Abstract
Although the immune response is often regarded as acting to suppress tumor growth, it is now clear that it can be both stimulatory and inhibitory. The interplay between these competing influences has complex implications for tumor development, cancer dormancy, and immunotherapies. In fact, early immunotherapy failures were partly due to a lack in understanding of the nonlinear growth dynamics these competing immune actions may cause. To study this biological phenomenon theoretically, we construct a minimally parameterized framework that incorporates all aspects of the immune response. We combine the effects of all immune cell types, general principles of self-limited logistic growth, and the physical process of inflammation into one quantitative setting. Simulations suggest that while there are pro-tumor or antitumor immunogenic responses characterized by larger or smaller final tumor volumes, respectively, each response involves an initial period where tumor growth is stimulated beyond that of growth without an immune response. The mathematical description is non-identifiable which allows an ensemble of parameter sets to capture inherent biological variability in tumor growth that can significantly alter tumor–immune dynamics and thus treatment success rates. The ability of this model to predict non-intuitive yet clinically observed patterns of immunomodulated tumor growth suggests that it may provide a means to help classify patient response dynamics to aid identification of appropriate treatments exploiting immune response to improve tumor suppression, including the potential attainment of an immune-induced dormant state.





Similar content being viewed by others
References
Adam JA, Bellomo N (1997) A survey of models for tumor–immune system dynamics. Birkhäuser, Boston
Al-Tameemi MM, Chaplain MA, d’Onofrio A (2012) Evasion of tumours from the control of the immune system: consequences of brief encounters. Biol Direct 7(1):31
Albini A (2005) Tumor inflammatory angiogenesis and its chemoprevention. Cancer Res 65(23):10637–10641
Balkwill FR, Coussens LM (2004) Cancer: an inflammatory link. Nature 431(7007):405–406
Betts G et al (2007) The impact of regulatory T cells on carcinogen-induced sarcogenesis. Br J Cancer 96(12):1849–1854
Cirit M, Haugh JM (2012) Data-driven modelling of receptor tyrosine kinase signalling networks quantifies receptor-specific potencies of PI3K- and Ras-dependent ERK activation. Biochem J 441(1):77–85
Cohen M et al (2010) Sialylation of 3-methylcholanthrene-induced fibrosarcoma determines antitumor immune responses during immunoediting. J Immunol 185(10):5869–5878
Condeelis J, Pollard JW (2006) Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124(2):263–266
De Palma M et al (2005) Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8(3):211–226
de Pillis LG, Radunskaya AE, Wiseman CL (2005) A validated mathematical model of cell-mediated immune response to tumor growth. Cancer Res 65(17):7950–7958
de Visser KE, Eichten A, Coussens LM (2006) Paradoxical roles of the immune system during cancer development. Nat Rev Cancer 6(1):24–37
den Breems NY, Eftimie R (2016) The re-polarisation of M2 and M1 macrophages and its role on cancer outcomes. J Theor Biol 390(C):23–39
DeLisi C, Rescigno A (1977) Immune surveillance and neoplasia—I. A minimal mathematical model. Bull Math Biol 39:201–221
DeNardo DG, Andreu P, Coussens LM (2010) Interactions between lymphocytes and myeloid cells regulate pro- versus anti-tumor immunity. Cancer Metastasis Rev 29(2):309–316
Diefenbach A et al (2001) Rae1 and H60 ligands of the NKG2D receptor stimulate tumour immunity. Nature 413(6852):165–171
d’Onofrio A (2005) A general framework for modeling tumor–immune system competition and immunotherapy: mathematical analysis and biomedical inferences. Phys D 208(3–4):220–235
d’Onofrio A, Ciancio A (2011) Simple biophysical model of tumor evasion from immune system control. Phys Rev E 84(3):031910
Eftimie R, Bramson JL, Earn DJ (2010) Interactions between the immune system and cancer: a brief review of non-spatial mathematical models. Bull Math Biol 73(1):2–32
Enderling H, Hlatky LR, Hahnfeldt P (2012) Immunoediting: evidence of the multifaceted role of the immune system in self-metastatic tumor growth. Theor Biol Med Model 9:31
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140(6):883–899
Hahnfeldt P et al (1999) Tumor development under angiogenic signaling: a dynamical theory of tumor growth, treatment response, and postvascular dormancy. Cancer Res 59:4770–4775
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Hoos A (2012) Evolution of end points for cancer immunotherapy trials. Ann Oncol 23(Suppl 8):viii47–viii52
Jackaman C et al (2003) IL-2 intratumoral immunotherapy enhances CD8+ T cells that mediate destruction of tumor cells and tumor-associated vasculature: a novel mechanism for IL-2. J Immunol 171(10):5051–5063
Ji R-C (2012) Macrophages are important mediators of either tumor- or inflammation-induced lymphangiogenesis. Cell Mol Life Sci 69(6):897–914
Joshi B et al (2009) On immunotherapies and cancer vaccination protocols: a mathematical modelling approach. J Theor Biol 259(4):820–827
Kareva I, Wilkie KP, Hahnfeldt P (2014) The power of the tumor microenvironment: a systemic approach for a systemic disease. In: Mathematical oncology 2013. Modeling and simulation in science, engineering and technology. Springer, New York, pp 181–196
Kirschner DE, Panetta JC (1998) Modeling immunotherapy of the tumor–immune interaction. J Math Biol 37(3):235–252
Koebel CM et al (2007) Adaptive immunity maintains occult cancer in an equilibrium state. Nature 450(7171):903–907
Kraus S, Arber N (2009) Inflammation and colorectal cancer. Curr Opin Pharmacol 9(4):405–410
Kuznetsov VA (1988) Mathematical modeling of the development of dormant tumors and immune stimulation of their growth. Cybern Syst Anal 23(4):556–564
Kuznetsov VA et al (1994) Nonlinear dynamics of immunogenic tumors: parameter estimation and global bifurcation analysis. Bull Math Biol 56(2):295–321
Lefever R, Horsthemke W (1978) Bistability in fluctuating environments. Implications in tumor immunology. Bull Math Biol 41:469–490
Li LM, Nicolson GL, Fidler IJ (1991) Direct in vitro lysis of metastatic tumor cells by cytokine-activated murine vascular endothelial cells. Cancer Res 51(1):245–254
Louzoun Y et al (2014) A mathematical model for pancreatic cancer growth and treatments. J Theor Biol 351:74–82
Mantovani A et al (2008) Tumour immunity: effector response to tumour and role of the microenvironment. Lancet 371(9614):771–783
Matzavinos A, Chaplain MAJ, Kuznetsov VA (2004) Mathematical modelling of the spatio-temporal response of cytotoxic T-lymphocytes to a solid tumour. Math Med Biol 21(1):1–34
Meshkat N, Anderson C, Distefano JJ (2011) Finding identifiable parameter combinations in nonlinear ODE models and the rational reparameterization of their input–output equations. Math Biosci 233(1):19–31
Miao H et al (2011) On identifiability of nonlinear ode models and applications in viral dynamics. SIAM Rev 53(1):3–39
Nelson D, Ganss R (2006) Tumor growth or regression: powered by inflammation. J Leukoc Biol 80(4):685–690
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. doi:10.1038/nrc3239
Phillips C (2012) Clinical Trials Network aims to strengthen cancer immunotherapy pipeline. NCI Cancer Bulletin—National Cancer Institute. 9(4). http://www.cancer.gov/ncicancerbulletin/022112/page6?cid=FBen+sf3319032. Accessed 28 Feb 2012
Prehn RT (1972) The immune reaction as a stimulator of tumor growth. Science 176:170–171
Prehn RT (2007) Immunostimulation and immunoinhibition of premalignant lesions. Theor Biol Med Model 4(1):6
Quesnel B (2008) Tumor dormancy and immunoescape. APMIS 116:685–694
Rakoff-Nahoum S (2006) Why cancer and inflammation? Yale J Biol Med 79(3–4):123–130
Raue A et al (2011) Addressing parameter identifiability by model-based experimentation. IET Syst Biol 5(2):120–130
Robert CP, Casella G (2010) Introducing Monte Carlo methods with R. Springer, New York
Roose T, Chapman SJ, Maini PK (2007) Mathematical models of avascular tumor growth. SIAM Rev 49(2):179–208
Sampson D et al (1977) Dose dependence of immunopotentiation of tumor regression induced by levamisole. Cancer Res 37(10):3526–3529
Takayanagi T, Kawamura H, Ohuchi A (2006) Cellular automaton model of a tumor tissue consisting of tumor cells, cytotoxic T lymphocytes (CTLs), and cytokine produced by CTLs. IPSJ Digit Cour 2:138–144
Tanooka H, Tanaka K, Arimoto H (1982) Dose response and growth rates of subcutaneous tumors induced with 3-methylcholanthrene in mice and timing of tumor origin. Cancer Res 42(11):4740–4743
Wilkie KP (2013) A review of mathematical models of cancer–immune interactions in the context of tumor dormancy. In: Enderling H, Almog N, Hlatky L (eds) Systems biology of tumor dormancy. Springer, New York, pp 201–234
Wilkie KP, Hahnfeldt P (2013) Tumor–immune dynamics regulated in the microenvironment inform the transient nature of immune-induced tumor dormancy. Cancer Res 73(12):3534–3544. http://cancerres.aacrjournals.org/content/early/2013/03/22/0008-5472.CAN-12-4590.short
Wolchok JD et al (2009) Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clinical Cancer Res 15(23):7412–7420
Acknowledgements
The authors would like to thank Dr. M. La Croix for his helpful discussions and for creating Fig. 1. The work of K.W. and P.H. was supported by the National Cancer Institute under Award Number U54CA149233 (to L. Hlatky) and by the Office of Science (BER), U.S. Department of Energy, under Award Number DE-SC0001434 (to P.H.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding authors
Appendices
Appendix 1
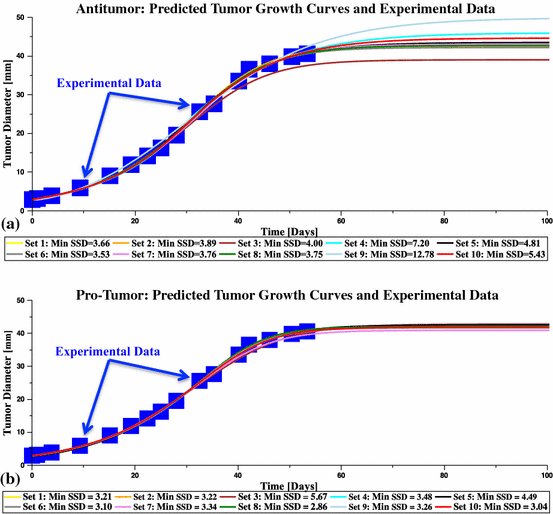
See Fig. 6.
Results of the data fitting: predicted tumor growth curves are shown over-laid with the experimental data for both antitumor and pro-tumor parameter set ensembles. Values of the parameters for each set are listed in Table 1 (Color figure online)
Appendix 2
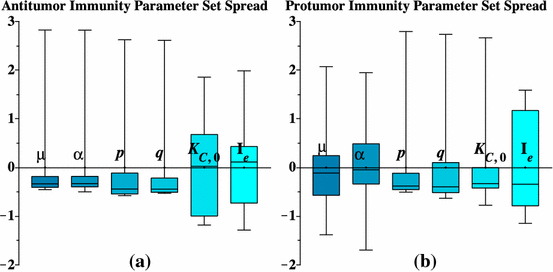
See Fig. 7.
Box plots showing the variability in the pro-tumor and antitumor parameter ensembles. Parameter values are rescaled for easier comparison by the normalization formula: \(x_i \rightarrow \frac{x_i -\mu _x }{\sigma _x }\) where \(\mu _x \) is the average, and \(\sigma _x \) is the standard deviation, of the set of values for parameter x (Color figure online)
Appendix 3
See Fig. 8.
Cancer–immune phase portraits demonstrate the variability across the 10 parameter sets in the antitumor (a) and pro-tumor (b) immunity ensembles (values listed in Table 1). Tumors are simulated to grow from an initial injection of \(C_0 =10^{4}\) cancer cells and varying numbers of immune cells \((I_0 =\gamma C_0 )\). The ranges (values of \(\gamma )\) that divide the behavior between tumor growth (red) or tumor suppression (blue) are listed below the plots. Simulations result from solving the full system of equations (1)–(4) with direct predation through \(\varPsi \), Eq. (5). Axes indicate diameter of spherical population in mm (Color figure online)

Rights and permissions
About this article
Cite this article
Wilkie, K.P., Hahnfeldt, P. Modeling the Dichotomy of the Immune Response to Cancer: Cytotoxic Effects and Tumor-Promoting Inflammation. Bull Math Biol 79, 1426–1448 (2017). https://doi.org/10.1007/s11538-017-0291-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0291-4