical trials and observational studies may relate to discrepancies in how e-cigarettes are used in a controlled study setting versus in the real world. Clinical trials evaluating a treatment or intervention under ideal conditions may differ from observational studies evaluating how a product is actually used in a real-world setting in study design, study population, study environment,26 and ability to control for potential confounders, which can compromise the generalisability of results of observational studies.27 These differences are potentially important for e-cigarettes, which, unlike prescription-only nicotine inhalers, are mass-marketed consumer products. We conducted a systematic review and meta-analysis of clinical trials and observational real-world studies to assess the association between e-cigarettes (as available and used) and cigarette smoking cessation among adults, including all smokers as well as only those interested in quitting smoking.
Methods
Data sources and searches
To identify studies, we began by manually searching the references from the three earlier meta-analyses12,13,20 and completed a comprehensive literature search of PubMed and the Web of Science Core Collection between April 27, 2015, and June 17, 2015. As detailed in the appendix, search terms included “electronic cigarette”, “e-cigarette”, “electronic nicotine delivery”, “stop”, “quit”, “cessation”, “abstain”, and “abstinence”. Search results were not limited by language, but all identified studies were in English. There was no search limitation on publication dates. We continued to monitor the scientific literature after completing the formal search; this report includes two studies that were published while it was in initial peer review.28,29 Both abstracts and full manuscripts were considered.
Study selection
One investigator (SK) did the search, data extraction, and risk of bias assessment, which was subsequently reviewed by a second investigator (SAG). Clinical trials, whether randomised and controlled or not, cohort studies, and cross-sectional studies were all considered. We included studies that evaluated the relationship between e-cigarette use and cigarette smoking cessation among adult cigarette smokers; therefore, two studies on adolescents were excluded.30,31 We considered all study populations that were defined as “adult” by the study authors (youngest age varied from 15 to 30 years in the studies that defined “adult”; detailed descriptions of each article are provided in the appendix). We included studies of participants who were interested in quitting cigarette smoking and studies of all smokers irrespective of interest in quitting. We excluded one cross-sectional study25 because the primary outcome was e-cigarette use, not smoking cessation.
Data extraction
Studies that included cigarette smoking cessation as a primary outcome were evaluated for inclusion. The definitions of cigarette smoking cessation included in this systematic review and meta-analysis included both self-reported abstinence from smoking cigarettes and biochemically-validated measures of abstinence (eg, cotinine or exhaled carbon monoxide measurements). All studies were included irrespective of the duration of abstinence from cigarettes. Those who quit cigarettes could have still been using e-cigarettes; quitting e-cigarettes was not used as an outcome.
Data extracted from each study included study location, design, population, definition and prevalence of e-cigarette use, comparison group (if applicable), cigarette consumption, level of nicotine dependence, other confounders measured, definition of quitting smoking, and odds of quitting smoking. We attempted to contact study investigators for missing information. Risk of bias was assessed using a modification of the ACROBAT-NRSI tool32 for observational studies and the Cochrane Risk of Bias Tool33 for clinical trials, implemented as detailed in the appendix.
Data synthesis
For studies comparing nicotine e-cigarettes with nicotine replacement therapy (NRT), no-nicotine e-cigarettes, or no cessation aid, all ORs are presented; when several ORs were provided, we included only the comparison with no e-cigarette use (when available) or no cessation aid in the meta-analysis. Adjusted ORs were used when available, otherwise unadjusted ORs were used. For one cohort study,7 two different ORs were reported for e-cigarette users of different intensities and a pooled estimate was not provided; the ORs for those two groups are presented separately.
Statistical analysis
We computed pooled estimates of the odds of smoking cessation among smokers using e-cigarettes compared with smokers not using e-cigarettes using a random effects meta-analysis with the metan command in Stata version 13.0. Adjusted ORs were used when available, with unadjusted ORs for the remaining studies in the meta-analysis. Statistical heterogeneity was assessed using the I2 statistic.
We did a sensitivity analysis of the effects of study type (real world vs clinical), longitudinal versus cross-sectional data analysis, sample frame (smokers interested in quitting vs all smokers), control group (NRT users vs all no-e-cigarette users), study population (mental illness or no mental illness), whether the study controlled for level of nicotine dependence, time of e-cigarette assessment (whether e-cigarette use was assessed at baseline or follow-up in longitudinal studies), whether abstinence was biochemically defined, and whether the definition of e-cigarette use was current (past 30 day) use versus ever-use or not within the past 30 days on the results using separate random effects meta-regressions with each factor entered as a dummy variable with the Stata metareg command. We considered the nine sensitivity analyses to be a family of comparisons and controlled for multiple comparisons using the Holm-Sidak method to obtain adjusted p values. We tested for the presence of publication bias using a funnel plot and Egger’s test.
The meta-analysis was registered with PROSPERO on May 11, 2015, number CRD42015020382.
Role of the funding source
The funders had no role in the study design, collection, analysis, or interpretation of the data, or writing of the report. Both authors had access to the raw data. Both authors had full access to all of the data the study and had final responsibility for the decision to submit for publication.
Results
Of 577 studies identified, 38 were included in the systematic review (appendix) and 20 (table 1) in the meta-analysis. Studies excluded from the systematic review did not include smoking cessation as an outcome, did not include adults as the study population, were opinion pieces or commentaries, or were review articles (figure 1). Of the 38 studies included in the systematic review, 16 were excluded from the meta-analysis because they lacked a control group that did not use e-cigarettes,15–17,19,45–56 and two57,58 were excluded because they used the same dataset as another study included in the meta-analysis.14,38 15 of the studies included in the meta-analysis were longitudinal cohort studies (ten assessed e-cigarette use at baseline, five assessed e-cigarette use only at follow-up), three were cross-sectional studies, and two were clinical trials.
Table 1
Summary of studies on the relationship between e-cigarette use and cigarette smoking
| Location | Years | Population | Prevalence and definition of e-cigarette use | Comparison group | Confounders
| Definition of quitting | Odds of quitting (95% CI) | ||
|---|---|---|---|---|---|---|---|---|---|
| Level of dependence | Other | ||||||||
| Cohort studies of real-world use of e-cigarettes, e-cigarette use assessed at baseline
| |||||||||
| Borderud et al (2014)34 | USA | 2012–13 and follow-up 6–12 months later | 1074 smokers with cancer in a tobacco treatment programme | 285 (27%) reported any e-cigarette use in the past 30 days at enrolment | No e-cigarette use in the past 30 days at baseline | Fagerstrom Test for Nicotine Dependence; e-cigarette users: mean 4·6 (SD 2·7); non-e-cigarette users: 3·3 (2·7) | Past quit attempts, cancer diagnosis | Self-reported 7 day cigarette abstinence | 1·0 (0·6–2·0), adjusted; ITT analysis: 0·5 (0·3–0·8), adjusted |
| Choi et al (2014)21 | USA | 2010–11 and follow-up 1 year later | 346 young adult smokers in Midwestern USA | Used e-cigarettes >1 day in the past 30 days at baseline | Never e-cigarette use | ·· | Demographics and baseline cigarette consumption | Self-reported 30 day cigarette abstinence | 0·93 (0·19, 4·63), adjusted |
| Grana et al (2014)22 | USA | 2011 and follow-up 1 year later | 949 current adult smokers | 88 (9%) reported using e-cigarettes in the past 30 days (even once) at baseline | No e-cigarette use in the past 30 days (at baseline) | <30 min to first cigarette smoked: (69%) baseline e-cigarette users, (58%) baseline non-e-cigarette users | Intention to quit, cigarette consumption | Response of “Yes, I do not smoke now” to the question “Have you ever tried to quit smoking?” and reporting no past 30 day e-cigarette use | 0·76 (0·36–1·60), adjusted |
| Prochaska et al (2014)35 | USA | 2009–13 | 956 adult daily smokers (at least five cigarettes per day) with serious mental illness in the San Francisco Bay Area (part of a randomised controlled trial) | Reported e-cigarette use when asked about “other tobacco use”: 0/35 (0%) participants in 2009, 5/348 (1%) in 2010, 21/225 (9%) in 2011, 38/202 (19%) in 2012, and 37/146 (25%) in 2013 | Non-e-cigarette users | 78·5% smoked within 30 min of waking | Demographics, study factors, psychiatric variables, and tobacco-related variables | Past 7 day tobacco abstinence (self-report and biochemical verification) | 1·16 (0·65–2·05),* adjusted |
| Al-Delaimy et al (2015)36 | USA | 2011–12 and follow-up 1 year later | 1000 current adult smokers in California aged 18–59 years; 368 individuals used in final analysis | 236 (24%) reported they “have used e-cigarettes” at baseline | “Will never use e-cigarettes” at baseline and follow-up | <30 min to first cigarette smoked: 595 (61%) among entire study sample | Demographics and intention to quit | Self-reported 30 day cigarette abstinence | 0·41 (0·18–0·93), adjusted |
| Harrington et al (2015)37 | USA | 2012–13 (baseline and 6 month follow-up) | 979 hospitalised cigarette smokers (825 with follow-up) at one hospital | 171 (21%) reported current e-cigarette use at baseline, 247 (30%) reported e-cigarette use at follow-up (98 [12%] at both times) | Did not use e-cigarettes at baseline | ·· | ·· | Those who reported quitting smoking | 0·90 (0·54–1·50),† unadjusted |
| Hitchman et al (2015)38 | UK | 2012 and follow-up in 2013 | 1643 current adult cigarette smokers | 348 (21%) reported any e-cigarette use at baseline, 587 (36%) at follow-up | Never e-cigarette use at baseline | Strength of urges to smoke (5 point scale from no urges to extremely strong urges) at initial assessment (0: 126 [8%], 1: 212 [13%], 2: 764 [47%], 3: 387 [24%], 4: 105 [6%], 5: 49 [3%]) | Motivation to stop smoking at baseline and demographics | Those who reported that they “do not smoke cigarettes at all” or “stopped smoking completely” | 0·83 (0·52–1·30), adjusted |
| Manzoli et al (2015)39 | Italy | 2013 with follow-up 1 year later | 236 e-cigarette only users, 491 cigarette smokers, and 232 dual users of cigarettes and e-cigarettes | 232 dual users of cigarettes and e-cigarettes (used cigarettes and e-cigarettes within the same week for past 6 months); 81 dual users continued using e-cigarettes at 1 year | Cigarette smokers not using e-cigarettes | ·· | Demographics, body-mass index, alcohol use, self-rated health, medical comorbidities, and years of tobacco smoking | Self-reported past 30 day smoking abstinence (25% verified with CO) | 0·83 (0·53–1·29), adjusted |
| Pavlov et al (2015)40 | Canada | Not available (baseline and 3 month follow-up) | 3073 cigarette smokers enrolled in a smoking cessation programme at a primary care clinic in Toronto, CA | 363 (12%) did not use e-cigarettes at enrolment but began using e-cigarettes during the programme | Participants who did not start using e-cigarettes during the programme | Heaviness of smoking index (unclear if adjusted for) | Education, employment, income, quit confidence, and motivation to quit (unclear if adjusted for) | Definition unclear | 0·68 (0·54–0·87) unclear if adjusted |
| Sutfin et al (2015)41 | USA | 2010–13 | 271 college students in North Carolina and Virginia who smoked cigarettes and had not tried an e-cigarette at baseline | 118 (44%) had ever tried e-cigarettes by study wave 5 (none at baseline) | Did not use e-cigarettes during study | ·· | Baseline smoking frequency, lifetime other tobacco use, demographics, membership in Greek organisation,‡ sensation seeking at baseline, and number of friends and family who smoke | Did not continue to smoke at study wave 6 (derived from inverse of those who continued to smoke) | 0·40 (0·21–0·76),† adjusted |
|
| |||||||||
| Cohort studies of real-world use of e-cigarettes, e-cigarette use assessed at follow-up | |||||||||
|
| |||||||||
| Adkison et al (2013)23 | Canada, USA, UK, Australia | 2008–09 and follow-up 1 year later | 5939 adults age >18 years who were current or former cigarette smokers | 3% reported currently using an e-cigarette | Current non-users of e-cigarettes at follow-up | ·· | ·· | Self-reported quitting since previous study wave | 0·81 (0·43–1·53),* unadjusted |
| Vickerman et al (2013)24 | USA | 2011–12 | 2758 adult tobacco quitline callers age >18 years in six US states surveyed 7 months after enrolment | 765 (31%) had ever used e-cigarettes | Never use of e-cigarettes | ·· | ·· | Self-reported 30 day tobacco abstinence at 7 month follow-up | 0.50 (0·40–0·63),† unadjusted |
| Pearson et al (2014)42 | USA | 2012–13, and 3 month follow-up | 2123 current smokers in the USA participating in a web-based smoking cessation trial | 672 (32%) reported using an e-cigarette to quit smoking cigarettes in the past 3 months | No e-cigarette use in the past 3 months | Fagerstrom score: e-cigarette for cessation, median 6 (IQR 4–7); no e-cigarette for cessation, 5 (4–7) | Demographics, quitting-related variables, other quit methods | Self-reported 30 day abstinence from cigarettes | 0·68 (0·53–0·87), adjusting for all factors but other quit methods; 0·77 (0·59–1·00), adjusting also for other quit methods |
| Biener et al (2015)7 | USA | 2011–12, and follow-up in 2014 | 695 adult smokers in two US metropolitan areas (Texas and Indiana) | 111 (23%) were intensive e-cigarette users (defined as daily e-cigarette use for at least 1 month), 220 (29%) were intermittent e-cigarette users (defined as regular but not daily use for more than 1 month), and 364 (48%) were either non-users of e-cigarette or had tried e-cigarettes no more than two times (self-trial) | Never used e-cigarettes or tried e-cigarettes no more than two times | Heavy smoker (>20 cigarettes per day and first cigarette <30 min of waking): 21% (95% CI 11–34) in non-users/self-trial; 28% (11–54) in intermittent users; 68% (40–87) in intensive users | Demographics | Self-reported 30 day abstinence from cigarettes | Intensive users: 6·07 (1·11–33·18), adjusted; intermittent users: 0·31 (0·04–2·80), adjusted |
| Shi et al (2015)43 | USA | 2010, and follow-up 1 year later | 2454 US cigarette smokers in the 2010 Tobacco Use Supplement to the Current Population Survey | 279 (11%) reported having ever used e-cigarettes | No smoking cessation aid use | Baseline cigarette dependence level assessed and controlled for in analysis | Demographics | Cigarette abstinence for >30 days | 0·44 (0·24–0·79), adjusted |
|
| |||||||||
| Cross-sectional studies of real-world use of e-cigarettes | |||||||||
|
| |||||||||
| Brown et al (2014)18 | UK | 2009–14 | 5863 adult smokers in England in 2009–14 as part of the Smoking Toolkit Study | 489 (8%) answered e-cigarettes to which “of the following did you try to help you stop smoking during the most recent serious quit attempt?” | Over the counter NRT or no aid | Strength of urges to smoke (0–5 score): e-cigarettes, mean 2·0 (SD 1·2); NRT, 2·2 (1·1); no aid, 1·8 (1·1); time spent with urges to smoke (0–5); e-cigarettes, 1·9 (1·3); NRT, 2·2 (1·3); no aid, 1·8 (1·3) | Demographics, time since start of quit attempt, past year quit attempts, abrupt vs gradual quitting, year, and interaction terms | Those who answered “I am still not smoking” when asked “how long did your most recent serious quit attempt last before you went back to smoking” | 1·63 (1·17–2·27) compared with NRT, adjusted; 1·61 (1·19–2·18) compared with no aid, adjusted |
| Christensen et al (2014)44 | USA | 2012–13 | 9656 adults via the telephone-based Kansas Adult Tobacco Survey | 12% had ever used an e-cigarette; 3% had used an e-cigarette at least once in the past month | Never tried e-cigarettes or currently do not use e-cigarettes | ·· | Demographics | Self-reported cigarette abstinence | Ever e-cigarette use: 0·43 (0·24–0·79), adjusted; current e-cigarette use: 0·16 (0·07–0·36), adjusted |
| McQueen et al (2015)28 | USA | 2014 | 106 patients with head and neck cancer at an otolaryngology clinic in Alabama aged >19 years who were current or past daily tobacco users | 23 (22%) reported using e-cigarettes as part of their quit programme | Did not use e-cigarettes as part of their quit programme | ·· | ·· | Self-reported 30 day abstinence from all tobacco products, including e-cigarettes | 0·25 (0·09–0·65),† unadjusted |
|
| |||||||||
| Randomised clinical trials with control groups | |||||||||
|
| |||||||||
| Bullen et al (2013)14 | New Zealand | 2011–13 | 657 adult cigarette smokers interested in quitting, recruited through newspaper advertising | Intervention arm: 16 mg nicotine e-cigarettes | 21 mg nicotine patches (n=295), no-nicotine e-cigarettes (n=73) | Fagerstrom mean 5·6 (SD 2) in e-cigarette group, 5·5 (2) in patch group, 5·5 (2) in no-nicotine e-cigarette group | Randomised groups with similar demographics, age started smoking, years smoking, type of tobacco smoked, past year quit attempt, living with smokers, smoking behavioural questionnaire, autonomy over smoking scale, self-efficacy to quit | Smoking abstinence 6 months after quit day, defined as self-reported abstinence of the follow-up period with <5 cigarettes total, verified by exhaled carbon monoxide | RR 1·77 (0·54-5·77) compared with non-nicotine e-cigarette; unadjusted; RR 1·26 (0·68–2·34) compared with patches, unadjusted |
|
| |||||||||
| Non-randomised clinical trials with control groups | |||||||||
|
| |||||||||
| Hajek et al (2015)29 | UK | 2015 | 100 smokers accessing Stop Smoking Services | 69 (69%) accepted e-cigarettes as part of their smoking cessation treatment | Did not accept e-cigarettes | ·· | ·· | Self-reported abstinence from cigarettes at 4 weeks, biochemically verified by exhaled carbon monoxide | RR 1·44 (0·94–2·21),† unadjusted |
ORs were adjusted for all factors in the confounders column except as noted. ORs provided for observational studies, RRs provided for randomised trials. OR=odds ratio. NRT=nicotine replacement therapy. RR=relative risk.
Odds of quitting (only provided for studies with a control group) are described with point estimates and 95% CIs and whether these point estimates differed significantly from 1·00.
15 longitudinal real-world studies assessing smoking cessation in e-cigarette users compared with those who did not use e-cigarettes.7,21–24,34–43 Point estimates in 13 of the 15 studies indicated decreased smoking cessation among those who used e-cigarettes, six of which reported statistically significant results.24,34,36,40,41,43 One study7 found that intensive e-cigarette users (those who used e-cigarettes daily for at least 1 month) had significantly increased smoking cessation and that intermittent e-cigarette users had a non-significant decrease in smoking cessation. In three studies without control groups (excluded from the meta-analysis),16,45,46 smoking cessation rates among e-cigarette users ranged from 17% at 8 weeks,45 to 41% at 1 year,46 to 46% at 1 year.16
Three cross-sectional studies compared e-cigarette users to those who did not use e-cigarettes:18,28,44 two28,44 showed significantly lower smoking cessation among smokers using e-cigarettes compared with those who did not, and the other study18 (of smokers who had made a quit attempt in the past year) found significantly higher smoking cessation in those who used e-cigarettes compared with those who used NRT or no smoking cessation aid. Cigarette quit rates in cross-sectional studies that included only e-cigarette users (ie, studies without control groups, which were excluded from the meta-analysis) ranged from 10% to 66%.19,47–49
The one randomised controlled trial comparing cigarette quit rates of e-cigarette users with those of NRT users showed a non-significant increase in quitting associated with e-cigarette use.14 A secondary analysis of a subset of participants with mental illness in this study showed a non-significant decrease in quitting among those who used e-cigarettes compared with those who used NRT.58 A non-randomised clinical trial found a non-significant increase in quitting among individuals electing to use e-cigarettes for smoking cessation compared with those not using e-cigarettes.29 For trials without control groups17,50–56 (ie, all participants used e-cigarettes), cigarette quit rates ranged from 12·5% to more than 40%. A randomised trial comparing users of e-cigarettes with and without nicotine (without a control group of non-e-cigarette users or conventional smoking cessation therapy) found a non-significant increase in quitting cigarettes for those using nicotine-containing e-cigarettes compared with those using non-nicotine e-cigarettes.15
Combining the 18 real-world studies7,18,21–24,28,34–44 (treating one study7 with estimates for two types of e-cigarette users as two studies, yielded 19 real-world estimates of the relationship between e-cigarette use and quitting smoking) and the two clinical trials14,29 in a random effects meta-analysis (figures 2, ,3)3) indicated that the odds of quitting smoking were 28% lower in those who used e-cigarettes compared with those who did not use e-cigarettes (OR 0·72, 95% CI 0·57–0·91).
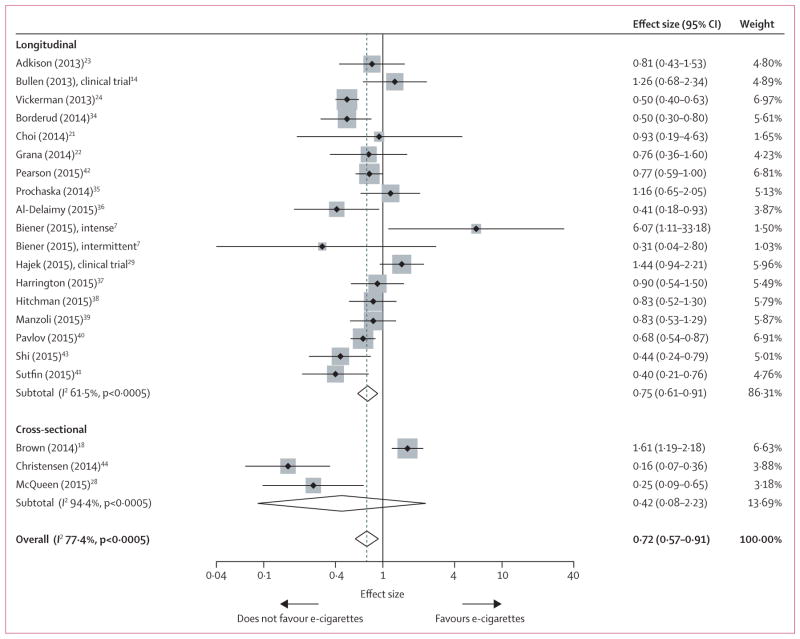
Figure shows odds of quitting among e-cigarette users compared with non-e-cigarette users. The overall odds of quitting cigarettes is 0·72 (95% CI 0·57–0·91) irrespective of how studies are stratified.
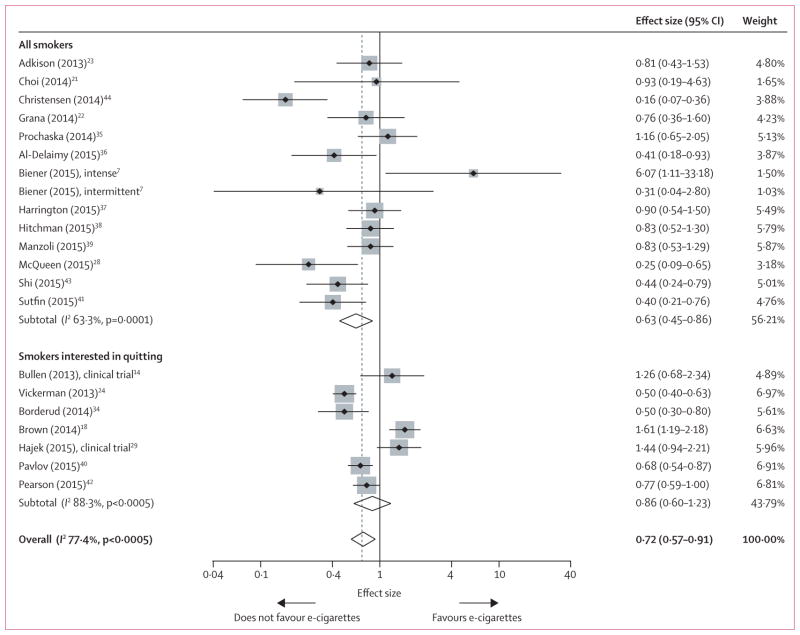
Figure shows odds of quitting among e-cigarette users compared with non-e-cigarette users. The overall odds of quitting cigarettes is 0·72 (95% CI 0·57–0·91) irrespective of how studies are stratified.
Studies that included only smokers interested in quitting cigarettes yielded a pooled OR for quitting of 0·86 (0·60–1·23) for those using e-cigarettes compared with those not using e-cigarettes. Studies of all smokers (irrespective of motivation to quit) yielded a pooled OR of 0·63 (0·45–0·86), which is not significantly different from studies limited to smokers interested in quitting (p=0·94).
All of the observational studies had low risk of selection bias, half (nine of 18) controlled for confounders, and seven of 15 longitudinal studies had follow-up periods of at least 6 months (appendix). The overall risk of bias from exposure measurement was unclear, given that the definition of e-cigarette use in all but two7,39 of the studies could have included people who only used e-cigarettes once; risk of bias from outcome assessment was unclear or high due to objective or poorly defined measurements.
The one randomised clinical trial14 had a low risk of selection, detection, and reporting bias, but a high risk of performance and attrition bias, because participants randomly assigned to e-cigarettes were provided with the e-cigarettes, whereas the individuals randomly assigned to nicotine patches were provided a voucher that they could take to a pharmacy to obtain the patches; additionally, the nicotine patch group had a higher loss to follow-up than did the e-cigarette group (appendix). The non-randomised controlled trial29 had low risk of detection, attrition, and reporting bias, but high risk of selection and performance bias because participants were not randomised and had chosen to use e-cigarettes.
We did not find evidence of publication bias by Egger’s test (p=0·91) or by visualisation of the funnel plot (appendix).
There was evidence of heterogeneity of the studies (I2 77·4%, p<0·0005). In particular, heterogeneity was higher for the cross-sectional studies (I2 94·4%, p<0·0005) than the longitudinal studies (I2 61·5%, p<0·0005). In addition to using a random effects model to control for heterogeneity, we assessed the possible reasons for heterogeneity in a sensitivity analysis (table 2). In the sensitivity analyses, sample frame, study type (longitudinal vs cross-sectional data analysis), control group, study population, level of nicotine dependence, time of e-cigarette assessment (in longitudinal studies), biochemical verification of smoking cessation, and definition of e-cigarette use were not associated with overall effect size (p≥0·77 in all cases).
Table 2
Sensitivity analysis
| n | OR (95% CI) | p value* | |
|---|---|---|---|
| Sample frame | 0·94 | ||
 All smokers All smokers | 14 | 0·63 (0·45–0·86) | |
 Smokers interested in quitting cigarettes Smokers interested in quitting cigarettes | 7 | 0·86 (0·60–1·23) | |
|
| |||
| Study type | 0·90 | ||
 Real world Real world | 19 | 0·67 (0·52–0·85) | |
 Clinical trial Clinical trial | 2 | 1·38 (0·97–1·96) | |
|
| |||
| Longitudinal vs cross-sectional | 0·98 | ||
 Longitudinal Longitudinal | 18 | 0·75 (0·61–0·91) | |
 Cross-sectional Cross-sectional | 3 | 0·42 (0·08–2·23) | |
|
| |||
| Control group for study | 0·90 | ||
 All non-e-cigarette users All non-e-cigarette users | 20 | 0·70 (0·55–0·89) | |
 NRT NRT | 1 | 1·26 (0·68–2·34) | |
|
| |||
| Study population | 0·89 | ||
 All individuals All individuals | 20 | 0·70 (0·55–0·89) | |
 Individuals with mental illness Individuals with mental illness | 1 | 1·16 (0·65–2·05) | |
|
| |||
| Controlling for level of nicotine dependence | 0·79 | ||
 Controlled for dependence Controlled for dependence | 11 | 0·84 (0·60–1·17) | |
 Did not control for dependence Did not control for dependence | 10 | 0·62 (0·45–0·85) | |
|
| |||
| Time of e-cigarette assessment in cohort studies | 0·93 | ||
 Baseline Baseline | 10 | 0·71 (0·59–0·85) | |
 Follow-up Follow-up | 6 | 0·66 (0·45–0·95) | |
|
| |||
| Biochemical verification of abstinence | 0·77 | ||
 Biochemical verification Biochemical verification | 4 | 1·14 (0·88–1·48) | |
 No biochemical verification No biochemical verification | 17 | 0·63 (0·49–0·82) | |
|
| |||
| Definition of e-cigarette use | 0·87 | ||
 Current or past 30 days Current or past 30 days | 11 | 0·80 (0·54–1·17) | |
 Ever use or not within the past 30 days Ever use or not within the past 30 days | 10 | 0·66 (0·49–0·90) | |
OR=odds ratio. NRT=nicotine replacement therapy.
Discussion
In this systematic review and meta-analysis, pooled results from 18 real-world observational studies and two clinical trials showed 28% (OR 0·72, 95% CI 0·57–0·91) lower odds of cigarette smoking cessation among those who used or had used e-cigarettes compared with those who had not used e-cigarettes. This conclusion was insensitive to a wide range of study design factors, including whether or not the study population consisted only of smokers interested in smoking cessation, or all smokers (irrespective of quit intention).
The results of this meta-analysis are consistent with those of the previous meta-analysis of five real-world studies,20 which found a pooled OR for quitting of 0·61 (95% CI 0·50–0·75) for those who used e-cigarettes compared with those who did not use e-cigarettes.
By contrast, two previous meta-analyses,12,13 which both included the same two clinical trials to compare nicotine e-cigarette users with non-nicotine e-cigarette users,14,15 showed the odds of quitting cigarettes to be twice as high in those using e-cigarettes with nicotine compared with those using e-cigarette without nicotine (2·29, 95% CI 1·05–4·96).12 One of the previous meta-analyses,13 based on six studies, found that 20% (95% CI 11–28) of users of nicotine-containing e-cigarettes went on to quit cigarettes. By contrast with our analysis, the estimates did not include a comparison to standard therapy or no e-cigarette use, so they cannot be used to determine whether e-cigarettes are associated with greater cigarette abstinence than current practice.
So far, no clinical trials have done a true head-to-head comparison of e-cigarettes with standard therapies (ie, nicotine patch, gum, or inhaler) approved by the US Food and Drug Administration for smoking cessation. The one randomised clinical trial included in this meta-analysis14 compared e-cigarettes with nicotine patches, but was subject to performance bias because individuals randomly assigned to e-cigarettes were provided with them by the investigators, whereas participants randomly assigned to NRT were given only a voucher that they could redeem at a pharmacy to obtain NRT patches. Although this practice is consistent with standard care in the country in which the study was done (New Zealand), it potentially biases the study against NRT. As a result, true head-to-head comparisons of e-cigarettes with approved therapies in a clinical setting are needed to evaluate the usefulness of e-cigarettes for smoking cessation. In the USA, such studies require Investigational New Drug approval from the Food and Drug Administration Center for Drug Evaluation and Research. If e-cigarette companies do not request this approval (as has been the case to date), such studies will be difficult, if not impossible, to conduct, unless at the request of the companies.
Heterogeneity was higher for the cross-sectional studies (I2 94·4%, p<0·0005) than the longitudinal studies (I2 61·5%, p<0·0005). This increased heterogeneity might result from the fact that there were only three cross-sectional studies (compared with 18 longitudinal estimates), and that they differed in terms of countries (one in the UK18 and two in the USA28,44), study population (two only of smokers interested in quitting,18,28 one of all smokers44), comparator group (one compared e-cigarettes with NRT18 and the other two compared with no e-cigarette use28,44), and adjustment for nicotine dependence (one18 was adjusted, two28,44 were not). There was no evidence of study type (longitudinal vs cross-sectional) being associated with the overall effect size.
There could be a number of explanations for why e-cigarette use was associated with less quitting in this meta-analysis combining observational and clinical studies. When considering e-cigarettes as a potential smoking cessation aid, the fact that they are freely available consumer products could be important. The situation may be similar to the differences between the clinical efficacy of approved NRT therapies for smoking cessation in clinical trials versus actual use in non-clinical settings. A few studies have evaluated the association of other forms of over-the-counter nicotine with smoking cessation. Data from the large population-based California Tobacco Surveys, showed that NRT was associated with long-term success in quitting cigarettes when available by prescription only, but this association was lost when NRT became available over the counter.59 In a prospective cohort study of adult smokers in England, prescription medication combined with behavioural counselling was associated with increased smoking cessation, whereas over-the-counter NRT was associated with a significant reduction in smoking cessation (OR 0·68, 95% CI 0·49–0·94) that was similar to the association between e-cigarette use and quitting smoking that we observed.60
In observational studies evaluating e-cigarette use, participants are choosing to use e-cigarettes unlike clinical trials where they are being randomly assigned to receive them. From one perspective, this self-selection of product use by individuals would be a potential source of selection bias61 when evaluating e-cigarettes as a smoking cessation aid. However e-cigarettes are not being used just as smoking cessation devices in the real world. Indeed, one important motivation for using e-cigarettes is to self-administer nicotine in places where smoking is prohibited.3,62
Interest in quitting cigarette smoking is a common reason for using e-cigarettes,3,4 probably because claims of efficacy as a cessation aid have appeared in e-cigarette advertisements in the USA,9 UK,8 and China10 even though such claims had not been accepted by regulatory authorities. E-cigarettes are also marketed as a way to circumvent smoke-free policies,8,9 and could be used as such by nicotine-dependent individuals who have lower baseline intentions of quitting.
Subgroup analyses in two of the studies suggest that specific use patterns may be important. Biener and colleagues7 found that intermittent e-cigarette users (more than once or twice but less than daily use) were less likely to quit smoking one year later than non-e-cigarette users, but those who had used e-cigarettes daily for at least one month were significantly more likely to quit cigarettes. Similarly, Hitchman and colleagues38 found that all “cig-alike” users and non-daily tank system users had lower odds of quitting cigarettes, whereas daily tank system users were significantly more likely to quit. By contrast, Manzoli and colleagues39 found a non-significant (OR 0·83, 95% CI 0·53–1·29) reduction in quitting cigarettes among cigarette smokers who regularly used e-cigarettes (defined as using both cigarettes and e-cigarettes within the same week for 6 months) compared with cigarette-only smokers. These data suggest that e-cigarette use patterns might be important in the association between e-cigarettes and smoking cessation.
E-cigarette regulation has the potential to influence marketing and reasons for use. The inclusion of e-cigarettes in smoke-free laws and voluntary smoke-free policies could help decrease use of e-cigarettes as a cigarette substitute and, perhaps, increase their effectiveness for smoking cessation. The way e-cigarettes are available on the market—for use by anyone and for any purpose—creates a disconnect between the provision of e-cigarettes for cessation as part of a monitored clinical trial and the availability of e-cigarettes for use by the general population. Therefore, careful attention to how the products are marketed and actually used will be necessary in evaluating e-cigarettes as a smoking cessation aid and their ultimate public health impact.63
The studies we reviewed controlled for many confounding variables, including level of nicotine dependence, whether the subject was trying to quit smoking cigarettes, demographics, past quit attempts, medical and psychiatric comorbidities, cigarette consumption, intention or motivation to quit, study factors, tobacco-related variables and behaviours, alcohol use, sensation seeking, number of friends and family who smoke, use of other quit methods, and type of tobacco smoked (table 1). Future research should focus on determining standard definitions of e-cigarette use; evaluating the association of different extents of use and different devices with smoking cessation; conducting more randomised clinical trials comparing e-cigarettes to standard therapies such as NRT; evaluating the effect of e-cigarette use on factors such as motivation to quit; and distinguishing e-cigarette users by their reasons for using the products. An ideal study (whether a clinical trial or observational study) would control for all these variables, and be a longitudinal study that assessed e-cigarette use at both the start and end, including product type used and frequency of use. Additionally, having clear definitions of e-cigarette use (ever use, past 30 day use, whether experimentation with e-cigarettes once or twice are included) is important, as is how cessation is defined. It will, unfortunately, likely be difficult to obtain all these details in any one study.
This study is subject to eight limitations. First, given that there have been very few controlled clinical trials exploring e-cigarette use for smoking cessation; all but two of the studies used in this meta-analysis were observational studies. Second, the definition of smoking cessation used in the studies included in the meta-analysis varied in terms of length of abstinence from cigarettes and how smoking cessation was defined. Because we aimed for a comprehensive review, we included all definitions of quit as defined by the authors. Third, most—but not all—of the observational studies controlled for confounders such as nicotine dependence. Sensitivity analysis showed that controlling for nicotine dependence was not associated with overall effect size. Other potential confounding variables were controlled for in some of the studies, however, sensitivity analyses could not be conducted on each of these variables. Nevertheless, it is always possible that other unidentified confounders, including biases introduced by those who self-selected to use e-cigarettes, might affect the results. Fourth, only two studies7,39 assessed the extent of e-cigarette use in their analyses. It is possible that in some included studies, e-cigarettes were only used once, which would not be a good predictor of smoking cessation. Fifth, there was variability in the quality of the studies. We included them all to provide a comprehensive review of the literature and avoid concerns that the results of the analysis were affected by bias in selecting which studies to exclude and include. The sensitivity analysis showed that the results of the meta-analysis were insensitive to a wide range of aspects of study design. Sixth, in the cross-sectional studies, e-cigarette use and cessation were assessed at the same time, raising concerns about recall bias. Seventh, e-cigarettes are rapidly evolving products and different types and generations of e-cigarettes may have different effects in terms of cessation. Finally, both e-cigarette products and the marketing and regulatory environment are rapidly evolving, all of which could affect the relationship between e-cigarette use and quitting smoking.
As use patterns and product types continue to evolve, the association between e-cigarettes and cigarette quit rates may change. In the current regulatory environment, e-cigarette use is increasing and, although quitting smoking is a common marketing claim and is often cited as a reason for use among cigarette smokers, the overall conclusion from the available studies is that e-cigarette use is associated with reduced smoking cessation in the real world.
Acknowledgments
We thank Pamela M Ling, Margarete C Kulik, Lauren M Dutra, and I Elaine Allen (University of California San Francisco, CA, USA) for their review of and feedback on the manuscript. SK’s work was supported by NIH NRSA T32HP19025. SAG’s work reported in this publication was supported by grant 1P50CA180890 from the National Cancer Institute and FDA Center for Tobacco Products (CTP). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the Food and Drug Administration.
Funding: National Institutes of Health, National Cancer Institute, FDA Center for Tobacco Products.
Footnotes
Contributors
Both authors contributed to all aspects of this study, including conducting the literature search, study design, data collection, data analysis, data interpretation, and writing of the report.
Declaration of interests
SAG is Truth Initiative Foundation Distinguished Professor of Tobacco Control. We declare no competing interests.
References
Full text links
Read article at publisher's site: https://doi.org/10.1016/s2213-2600(15)00521-4
Read article for free, from open access legal sources, via Unpaywall:
https://europepmc.org/articles/pmc4752870?pdf=render
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics
Smart citations by scite.ai
Explore citation contexts and check if this article has been
supported or disputed.
https://scite.ai/reports/10.1016/s2213-2600(15)00521-4
Article citations
Changing patterns of cigarette and ENDS transitions in the USA: a multistate transition analysis of youth and adults in the PATH Study in 2015-2017 vs 2017-2019.
Tob Control, 33(5):570-579, 25 Sep 2024
Cited by: 5 articles | PMID: 36977570
Examining longitudinal associations between initial perceptions and experiences with electronic nicotine delivery system (ENDS) use and use patterns among adults who smoke and recently initiated ENDS.
Tob Induc Dis, 22, 27 Sep 2024
Cited by: 0 articles | PMID: 39345875
Knowledge, Attitude, and Practice of College Students Toward E-cigarettes: A Study From the Northern Part of Karnataka, India.
Cureus, 16(9):e68403, 01 Sep 2024
Cited by: 0 articles | PMID: 39360072
Pharmacokinetics and Pharmacodynamics of Inhaled Nicotine Salt and Free-Base Using an E-cigarette: A Randomized Crossover Study.
Nicotine Tob Res, 26(10):1313-1321, 01 Sep 2024
Cited by: 2 articles | PMID: 38597729
Acute exposure to electronic cigarette components alters mRNA expression of pre-osteoblasts.
FASEB J, 38(17):e70017, 01 Sep 2024
Cited by: 0 articles | PMID: 39213037
Go to all (475) article citations
Other citations
Data
Data behind the article
This data has been text mined from the article, or deposited into data resources.
BioStudies: supplemental material and supporting data
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Use of E-Cigarettes Among Current Smokers: Associations Among Reasons for Use, Quit Intentions, and Current Tobacco Use.
Nicotine Tob Res, 17(10):1228-1234, 14 Jan 2015
Cited by: 122 articles | PMID: 25589678 | PMCID: PMC4592339
Concurrent E-Cigarette Use During Tobacco Dependence Treatment in Primary Care Settings: Association With Smoking Cessation at Three and Six Months.
Nicotine Tob Res, 19(2):183-189, 31 Aug 2016
Cited by: 24 articles | PMID: 27613911
Reasons for quitting cigarette smoking and electronic cigarette use for cessation help.
Psychol Addict Behav, 29(1):114-121, 01 Sep 2014
Cited by: 12 articles | PMID: 25180551 | PMCID: PMC4511704
Electronic Cigarettes for Smoking Cessation: A Systematic Review.
Nicotine Tob Res, 18(10):1926-1936, 25 Apr 2016
Cited by: 158 articles | PMID: 27113014
Review
Funding
Funders who supported this work.
NCI NIH HHS (2)
Grant ID: P50 CA180890
Grant ID: 1P50CA180890
PHS HHS (1)
Grant ID: T32HP19025