Abstract
Epidemiological and basic research suggests that nonsteroidal anti-inflammatory drugs (NSAIDs) should protect against the most common forms of Alzheimer's disease (AD). Ibuprofen reduces amyloid (Aβ) pathology in some transgenic models, but the precise mechanisms remain unclear. Although some reports show select NSAIDs inhibit amyloid production in vitro, the possibility that in vivo suppression of amyloid pathology occurs independent of Aβ production has not been ruled out. We show that ibuprofen reduced Aβ brain levels in rats from exogenously infused Aβ in the absence of altered Aβ production. To determine whether ibuprofen inhibits pro-amyloidogenic factors, APPsw (Tg2576) mice were treated with ibuprofen for 6 months, and expression levels of the Aβ and inflammation-related molecules α1 antichymotrypsin (ACT), apoE, BACE1, and peroxisome proliferator-activated receptor γ) (PPARγ) were measured. Among these, ACT, a factor whose overexpression accelerates amyloid pathology, was reduced by ibuprofen both in vivo and in vitro. IL-1β, which was reduced in our animals by ibuprofen, induced mouse ACT in vitro. While some NSAIDs may inhibit Aβ42 production, these observations suggest that ibuprofen reduction of Aβ pathology may not be mediated by altered Aβ42 production. We present evidence supporting the hypothesis that ibuprofen-dependent amyloid reduction is mediated by inhibition of an alternate pathway (IL-1β and its downstream target ACT).
Similar content being viewed by others
INTRODUCTION
Although Alzheimer's disease (AD) is classified as a neurodegenerative rather than inflammatory disorder, the associated chronic inflammation of the brain is thought to play a role in pathogenesis and progression (Yasojima et al, 1999); see reviews (Akiyama et al, 2000; McGeer and McGeer, 2003). Although epidemiological data suggesting nonsteroidal anti-inflammatory drugs (NSAIDs) are protective against AD (Breitner et al, 1994; McGeer et al, 1996; Stewart et al, 1997; in t' Veld et al, 2001; Zandi et al, 2002) have been used to argue for the pathogenic significance of neuroinflammation in AD, it is not resolved whether the beneficial targets of NSAIDs are inflammation-related. While some studies argue for inflammation-independent pathways targeted by NSAIDs in AD prevention (Weggen et al, 2001), other studies support the argument that inflammatory responses (known to be induced by β-amyloid protein (Aβ), neurodegeneration, and Aβ-comprised neuritic plaques) may contribute to further neurodegeneration and induce additional amyloid pathology (Lim et al, 2000; Lim et al, 2001; Xiang et al, 2002).
We previously reported that ibuprofen treatment reduced amyloid pathology in the APPsw Tg2576 mice (Lim et al, 2000). Although ibuprofen has failed to impact plaques in some more aggressive models, others have confirmed ibuprofen reduction of plaques in APP transgenic (APP Tg) Alzheimer model mice (Jantzen et al, 2002; Yan et al, 2003). The major inflammatory target of NSAIDs, including ibuprofen, is cyclooxygenase (COX), and in our model ibuprofen-induced plaque reduction was associated with reductions in brain interleukin-1β, a downstream target of COX. Although this and other evidence suggests a possible role of inflammation in controlling plaque pathogenesis, this hypothesis has been challenged by discovery of an unexpected COX-independent function of a subset of NSAIDs (including ibuprofen) in reducing Aβ42 production in vitro (Weggen et al, 2001, 2003a, 2003b; Morihara et al, 2002; Eriksen et al, 2003; Sagi et al, 2003; Takahashi et al, 2003; Zhou et al, 2003). Adding to the controversy whether the mechanism(s) of plaque reduction are inflammation-dependent or -independent, it is unclear whether the high ibuprofen concentrations required in vitro to reduce Aβ42 production can be attained in the human brain with doses shown epidemiologically to reduce AD risk. Further, epidemiological studies suggest that some of the NSAIDs that appear to show AD risk reduction cannot reduce Aβ42. In summary, it remains uncertain whether the suppression of Aβ42 production by γ-secretase is the main mechanism for the protective effects of NSAIDs in the epidemiological studies and for the reduction of amyloid plaques in chronic NSAID-treated APP Tg mice.
An alternative possible NSAID mechanism for suppressing Aβ production is targeting a key regulator of Aβ production in the brain: BACE1, the main enzyme that cleaves APP at the β-secretase site. BACE1 deficiency abolishes Aβ production in BACE1 knockout mouse (Cai et al, 2001; Luo et al, 2001; Roberds et al, 2001) and in cell cultures (Cai et al, 2001; Roberds et al, 2001). BACE mRNA and protein levels and β-secretase activities were reported to be induced by a combination of proinflammatory cytokines and suppressed by NSAIDs and peroxisome proliferator-activated receptor gamma (PPARγ) agonist in vitro (Sastre et al, 2003).
Besides suppressing Aβ42 production, anti-inflammatory effects of NSAIDs, particularly those achieved with lower NSAID doses, could exert protection from AD by reducing amyloid deposition without altering Aβ42 production. Amyloid pathology can be affected by multiple inflammation-driven pathways, notably those involving acute phase proteins. Inflammation-related astrocyte molecules like apolipoprotein E (apoE) (Bales et al, 1997) and α1-antichymotrypsin (ACT) (Mucke et al, 2000; Nilsson et al, 2001) regulate amyloid pathology in APP Tg mice. ACT is an acute phase protein that accelerates amyloid pathology in APP Tg mice and is elevated in AD (Abraham et al, 1988; Pasternack et al, 1989; Koo et al, 1991; Shoji et al, 1991; Licastro et al, 1998), where it accumulates with amyloid plaques. ApoE, expressed primarily by astrocytes, can be induced during glial activation associated with cell damage or inflammation. ApoE has been shown to dose-dependently and isotype-specifically promote Aβ deposition in APP Tg mice (Bales et al, 1997; Fagan et al, 2002). Chronic proinflammatory LPS injection into APP Tg apoE wild-type mice has been reported to induce extensive GFAP-positive astrocytes and to accelerate amyloidosis, while LPS failed to increase amyloid deposition in APP Tg apoE-knockout mice (Qiao et al, 2001). Since ibuprofen treatment reduced GFAP in our APP trangenics (Lim et al, 2000), we reasoned that control of inflammation and gliosis might reduce apoE production. Although some reports show an association between increased apoE mRNA (or promoter polymorphisms promoting such increases) and AD risk or amyloid plaques (Diedrich et al, 1991; Yamada et al, 1995; Bullido et al, 1998; Lambert et al, 1998; Laws et al, 1999), other reports show that this relation is restricted to apoE4 cases (Lambert et al, 1997; Roks et al, 1998; Song et al, 1998; Pahnke et al, 2003).
Therefore in this study, we explored the potential of ibuprofen to alter post-amyloid production effects of NSAIDs using the Tg2576 mouse and in vitro model systems. We focused on the impact of chronic ibuprofen treatment on expression of a set of molecules related to both activated glia and amyloid pathology: BACE1, apoE, Il-1β, α1 ACT. We also evaluated the effect of ibuprofen on Aβ deposition from exogenously infused Aβ, independent of effects on production.
MATERIAL AND METHODS
Immunohistochemistry for Amyloid Burden in Aβ-infused Rats
Female SpragueâDawley rats (19 months old) were fed chow with (n=10) or without (n=10) ibuprofen (375âppm) for 60 days. They were then intracerebroventricularly infused bilaterally into the lateral ventricles with a mixture of Aβ40 (20âμg total) and Aβ42 (5âμg total) with HDL as carrier and 4âmM HEPES, pH 8.0 buffer for 30 days, as previously described (Frautschy et al, 2001). The 4â:â1 ratio used approximates the typical ratio of secreted Aβ40â:â42. Rats were then killed and brain sections prepared for histochemical analysis. Aβ deposits were labeled with 10G4 anti-Aβ antibody and detected by ABC ELITE-peroxidase kits (Vector) using metal diaminobenzidine (DAB, Pierce, Rockford, IL). Captured immunohistochemical images were analyzed by NIH Image software with custom macro subroutines, as previously described (Frautschy et al, 1999).
Mice for Chronic Ibuprofen Treatment
Tg2576 transgenic mice, 10-month-old huAPPsw line, were used. Transgene negative littermates (Tgâ; n=10) were fed control chow and transgene positive (Tg+) mice were fed control chow (n=12) or chow with 375âppm ibuprofen (Sigma St Louis, MO) for 6 months (n=8) or 3 months (n=4) as reported previously (Lim et al, 2000). Cortical tissue was snap frozen in liquid nitrogen and powderized for mRNA analysis. Half of the powdered tissue was used for protein analysis reported previously for Aβ and IL-1β (Lim et al, 2000) and half was analyzed for mRNA in this study. APPsw Tg+ (n=7) and Tgâ control mice (n=9), 22-month old, on control chow were also analyzed. Positive controls for verification of CNS inflammation were made by injecting LPS (6âμg) into the right lateral ventricle of Tgâ mice and killing after 24âh. The protocols using mice and rats were approved by the UCLA and VA IACUCs.
Real-Time PCR of mACT and apoE mRNA
Total RNA was prepared using RNAqueous kits (Ambion, Austin TX). The absence of RNA degradation was confirmed by gel electrophoresis and total RNA was DNase treated (DNA-free, Ambion). cDNA was generated from 1.4âμg total RNA in two reaction volumes of RETROscript (Ambion) using dT primer and then aliquoted for single uses. TaqMan real-time PCR assays for mACT and apoE mRNA were developed. Primers (Sigma; IDT) and fluorescent probes (Biosource, Camarillo CA; IDT, Coralville IA) were designed using Primer Express 1.1 (Applied Biosystems, Foster City CA). BACE1 and COX-2 was measured using Assay on Demand (Mm00478664_m1 and Mm00478374_m1, Applied Biosystems, Foster City, CA). PPARγ primer and probes were designed in Dr P Tontonoz's lab (UCLA). Sequences and optimized concentrations of primers and probes are shown in Table 1. The absence of nonspecific PCR was confirmed by gel electrophoresis using 45 cycle PCR products (data not shown). cDNA for relative standard curves was prepared from Tgâ mouse brain with the same protocols with double RNA concentration in the reverse transcript step. Real-time PCR was performed by SDS7700 (Applied Biosystems) and TaqMan Universal PCR Master Mix (Applied Biosystems), using triplicates for samples and quadruplicates for standards. GAPDH was measured as the internal control using TaqMan Rodent GAPDH control (Applied Biosystems). Consistent GAPDH levels confirm the absence of RNA and/or cDNA degradation (data not shown). The absence of contamination was checked using RT products made without M-MLV reverse transcriptase. The R2 of relative standard curves in TaqMan PCR was above 0.99. The R2 of mRNA levels from the same samples but measured on different days was around 0.9. In spite of the great reproducibility and use of the same five-point standards in every plate, around 5% difference in average values between different PCR plates with same samples was observed (data not shown). For optimum reliable comparisons, only sample values from the same PCR plate were statistically analyzed and shown here.
Multiplex RT-PCR of mACT mRNA
Murine ACT (EB22/4, mACT) was amplified by PCR along with β-actin in the same tubes. The sequences of primers are shown in Table 1. cDNA (1âμl) was amplified by 1.5âU of thermostable Taq Polymerase (#0032 003.416 Eppendorf, Hamburg Germany) in 20âμl reaction volumes (1âμCi 32P dCTP, 0.2âmM dNTPs, 50âmM KCl, 10âmM Tris-HCl pH 8.3, and 1.5âmM MgCl). Since the expression levels of β-actin mRNA were much higher than those of mACT mRNA, mACT PCR amplification was distorted in the same PCR tubes. To avoid this PCR interruption, mACT primers alone were added at the beginning of PCR and β-actin primers were added into the PCR tubes after nine cycles of PCR. The optimized total PCR cycles were 24 for ACT. The absence of PCR interactions and the expected exponential amplification of both products were confirmed during these optimizations (data not shown). PCR products were analyzed by acrylamide gel electrophoresis and exposed autoradiogram and quantified using Molecular Imager system (BioRad, Hercules, CA).
ApoE Western Blotting
Cortex was homogenized in TBS and then spun down at 55â000 RPM (100â000âg) for 20âmin at 4°C. The supernatant was used as TBS fraction. The pellet was solublized in 2% SDS and used as SDS fraction. Western blotting was performed using rabbit anti-rat apoE antibody R243 (gift of Dr Schonfeld, Wash. U., St Louis, MO, 1â:â1000) and anti-rabbit antibody conjugated to HRP (Pierce #31463, 1â:â10â000).
Human Astrocytoma U373 Cell Cuture and ACT Western Blotting
Human astrocytoma U373 cells were cultured to semiconfluence in MEM supplemented with 10% fetal bovine serum, 1âmM Na-pyruvate, 2âmM GlutaMax-1 (Gibco BRL, Grand Island NY), 16âmM HEPES pH 7.4, penicillin 100âU, and streptomycin 100âμg/ml. Cells were washed twice with serum-free media before the addition of test samples. Medium was replaced by serum-free media containing 0.1% EtOH with or without ibuprofen. After 1âh incubation, half the volume of media was replaced with the same media with or without recombinant human IL-1β (final concentration was 2â10âng/ml) (Sigma St Louis MO) was added. After 24âh incubation, media were collected. Western analysis employed our standard protocol (Lim et al, 2000) using anti-ACT polyclonal antibody (Calbiochem, CA #178216) (1â:â2500) and anti-rabbit antibody conjugated to HRP (Pierce #31463) (1â:â10â000).
Mouse Primary Astrocytes
Primary astrocyte cultures were prepared from cortices of postnatal 1â2-day-old mice (McCarthy and de Vellis, 1980). Briefly, mixed glia cells were obtained from tissue suspensions after incubation with trypsin. Confluent cultures were plated into six-well plates. Microglia contamination was limited by shaking flasks every 2 days and washing off cells. This yields cultures of 98% pure astrocytes (by GFAP ICC; data not shown). Ibuprofen in EtOH (final EtOH was 0.1%) was added 24âh before mouse IL-1β (final concentration was 2â10âng/ml (Sigma, St Louis, MO)) or LPS (final 5âμg/ml #L-8274 Sigma, St Louis, MO) stimulation. After 24âh stimulation, RNA was prepared using QIA shredder (QIAGEN, Valencia, CA) and an RNAqueous kit (Ambion, Austin, TX). One step QPCR was performed in triplicate from 10ng total RNA. LDH was assayed (Promega, Madison, WI) in the medium to measure toxicity.
RESULTS
Ibuprofen can Suppress Exogenous Amyloid Accumulation In Vivo
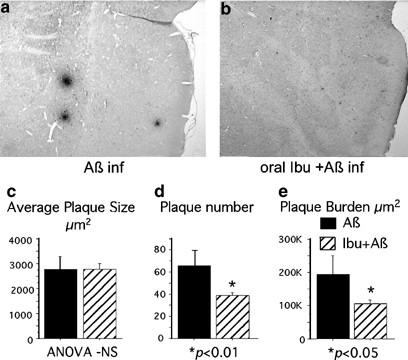
While ibuprofen at extremely high concentrations can reduce Aβ42 production in vitro, epidemiological studies suggest AD risk reduction is achieved at lower NSAID doses and with NSAIDs that cannot reduce Aβ42 production. Whether ibuprofen can suppress amyloid pathology beyond the level of Aβ production was tested in an Aβ CNS infusion rat model where infused exogenous Aβ accumulates in plaque-like deposits throughout the cortex and thalamus. Rats fed chow with (n=10) or without ibuprofen (375âppm) (n=10) for 60 days were then infused with human Aβ using Alzet pumps cannulated into the ventricles for 30 days, and then killed. Diffuse Aβ deposits (Figure 1a and b) were analyzed by immunohistochemistry using image analysis. While the average size of Aβ deposits was not changed with ibuprofen (Aβ: 2770±22âμm2 vs Aβ/Ibu: 2788±498âμm2; Figure 1c), the number of Aβ deposits (40.9% reduction, p<0.05, Aβ: 65.3±13.2 vs Aβ/Ibu: 38.6±2.6; Figure 1d) and the total Aβ-ir area (45% reduction, p<0.05, Aβ: 192â800±57â000âμm2 vs Aβ/Ibu: 106â000±10â500âμm2; Figure 1e) were significantly reduced by ibuprofen. These results show that ibuprofen can also suppress Aβ accumulation at a post Aβ-production step. This suggests the hypothesis that ibuprofen inhibits pro-amyloidogenic factors at this CNS-achievable dose.
Amyloid deposition in Aβ-infused rats. Rats were fed with or without ibuprofen 3 month prior to and 1 month during intracerebroventricular infusion with Aβ (Aβ40 and Aβ42). Brain Aβ immunohistochemistry of rat without (a) and with ibuprofen (b). Average plaque size (c), number of Aβ deposits (d), and total Aβ-ir area (e) were analyzed.
ApoE Levels were not Significantly Reduced by Ibuprofen Treatment
ApoE levels were measured in three different ways because apoE is amyloid binding and apoE levels might be changed as a secondary effect of reduced plaques where apoE protein accumulates. ApoE levels potently control the rate of amyloidosis by modulating Aβ deposition in APP transgenics, and apoE expression is potentially subject to inflammatory control. Since ibuprofen treatment reduced the inflammation marker GFAP in our APP transgenics (Lim et al, 2000), we reasoned that control of inflammation and gliosis might reduce apoE production. However, apoE levels were not affected by ibuprofen treatment in APPsw mice (Figure 2aâc). ApoE protein levels in both SDS soluble fraction (+3.3%, p=0.78) and TBS soluble fraction (â17.5%, p=0.21) were not significantly changed by ibuprofen (Figure 2a and b). ApoE mRNA levels, which avoids amyloid binding issues, were also measured using real-time PCR. ApoE mRNA levels were not significantly suppressed by ibuprofen (Figure 2c). Thus, ibuprofen treatment did not significantly impact levels of apoE protein or mRNA.
Relative mRNA expression and protein levels in mice cortex. mRNA levels were normalized to GAPDH and the average levels of control mice were set to 100. (e, g, and h) Open bars are Tgâ mice with control diet (n=10), closed bars are APP Tg+ mice with control diet (n=12), and hatched bars are APP Tg+ mice with ibuprofen diet (375âppm) for 6 months (n=8). ApoE protein in SDS and TBS fractions (a and b) and mRNA levels (c), BACE1 mRNA levels (d), and PPARγ mRNA levels (e) in APP Tg mice at the age of 16 months were not significantly changed by ibuprofen. mACT mRNA levels (f) in control (open bar) were increased by CNS LPS injection (closed bar) in mice. mACT mRNA (g and h) levels in mice at the age of 22 months (h) increased over levels at 16 months (g). Error bars indicate SD.
BACE mRNA was not Suppressed by Ibuprofen
Since BACE1 is critical for Aβ production and its expression is subject to inflammatory and NSAID control (Sastre et al, 2003), we measured BACE mRNA levels in ibuprofen-treated mice. Surprisingly, BACE mRNA was not reduced (Figure 2d). PPARγ, an ibuprofen target whose agonists suppress BACE (Sastre et al, 2003), was also not changed in its mRNA levels by ibuprofen treatment (Figure 2e); however, this does not rule out an effect of ibuprofen on PPARγ activity since PPARγ mRNA levels do not always reflect PPARγ activity.
A Murine Homologue of ACT, an Acute Phase Protein and Pro-Amyloidgenic Molecule, was Induced in APP Tg+ Mice and Suppressed by Ibuprofen
ACT is an acute phase protein induced by inflammation (Akiyama et al, 2000) as well as a pro-amyloidogenic molecule (Mucke et al, 2000; Nilsson et al, 2001). In our experiments, the analysis of ACT was performed in human and mouse material rather than in the rat infusion model, because a rat homolog of ACT has not been adequately identified. The murine ACT homologue mRNA (mACT, EB22/4) rather than protein was measured, because anti-mouse ACT antibody is not available and anti-human ACT antibody did not detect mouse ACT (data not shown). Measurement of ACT mRNA also has an advantage in that it can avoid the complication of ACT protein accumulation associated with Aβ deposits, which were reduced by ibuprofen.
The mACT mRNA was induced 5.02-fold by LPS injection, showing that inflammation induces mACT mRNA (Figure 2f). mACT mRNA was elevated in Tg+ mice compared to Tgâ controls by 40% at 16 months of age, p=0.003 (ANOVA; F(2,25)=6.21, p=0.0065) and by 177% at 22 months of age, p=0.000008 (Figure 3g and h). The 40% elevated mACT mRNA in 16-month Tg+ mice was suppressed by 18% (p=0.03) by ibuprofen (Figure 3g). Since the changes in mACT mRNA levels were moderate, we remeasured mACT mRNA using two sets of independently prepared cDNA from the same mice and observed very similar results (data not shown). This mACT mRNA suppression by ibuprofen was also confirmed by carefully optimized multiplex RT-PCR with radioactivity using differently designed primers (Table 1), where we observed a 24% reduction (p=0.02). Using different internal controls (β-actin vs GAPDH), the two assays showed reasonable reproducibility (R2=0.797, data not shown).
ACT levels in mouse and human astrocyte cell culture. The average levels of nontreated cells (open bars) were set to 100. Error bars indicate SD. ACT mRNA levels (a and c) were normalized to GAPDH. (a) mACT mRNA levels in mouse primary astrocyte culture (n=3). Incubation with ibuprofen (20 or 100âμM) or vehicle (0.1% EtOH) started 24âh prior to incubation with or without LPS (5âμg/ml) for the following 24âh. (b) ACT protein levels in human astrocytoma U373 measured by Western blotting (n=3). Cells were incubated with or without human IL-1β (2âng/ml) for 24âh. (c) mACT mRNA levels in mouse primary astrocyte culture (n=3). Cells were incubated with or without mouse IL-1β (10âng/ml) for 24âh.
The Suppression of an ACT mRNA by Ibuprofen in the Absence of Aβ Plaques In Vitro
The reduction of mACT mRNA by ibuprofen could be an indirect effect of the loss of Aβ plaques that stimulate glial activation and ACT production. To address this issue, we used a cell culture system with no Aβ accumulation or plaques. Astrocytes were chosen since they are the major source of ACT in brain (Abraham et al, 1988; Pasternack et al, 1989; Koo et al, 1991). mACT mRNA was induced 100.9%, p<0.001 (ANOVA; F(3,8)=68.6, p<0.0001) by LPS in mouse primary astrocyte cultures. Ibuprofen suppressed the mACT mRNA induction moderately, but significantly (10.7%, p=0.02 at 20âμM and 20.7%, p=0.0005 at 100âμM ibuprofen; Figure 3a). These results suggested that ibuprofen can suppress inflammation-induced mACT independently of Aβ plaque reduction.
We previously reported that IL-1β was reduced 64.7% by ibuprofen in the same mouse brains (Lim et al, 2000). IL-1β induces ACT in human astrocytes, but this has not been reported for any mouse ACT homolog. So we examined whether IL-1β can induce mouse and human ACT in vitro. We confirmed human ACT induction by human IL-1ββ using the human astrocytoma U373-MG, as previously reported (Gitter et al, 2000). ACT protein in medium from these cells was 2.2-fold induced (p=0.01; Figure 3b). In low passage, young, subconfluent murine primary astrocytes cultures, mouse IL-1β induced mACT (mouse homolog EB22/4) mRNA by 82.1% (p=0.01; Figure 3c). Confluent mouse primary astrocytes showed smaller (39%) inductions of mACT mRNA (data not shown). Aged astrocytes grown >40 days in vitro expressed much higher mACT mRNA (7â8-fold) compared to young cultures, and IL-1β did not induce mACT mRNA, even in subconfluent conditions (data not shown). Interestingly, similar confluence-dependent phenomena have been shown with human primary astrocytes (Das and Potter, 1995). Ibuprofen (10â500âμM) failed to suppress the ACT induction by IL-1β in both human astrocytoma and mouse astrocytes (data not shown). These in vivo and vitro data suggest that mACT suppression by ibuprofen is likely mediated by a reduction in IL-1β, rather than inhibition of the ACT response to IL-1β.
DISCUSSION
In APP Tg mice, we previously reported that 375âppm ibuprofen in diet from 10 to 16 month of age suppressed amyloid pathology as measured by the number (â52.6%, p=0.02) and total area (â55.9%, p=0.02) of amyloid plaques and SDS-insoluble Aβ levels (â39%, p=0.03) (Lim et al, 2000). To evaluate possible mechanisms mediating this effect of ibuprofen on amyloid, we measured the effect of ibuprofen on several factors known to affect amyloid formation. Two factors that showed significant effects of ibuprofen were ACT (an acute phase protein) and IL-1β (an inflammatory cytokine known to regulate ACT). We show that in ibuprofen-treated mice with amyloid reduction, ibuprofen also reduced mACT mRNA by â¼20% and reduced IL-1β levels by â¼70%. These effects can be linked by the regulatory pathway of ibuprofen controlling COX and other targets which elicit anti-inflammatory effects including reduction of IL-1β and ACT; further, IL-1β can directly regulate ACT expression (as discussed below).
Using additional animals chronically fed with the same dose of ibuprofen (375âppm), we measured ibuprofen levels in brain and plasma. The ibuprofen level in brain was 4.07âμM. The ibuprofen levels in plasma (47.0âμM) were comparable to high analgesic or the low end of the therapeutic range for rheumatoid patients (10â50âμg/ml=48.5â242.4âμM) (Davies, 1998). Although the tolerance of these levels of ibuprofen for the vulnerable elderly might be an issue for some, the ibuprofen dose for our mice produces plasma values that are in a range that can be chronically achieved in many patients.
Three mechanisms that could account for amyloid pathology suppression by ibuprofen are: (1) the selective reduction of Aβ42 production independent of COX; (2) the reduction of total Aβ production dependent on PGE2; and, (3) the postproduction reduction of amyloid aggregation and plaque formation. Considering the first of these three mechanisms, high dose ibuprofen (100â500âμM) has been shown to reduce Aβ42 production in vitro, an effect also shown by other NSAIDs to be independent of their anti-inflammatory effects on regulating COX, PPAR, and NFκB (Weggen et al, 2001; Morihara et al, 2002; Sagi et al, 2003; Takahashi et al, 2003). Although the COX-dependency of ibuprofen's effects on Aβ42 suppression has not been tested, another NSAID sulindac has been more extensively analyzed and clearly reduces Aβ42 independent of COX (Weggen et al, 2001; Sagi et al, 2003; Takahashi et al, 2003). Aβ42 production was reduced in 3-month-old Tg2576 mice with a brain ibuprofen level of â¼2âμM, which, surprisingly, was much lower than dosing required in vitro (Eriksen et al, 2003). Since the CNS ibuprofen levels (4.07âμM±1.292, n=6) achieved in our animals are comparable, the suppression of amyloid pathology we observed, could, at least in part, be due to a reduction of Aβ42 production.
In relation to the second mechanism, low dose ibuprofen (1âμM) can reduce total Aβ generation in COX-overexpressing cultured cells (Qin et al, 2003). The mechanism depends on prostaglandins and is supported in vivo by overexpression of neuronal COX-2, which increased amyloid pathology in APP Tg mice (Xiang et al, 2002). While in our mice PGE2 levels as a measure of COX activity were not measured, 375âppm ibuprofen was likely to suppress locally elevated COX activity, for the following reasons: (1) Daily ibuprofen dosing (375âppm in chow) was above the single dose ED50 (2.3âmg/kg) that inhibited PGE2 synthesis in ex vivo mouse brain (Ferrari et al, 1990), even when extrapolated based on 2.5âh ibuprofen plasma half life. (2) The ibuprofen levels in brain (4.07âμM) in mice consuming 375âppm ibuprofen (are above the IC50 of COX-1 (2.1âμM) and COX-2 (1.6âμM) (Tegeder et al, 2001). (3) In general, NSAID doses at the IC50, including ibuprofen, are known to reduce COX activity by blocking COX enzyme. However, ibuprofen can also reduce COX-2 mRNA levels (Kannan et al, 2004; Lo et al, 1998). COX-2 mRNA levels were measured in our ibuprofen treated mice, however, they were not significantly reduced (â13%, n=8, p=0.38; see supplemental data).
Taken together, these data argue that 375âppm ibuprofen should limit any local elevation of COX and PGE2 caused by the APP Tg. However, global elevations in PGE2 have not been detected in APP Tg mice at 14 or 20 months of age (Quinn et al, 2003). Whether or not COX-2 mRNA or activity is locally elevated in APP Tg and suppressed by ibuprofen in selected neuronal layers or periplaque regions is worthy of further study.
These two mechanisms related to Aβ production are, however, unlikely to be the only mechanisms for amyloid pathology reduction by ibuprofen, because ibuprofen reduced exogenous Aβ deposition in Aβ infused rats (Figure 1), demonstrating ibuprofen's ability to limit Aβ deposition independent of Aβ42 or Aβ production. Other reported results also suggest mechanisms beyond reduction of Aβ42 production. First, there are no published epidemiological studies to suggest substantial differences in the apparent neuroprotective (antidementia) effects between ibuprofen and naproxen, the latter being a drug that does not reduce Aβ42 production (Breitner JC, personal communication, 17 May 2004). In fact, the Rotterdam Study (in t' Veld et al, 2001) found a five-fold reduction in AD incidence with 2 or more years' administration (at least 2 years before onset) of any NSAID, including diclofenac, naproxen, and other drugs without âAβ42 loweringâ activity. Second, COX-independent Aβ42 reduction in vitro requires unrealistically high ibuprofen concentrations that are probably not relevant epidemiologically, while low dose ibuprofen has anti-inflammatory effects. For example, in vitro, ibuprofen and other NSAIDs can inhibit COX and suppress inflammation at lower doses than those for Aβ42 suppression (Weggen et al, 2001). It is thus likely that low dose ibuprofen can decrease amyloid pathology through IL-1β and ACT suppression based on anti-inflammation (our third mechanism), even when the ibuprofen dose is not high enough to suppress Aβ42 production. With high enough dosing, all three mechanisms should act coordinately to reduce amyloidogenesis.
ACT is not only an acute phase molecule but also a pro-amyloidogenic molecule. Overexpression of human ACT accelerates amyloid pathology in human APP Tg mice (Mucke et al, 2000; Nilsson et al, 2001). Although ACT functions remain unclear and some reports about biochemical interactions with Aβ are controversial, most evidence argues that ACT can play a causal role in amyloid aggregation and plaque formation, which is post-Aβ-production (Ma et al, 1994). mACT mRNA was reduced by ibuprofen both in vivo and in vitro. We confirmed that mACT mRNA was induced in brain by the inflammatory effect of LPS injection (Figure 2d) and showed that ibuprofen suppressed the induction of mACT mRNA by LPS in mouse primary astrocytes (Figure 3a). The mACT mRNA reduction by ibuprofen (Figure 2e) was associated with, but unlikely to be simply secondary to the reduction in amyloid plaques. First, in the astrocyte culture where amyloid plaques are absent, mACT mRNA levels were suppressed by ibuprofen (Figure 3a), and induced by IL-1β (Figure 3b and c). Second, we measured mACT mRNA to avoid the caveat that ACT protein accumulates with amyloid plaques.
In AD, both ACT levels and genetic polymorphisms could contribute to its effects on amyloid formation, since AD patients have higher ACT levels in plasma and/or CSF (Matsubara et al, 1990; Harigaya et al, 1995; Licastro et al, 1995; Lieberman et al, 1995) and polymorphisms differentially modify AD risk (Wang et al, 2002a; Wang et al, 2002b). ACT mRNA is greatly elevated in AD (Abraham et al, 1988; Pasternack et al, 1989; Koo et al, 1991). Our data showed mACT mRNA was elevated in Tg2576 mice compared to non-Tg mice by 2.8-fold at 22 months of age and by 1.4-fold at 16 months, even though Aβ levels are as high in these mice as those of late stage AD patients (Kawarabayashi et al, 2001). The larger percent increase in ACT in AD compared to mouse models may contribute to a more significant role of ACT in AD than in mouse models. In addition to ACT levels, variations in the association of ACT polymorphisms with increased AD risk are observed, with some haplotypes associated with increased and others with decreased AD risk (Wang et al, 2002b).
Human gene polymorphism and promoter studies suggest a relationship between IL-1β, ACT, and AD (Akiyama et al, 2000; Nicoll et al, 2000). The common TT IL-1β polymorphism, which shows higher plasma IL-1β levels in AD patients, interacts with one polymorphism in ACT to significantly increase AD risk (Licastro et al, 2000). This genetic polymorphism data support the participation of IL-1 in cytokine cascades previously linked to AD pathogenesis (Mrak and Griffin, 2000, 2001). The human ACT gene has enhancer elements responsive to IL-1-β-stimulated NFκB and AP-1 (Kordula et al, 2000) and human ACT mRNA expression is directly controlled by NFκB (Lieb et al, 1996). As ibuprofen is a weak NFκB inhibitor (Tegeder et al, 2001), it is possible that higher levels of ibuprofen or the less toxic R-flurbiprofen (Morihara et al, 2002) could directly reduce human ACT expression levels by inhibiting NFκB.
The contribution of ibuprofen's effects on ACT and IL-1β to the suppression of amyloid pathology is strengthened by the lack of effect of ibuprofen on several other candidate factors known to modulate amyloid formation (submitted). Candidate amyloid-regulating molecules that can be controlled by NSAIDs include APP (Lim et al, 2000), BACE, apoE, TGF-β1, C1q, C3, CD11b, CD11c, iNOS, and PPARγ. APP and BACE relate to Aβ42 production, while apoE modulates amyloid deposition or clearance; no changes in these molecules were detected (Figure 2aâe) (Lim et al, 2000). Other molecules whose possible suppression by NSAIDs can interrupt the clearance of amyloid, for example, inflammation-related, phagocytic microglia markers, complement components (TGF-β1, C1q, C3, CD11b, CD11c, iNOS, PPARγ) were also not suppressed by ibuprofen in our mice (submitted). Among these markers, only CD11c (phagosytosis-related amyloid clearance) was increased by the APP transgene; its insensitivity to reduction by ibuprofen supports the importance of the ACT and IL-1β axis as a NSAID target that can directly modulate amyloid metabolism.
We found no reductions in apoE mRNA or protein levels in TBS soluble or insoluble fractions from cortical regions of ibuprofen-treated animals (Figure 2aâc). Although apoE is often increased by inflammatory stimuli, and this can be coordinate with upregulation of the canonical glial reactivity marker GFAP (Poirier et al, 1991; Laping et al, 1994a) (which was downregulated by ibuprofen in these mice (Lim et al, 2000)), in some inflammatory and neurodegenerative conditions apoE mRNA expression is not coordinately regulated with GFAP mRNA (Laping et al, 1994b; Schauwecker et al, 1998; Petegnief et al, 2001), including conditions related to amyloid plaques and AD (Shao et al, 1997; Zarow and Victoroff, 1998). In addition, apoE mRNA is uniquely downregulated by apoE and Aβ proteins (Soulie et al, 1999; Yamauchi et al, 2003).
In summary, ibuprofen specifically reduced pro-amyloidogenic ACT both in vivo and in vitro, an effect likely to be mediated by reduction of IL-1β. To our knowledge, this is the first report that suggests those molecules are involved in NSAID suppression of amyloid pathology in vivo and are regulated by ibuprofen through its anti-inflammatory effects, a mechanism that could apply to other AD-protective anti-inflammatory drugs that fail to lower Aβ42 production, including naproxen (Martin et al, 2002) and high dose aspirin.
References
Abraham CR, Selkoe DJ, Potter H (1988). Immunochemical identification of the serine protease inhibitor alpha 1-antichymotrypsin in the brain amyloid deposits of Alzheimer's disease. Cell 52: 487â501.
Akiyama H, Barger S, Barnum S, Bradt B, Bauer J, Cole GM et al (2000). Inflammation and Alzheimer's disease. Neurobiol Aging 21: 383â421.
Bales KR, Verina T, Dodel RC, Du Y, Altstiel L, Bender M et al (1997). Lack of apolipoprotein E dramatically reduces amyloid beta-peptide deposition. Nat Genet 17: 263â264.
Breitner JC, Gau BA, Welsh KA, Plassman BL, McDonald WM, Helms MJ et al (1994). Inverse association of anti-inflammatory treatments and Alzheimer's disease: initial results of a co-twin control study. Neurology 44: 227â232.
Bullido MJ, Artiga MJ, Recuero M, Sastre I, Garcia MA, Aldudo J et al (1998). A polymorphism in the regulatory region of APOE associated with risk for Alzheimer's dementia. Nat Genet 18: 69â71.
Cai H, Wang Y, McCarthy D, Wen H, Borchelt DR, Price DL et al (2001). BACE1 is the major beta-secretase for generation of Abeta peptides by neurons. Nat Neurosci 4: 233â234.
Das S, Potter H (1995). Expression of the Alzheimer amyloid-promoting factor antichymotrypsin is induced in human astrocytes by IL-1. Neuron 14: 447â456.
Davies NM (1998). Clinical pharmacokinetics of ibuprofen. The first 30 years. Clin Pharmacokinet 34: 101â154.
Diedrich JF, Minnigan H, Carp RI, Whitaker JN, Race R, Frey II W et al (1991). Neuropathological changes in scrapie and Alzheimer's disease are associated with increased expression of apolipoprotein E and cathepsin D in astrocytes. J Virol 65: 4759â4768.
Eriksen JL, Sagi SA, Smith TE, Weggen S, Das P, McLendon DC et al (2003). NSAIDs and enantiomers of flurbiprofen target gamma-secretase and lower Abeta 42 in vivo. J Clin Invest 112: 440â449.
Fagan AM, Watson M, Parsadanian M, Bales KR, Paul SM, Holtzman DM (2002). Related articles, links abstract human and murine ApoE markedly alters A beta metabolism before and after plaque formation in a mouse model of Alzheimer's disease. Neurobiol Dis 9: 305â318.
Ferrari RA, Ward SJ, Zobre CM, Van Liew DK, Perrone MH, Connell MJ et al (1990). Estimation of the in vivo effect of cyclooxygenase inhibitors on prostaglandin E2 levels in mouse brain. Eur J Pharmacol 179: 25â34.
Frautschy S, Sigel JJ, Harris-White ME, Chu T, Cole GM (1999). Methods for evaluating in vivo redent models for Alzheimer's disease. In: Timiras P, Sternberg H (eds). Methods in Brain Aging. Springer-Verlag: Berlin. pp 66â76.
Frautschy SA, Hu W, Kim P, Miller SA, Chu T, Harris-White ME et al (2001). Phenolic anti-inflammatory antioxidant reversal of Abeta-induced cognitive deficits and neuropathology. Neurobiol Aging 22: 993â1005.
Gitter BD, Boggs LN, May PC, Czilli DL, Carlson CD (2000). Regulation of cytokine secretion and amyloid precursor protein processing by proinflammatory amyloid beta (A beta). Ann N Y Acad Sci 917: 154â164.
Harigaya Y, Shoji M, Nakamura T, Matsubara E, Hosoda K, Hirai S (1995). Alpha 1-antichymotrypsin level in cerebrospinal fluid is closely associated with late onset Alzheimer's disease. Intern Med 34: 481â484.
in t' Veld BA, Ruitenberg A, Hofman A, Launer LJ, van Duijn CM, Stijnen T et al (2001). Nonsteroidal antiinflammatory drugs and the risk of Alzheimer's disease. N Engl J Med 345: 1515â1521.
Jantzen PT, Connor KE, DiCarlo G, Wenk GL, Wallace JL, Rojiani AM et al (2002). Microglial activation and beta-amyloid deposit reduction caused by a nitric oxide-releasing nonsteroidal anti-inflammatory drug in amyloid precursor protein plus presenilin-1 transgenic mice. J Neurosci 22: 2246â2254.
Kannan S, Kolhe P, Raykova V, Glibatec M, Kannan RM, Lieh-Lai M et al (2004). Dynamics of cellular entry and drug delivery by dendritic polymers into human lung epithelial carcinoma cells. J Biomater Sci Polym Ed 15: 311â330.
Kawarabayashi T, Younkin LH, Saido TC, Shoji M, Ashe KH, Younkin SG (2001). Age-dependent changes in brain, CSF, and plasma amyloid (beta) protein in the Tg2576 transgenic mouse model of Alzheimer's disease. J Neurosci 21: 372â381.
Koo EH, Abraham CR, Potter H, Cork LC, Price DL (1991). Developmental expression of alpha 1-antichymotrypsin in brain may be related to astrogliosis. Neurobiol Aging 12: 495â501.
Kordula T, Bugno M, Rydel RE, Travis J (2000). Mechanism of interleukin-1- and tumor necrosis factor alpha-dependent regulation of the alpha 1-antichymotrypsin gene in human astrocytes. J Neurosci 20: 7510â7516.
Lambert JC, Pasquier F, Cottel D, Frigard B, Amouyel P, Chartier-Harlin MC (1998). A new polymorphism in the APOE promoter associated with risk of developing Alzheimer's disease. Hum Mol Genet 7: 533â540.
Lambert JC, Perez-Tur J, Dupire MJ, Galasko D, Mann D, Amouyel P et al (1997). Distortion of allelic expression of apolipoprotein E in Alzheimer's disease. Hum Mol Genet 6: 2151â2154.
Laping NJ, Morgan TE, Nichols NR, Rozovsky I, Young-Chan CS, Zarow C et al (1994a). Transforming growth factor-beta 1 induces neuronal and astrocyte genes: tubulin alpha 1, glial fibrillary acidic protein and clusterin. Neuroscience 58: 563â572.
Laping NJ, Teter B, Nichols NR, Rozovsky I, Finch CE (1994b). Glial fibrillary acidic protein: regulation by hormones, cytokines, and growth factors. Brain Pathol 4: 259â275.
Laws SM, Taddei K, Martins G, Paton A, Fisher C, Clarnette R et al (1999). The â491AA polymorphism in the APOE gene is associated with increased plasma apoE levels in Alzheimer's disease. NeuroReport 10: 879â882.
Licastro F, Mallory M, Hansen LA, Masliah E (1998). Increased levels of alpha-1-antichymotrypsin in brains of patients with Alzheimer's disease correlate with activated astrocytes and are affected by APOE 4 genotype. J Neuroimmunol 88: 105â110.
Licastro F, Parnetti L, Morini MC, Davis LJ, Cucinotta D, Gaiti A et al (1995). Acute phase reactant alpha 1-antichymotrypsin is increased in cerebrospinal fluid and serum of patients with probable Alzheimer disease. Alzheimer Dis Assoc Disord 9: 112â118.
Licastro F, Pedrini S, Ferri C, Casadei V, Govoni M, Pession A et al (2000). Gene polymorphism affecting alpha1-antichymotrypsin and interleukin-1 plasma levels increases Alzheimer's disease risk. Ann Neurol 48: 388â391.
Lieb K, Fiebich BL, Schaller H, Berger M, Bauer J (1996). Interleukin-1 beta and tumor necrosis factor-alpha induce expression of alpha 1-antichymotrypsin in human astrocytoma cells by activation of nuclear factor-kappa B. J Neurochem 67: 2039â2044.
Lieberman J, Schleissner L, Tachiki KH, Kling AS (1995). Serum alpha 1-antichymotrypsin level as a marker for Alzheimer-type dementia. Neurobiol Aging 16: 747â753.
Lim GP, Yang F, Chu T, Chen P, Beech W, Teter B et al (2000). Ibuprofen suppresses plaque pathology and inflammation in a mouse model for Alzheimer's disease. J Neurosci 20: 5709â5714.
Lim GP, Yang F, Chu T, Gahtan E, Ubeda O, Beech W et al (2001). Ibuprofen effects on Alzheimer pathology and open field activity in APPsw transgenic mice. Neurobiol Aging 22: 983â991.
Lo CJ, Cryer HG, Fu M, Lo FR (1998). Regulation of macrophage eicosanoid generation is dependent on nuclear factor kappaB. J Trauma 45: 19â23 ; discussion 23â14.
Luo Y, Bolon B, Kahn S, Bennett BD, Babu-Khan S, Denis P et al (2001). Mice deficient in BACE1, the Alzheimer's beta-secretase, have normal phenotype and abolished beta-amyloid generation. Nat Neurosci 4: 231â232.
Ma J, Yee A, Brewer Jr HB, Das S, Potter H (1994). Amyloid-associated proteins alpha 1-antichymotrypsin and apolipoprotein E promote assembly of Alzheimer beta-protein into filaments. Nature 372: 92â94.
Martin BK, Meinert CL, Breitner JC (2002). Double placebo design in a prevention trial for Alzheimer's disease. Control Clin Trials 23: 93â99.
Matsubara E, Hirai S, Amari M, Shoji M, Yamaguchi H, Okamoto K et al (1990). Alpha 1-antichymotrypsin as a possible biochemical marker for Alzheimer-type dementia. Ann Neurol 28: 561â567.
McCarthy KD, de Vellis J (1980). Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue. J Cell Biol 85: 890â902.
McGeer EG, McGeer PL (2003). Inflammatory processes in Alzheimer's disease. Prog Neuropsychopharmacol Biol Psychiatry 27: 741â749.
McGeer PL, Schulzer M, McGeer EG (1996). Arthritis and anti-inflammatory agents as possible protective factors for Alzheimer's disease: a review of 17 epidemiologic studies. Neurology 47: 425â432.
Morihara T, Chu T, Ubeda O, Beech W, Cole GM (2002). Selective inhibition of Abeta42 production by NSAID R-enantiomers. J Neurochem 83: 1009â1012.
Mrak RE, Griffin WS (2000). Interleukin-1 and the immunogenetics of Alzheimer disease. J Neuropathol Exp Neurol 59: 471â476.
Mrak RE, Griffin WS (2001). Interleukin-1, neuroinflammation, and Alzheimer's disease. Neurobiol Aging 22: 903â908.
Mucke L, Yu GQ, McConlogue L, Rockenstein EM, Abraham CR, Masliah E (2000). Astroglial expression of human alpha(1)-antichymotrypsin enhances alzheimer-like pathology in amyloid protein precursor transgenic mice. Am J Pathol 157: 2003â2010.
Nicoll JA, Mrak RE, Graham DI, Stewart J, Wilcock G, MacGowan S et al (2000). Association of interleukin-1 gene polymorphisms with Alzheimer's disease. Ann Neurol 47: 365â368.
Nilsson LN, Bales KR, DiCarlo G, Gordon MN, Morgan D, Paul SM et al (2001). Alpha-1-antichymotrypsin promotes beta-sheet amyloid plaque deposition in a transgenic mouse model of Alzheimer's disease. J Neurosci 21: 1444â1451.
Pahnke J, Walker LC, Schroeder E, Vogelgesang S, Stausske D, Walther R et al (2003). Cerebral beta-amyloid deposition is augmented by the â491AA promoter polymorphism in non-demented elderly individuals bearing the apolipoprotein E epsilon4 allele. Acta Neuropathol (Berl) 105: 25â29.
Pasternack JM, Abraham CR, Van Dyke BJ, Potter H, Younkin SG (1989). Astrocytes in Alzheimer's disease gray matter express alpha 1-antichymotrypsin mRNA. Am J Pathol 135: 827â834.
Petegnief V, Saura J, de Gregorio-Rocasolano N, Paul SM (2001). Neuronal injury-induced expression and release of apolipoprotein E in mixed neuron/glia co-cultures: nuclear factor kappaB inhibitors reduce basal and lesion-induced secretion of apolipoprotein E. Neuroscience 104: 223â234.
Poirier J, Hess M, May PC, Finch CE (1991). Astrocytic apolipoprotein E mRNA and GFAP mRNA in hippocampus after entorhinal cortex lesioning. Brain Res Mol Brain Res 11: 97â106.
Qiao X, Cummins DJ, Paul SM (2001). Neuroinflammation-induced acceleration of amyloid deposition in the APPV717F transgenic mouse. Eur J Neurosci 14: 474â482.
Qin W, Ho L, Pompl PN, Peng Y, Zhao Z, Xiang Z et al (2003). Cyclooxygenase (COX)-2 and COX-1 potentiate beta-amyloid peptide generation through mechanisms that involve gamma-secretase activity. J Biol Chem 278: 50970â50977.
Quinn J, Montine T, Morrow J, Woodward WR, Kulhanek D, Eckenstein F (2003). Related articles, links abstract inflammation and cerebral amyloidosis are disconnected in an animal model of Alzheimer's disease. J Neuroimmunol 137: 32â41.
Roberds SL, Anderson J, Basi G, Bienkowski MJ, Branstetter DG, Chen KS et al (2001). BACE knockout mice are healthy despite lacking the primary beta-secretase activity in brain: implications for Alzheimer's disease therapeutics. Hum Mol Genet 10: 1317â1324.
Roks G, Cruts M, Bullido MJ, Backhovens H, Artiga MJ, Hofman A et al (1998). The â491 A/T polymorphism in the regulatory region of the apolipoprotein E gene and early-onset Alzheimer's disease. Neurosci Lett 258: 65â68.
Sagi SA, Weggen S, Eriksen J, Golde TE, Koo EH (2003). The non-cyclooxygenase targets of non-steroidal anti-inflammatory drugs, lipoxygenases, peroxisome proliferator-activated receptor, inhibitor of kappa B kinase, and NF kappa B, do not reduce amyloid beta 42 production. J Biol Chem 278: 31825â31830.
Sastre M, Dewachter I, Landreth GE, Willson TM, Klockgether T, van Leuven F et al (2003). Nonsteroidal anti-inflammatory drugs and peroxisome proliferator-activated receptor-gamma agonists modulate immunostimulated processing of amyloid precursor protein through regulation of beta-secretase. J Neurosci 23: 9796â9804.
Schauwecker PE, Cogen JP, Jiang T, Cheng HW, Collier TJ, McNeill TH (1998). Differential regulation of astrocytic mRNAs in the rat striatum after lesions of the cortex or substantia nigra. Exp Neurol 149: 87â96.
Shao Y, Gearing M, Mirra SS (1997). Astrocyteâapolipoprotein E associations in senile plaques in Alzheimer disease and vascular lesions: a regional immunohistochemical study. J Neuropathol Exp Neurol 56: 376â381.
Shoji M, Hirai S, Yamaguchi H, Harigaya Y, Ishiguro K, Matsubara E (1991). Alpha 1-antichymotrypsin is present in diffuse senile plaques. A comparative study of beta-protein and alpha 1-antichymotrypsin immunostaining in the Alzheimer brain. Am J Pathol 138: 247â257.
Song YQ, Rogaeva E, Premkumar S, Brindle N, Kawarai T, Orlacchio A et al (1998). Absence of association between Alzheimer disease and the â491 regulatory region polymorphism of APOE. Neurosci Lett 250: 189â192.
Soulie C, Mitchell V, Dupont-Wallois L, Chartier-Harlin MC, Beauvillain JC, Delacourte A et al (1999). Synthesis of apolipoprotein E (ApoE) mRNA by human neuronal-type SK N SH-SY 5Y cells and its regulation by nerve growth factor and ApoE. Neurosci Lett 265: 147â150.
Stewart WF, Kawas C, Corrada M, Metter EJ (1997). Risk of Alzheimer's disease and duration of NSAID use. Neurology 48: 626â632.
Takahashi Y, Hayashi I, Tominari Y, Rikimaru K, Morohashi Y, Kan T et al (2003). Sulindac sulfide is a noncompetitive gamma-secretase inhibitor that preferentially reduces Abeta 42 generation. J Biol Chem 278: 18664â18670.
Tegeder I, Pfeilschifter J, Geisslinger G (2001). Cyclooxygenase-independent actions of cyclooxygenase inhibitors. FASEB J 15: 2057â2072.
Wang X, DeKosky ST, Ikonomovic MD, Kamboh MI (2002a). Distribution of plasma alpha 1-antichymotrypsin levels in Alzheimer disease patients and controls and their genetic controls. Neurobiol Aging 23: 377â382.
Wang X, DeKosky ST, Luedecking-Zimmer E, Ganguli M, Kamboh IM (2002b). Genetic variation in alpha(1)-antichymotrypsin and its association with Alzheimer's disease. Hum Genet 110: 356â365.
Weggen S, Eriksen JL, Das P, Sagi SA, Wang R, Pietrzik CU et al (2001). A subset of NSAIDs lower amyloidogenic Abeta42 independently of cyclooxygenase activity. Nature 414: 212â216.
Weggen S, Eriksen JL, Sagi SA, Pietrzik CU, Golde TE, Koo EH (2003a). Abeta42-lowering nonsteroidal anti-inflammatory drugs preserve intramembrane cleavage of the amyloid precursor protein (APP) and ErbB-4 receptor and signaling through the APP intracellular domain. J Biol Chem 278: 30748â30754.
Weggen S, Eriksen JL, Sagi SA, Pietrzik CU, Ozols V, Fauq A et al (2003b). Evidence that nonsteroidal anti-inflammatory drugs decrease amyloid beta 42 production by direct modulation of gamma-secretase activity. J Biol Chem 278: 31831â31837.
Xiang Z, Ho L, Valdellon J, Borchelt D, Kelley K, Spielman L et al (2002). Cyclooxygenase (COX)-2 and cell cycle activity in a transgenic mouse model of Alzheimer's disease neuropathology. Neurobiol Aging 23: 327â334.
Yamada T, Kondo A, Takamatsu J, Tateishi J, Goto I (1995). Apolipoprotein E mRNA in the brains of patients with Alzheimer's disease. J Neurol Sci 129: 56â61.
Yamauchi K, Tozuka M, Hidaka E, Ueno I, Matsuda K, Katsuyama T (2003). Internalization of beta-amyloid causes downregulation of apolipoprotein E mRNA expression in neuroblastoma cells. Ann Clin Lab Sci 33: 68â78.
Yan Q, Zhang J, Liu H, Babu-Khan S, Vassar R, Biere AL et al (2003). Anti-inflammatory drug therapy alters beta-amyloid processing and deposition in an animal model of Alzheimer's disease. J Neurosci 23: 7504â7509.
Yasojima K, Schwab C, McGeer EG, McGeer PL (1999). Up-regulated production and activation of the complement system in Alzheimer's disease brain. Am J Pathol 154: 927â936.
Zandi PP, Anthony JC, Hayden KM, Mehta K, Mayer L, Breitner JC (2002). Reduced incidence of AD with NSAID but not H2 receptor antagonists: the Cache County Study. Neurology 59: 880â886.
Zarow C, Victoroff J (1998). Increased apolipoprotein E mRNA in the hippocampus in Alzheimer disease and in rats after entorhinal cortex lesioning. Exp Neurol 149: 79â86.
Zhou Y, Su Y, Li B, Liu F, Ryder JW, Wu X et al (2003). Nonsteroidal anti-inflammatory drugs can lower amyloidogenic Abeta42 by inhibiting Rho. Science 302: 1215â1217.
Acknowledgements
We thank Dr Peter Tontonoz and Dr Sean B Joseph (UCLA) for PPARγ primers and probe. We also thank the following colleagues for the excellent technical contributions: Walter Beech, Ping Ping Chen, Mychica Simmons, Oliver J Ubeda, and Sheryl Miller. This research was supported by grants from NIH AG13471 and NS43946 to GMC, AG00962 to BT, AG10685, AG16793 to SAF, Mitsubishi Pharma Research Foundation to TM and contributions from private donors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary information
Rights and permissions
About this article
Cite this article
Morihara, T., Teter, B., Yang, F. et al. Ibuprofen Suppresses Interleukin-1β Induction of Pro-Amyloidogenic α1-Antichymotrypsin to Ameliorate β-Amyloid (Aβ) Pathology in Alzheimer's Models. Neuropsychopharmacol 30, 1111â1120 (2005). https://doi.org/10.1038/sj.npp.1300668
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.npp.1300668
Keywords
This article is cited by
-
The brain consequences of systemic inflammation were not fully alleviated by ibuprofen treatment in mice
Pharmacological Reports (2021)
-
Cyclooxygenase-2 Induced the β-Amyloid Protein Deposition and Neuronal Apoptosis Via Upregulating the Synthesis of Prostaglandin E2 and 15-Deoxy-Î12,14-prostaglandin J2
Neurotherapeutics (2019)
-
Neuroinflammatory responses in Alzheimerâs disease
Journal of Neural Transmission (2018)
-
Amelioration of amyloid-β-induced deficits by DcR3 in an Alzheimerâs disease model
Molecular Neurodegeneration (2017)
-
Tracking neuroinflammation in Alzheimerâs disease: the role of positron emission tomography imaging
Journal of Neuroinflammation (2014)