Abstract
Functional traits reveal the adaptive strategies of species to their environment, and are relevant to the formation of communities, the function of ecosystems, and the mechanisms underlying biodiversity. However, trait databases have not been established for most biological taxa, especially for insects, which encompass a vast number of species. This study measured the morphological traits of 307 species of Heteroptera insects collected in 2019 from the “Xishuangbanna Priority Conservation Area” in Southwest China using sweep netting and light trapping methods. This study provides a dataset for 307 Heteroptera species, comprising 34 morphometric measurements and 17 morphological traits. The dataset contains information on species sex, abundance, and the average, maximum, and minimum values of traits. This dataset facilitates an enhanced understanding of the functional traits and ecological associations of Heteroptera insects and offers opportunities for exploring a more diverse range of research topics.
Similar content being viewed by others
Background & Summary
Research methodologies based on functional trait measurements offer a robust framework for comparing the ecological differences among species across various scales and for describing their community structure characteristics1,2,3. These methodologies hold promising applications in unveiling the mechanisms of species coexistence within communities4,5. There is a strong correlation between environmental filtering and the functional traits of species; the loss of traits often reflects the local biological community’s response to environmental changes1,3. Therefore, studying the functional traits of organisms within a community can provide insights into the assembly patterns of the community6, aiding in the understanding of the correlation between the environment and the functional structure of biological communities3,7. This, in turn, offers novel insights into the mechanisms underlying community formation. By focusing on functional traits, researchers can not only elucidate the factors contributing to the formation of insect communities but also identify the driving forces behind the evolution of community structures8.
Over the past few decades, regional and global trait datasets for various biological taxa have been sequentially established, with traits becoming an increasingly utilized research element within the fields of ecology, biodiversity conservation, ecosystem restoration, and landscape management1,5,9,10. Despite global efforts to construct trait databases, the majority of existing databases are primarily focused on plants, birds, and amphibians, representing relatively small taxonomic groups11,12,13,14,15. In contrast, focusing on insect taxa presents a formidable challenge due to the vast number of species, making the collection of research-valuable trait data a daunting task. Additionally, much of the existing research on insect traits is based on characteristics derived from literature16,17,18,19,20, such as trophic-defined groups or binary long- and short-winged groups, which are discrete and lose critical information. This is suboptimal for analyzing the impact of abiotic environmental filters on the composition of functional communities1,21,22. Therefore, it is imperative to measure accurate and comprehensive morphological characteristics of insects and establish a morphological trait database.
The Heteroptera represents a vast group within the hemimetabolous insects, with over 45,000 species identified globally23. Characterized by their extensive distribution and morphological diversity, they serve as ideal subjects for ecological studies21,24,25,26,27,28,29. Gossner et al. established a database containing mean values of 23 morphometric measurements for 179 heteropteran species collected from German grasslands30. This database laid the foundation for the development of a trait database for the Heteroptera, instilling confidence in subsequent research efforts. To expand upon this existing database, we measured 34 morphological characteristics of 307 Heteroptera species collected from a biodiversity priority area in China. To ensure the standardization and availability of trait datasets, this study employed the same measurement techniques and trait classification methods utilized in the morphological trait database of Heteroptera from German grasslands30. Furthermore, the value of measuring diverse morphological traits for further analysis of functional responses has been demonstrated by individual studies on single taxonomic groups, such as the effect of mantis hind tibia width31 and lepidopteran wing area32,33 on flight performance. Therefore, acquiring additional trait information is necessary to detect the response of arthropod communities to environmental changes or the impact of trait variation on ecosystem function. Thus, our study incorporated additional measurements of features such as the tibia width and wing area, enhancing the dataset’s comprehensiveness.
Methods
Sites
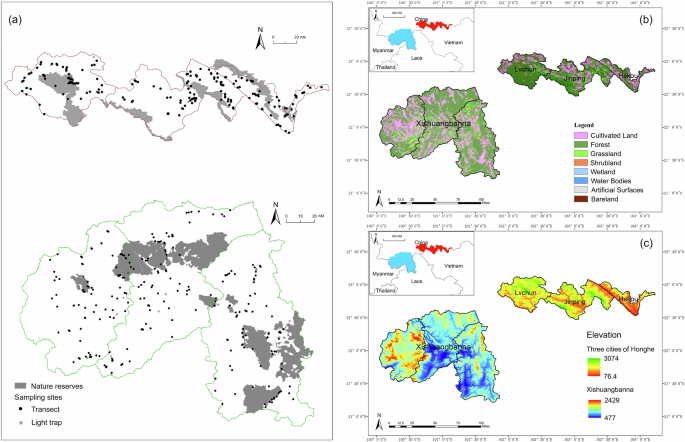
This study conducted sampling within two areas, Xishuangbanna Prefecture and three counties of Honghe Prefecture, located in the “Xishuangbanna Priority Conservation Area (42,858 km2, including six national nature reserves)” in the southwest region of China. The sampling sites included five national nature reserves (Fig. 1a). The study areas have been subject to long-term human activity, resulting in a complex landscape structure interlaced with urban areas, forests, and farmlands34,35 (Fig. 1b).
Xishuangbanna
Xishuangbanna Prefecture, located within Yunnan Province of China, encompasses an area of 19,124.5 km2, including two national nature reserves, with an elevation range from 477 meters to 2,429 meters (Fig. 1c). This region is characterized by a tropical monsoon climate.
Three cities in Honghe
Lüchun, Jinping, and Hekou are three counties located in the Honghe Prefecture of Yunnan Province, China, encompassing a total area of 8,105.86 km2, which includes three national nature reserves. Situated in a low-latitude subtropical plateau with a moist monsoon climate, their elevation ranges from 76.4 meters to 3,074 meters (Fig. 1c).
Experimental and sampling design
In this study, the research area was delineated into grid units of 10 kilometers each, with 1 to 2 sampling sites selected per working grid. At each sampling site, three transects of 100 meters in length were established. From July to October 2019, insect samples were collected using sweep netting and light trapping methods. Each study area underwent two rounds of sampling. All specimens collected were preserved in 75% ethanol solution and transported back to the laboratory for identification.
Traits acquisition and classification
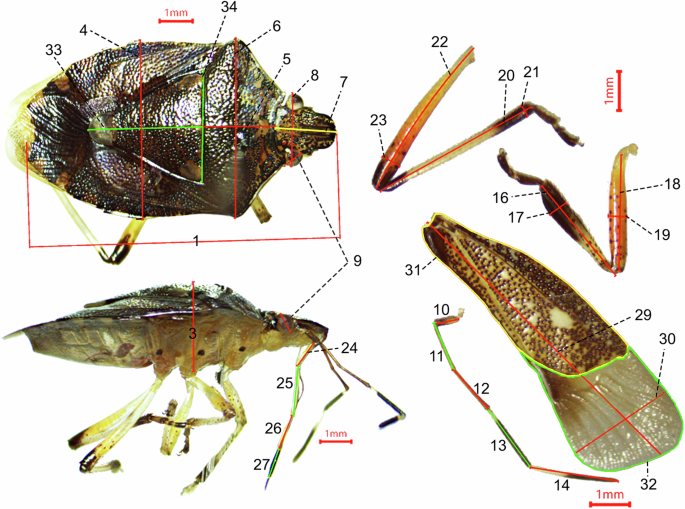
We employed a stereoscopic dissecting microscope equipped with a ToupCam digital camera (1/1.8” Sony Exmor CMOS sensor) and the ToupTek ToupView 3.7 software to take morphometric measures, the measurement precision is 0.01 mm. A total of 34 morphometric measurements were performed on each individual, with measurement locations and definitions following previous studies23,30,36. For details see Table 1 and Fig. 2.
Illustration of morphometric measurements taken on each specimen. The numbers correspond to morphometric measurement codes in Table 1.
For all sampled species within the Heteroptera, species with a sufficient number of individuals were subject to the morphological measurement of at least five specimens to incorporate intraspecific variability into our measurement of species characteristics. The sampling effort aimed to include both female and male individuals, with the sex ratio dependent on the sampling conditions. Individuals of different sexes were selected from the study samples and mounted on a work board using entomological pins to ensure the insect’s body remained parallel to the microscope objective. We disassembled the insect’s forelegs, hindlegs, antennae, and forewings for precise measurement, ensuring that the parts being measured were parallel to the objective to guarantee the reliability of the measurements. For species with an adequate number of samples, individuals with partial damage were excluded from measurement. For species with fewer than five individuals, only measurements of the damaged parts were omitted.
We identified 17 morphometric traits representing five characteristic categories30, including body size (body length, volume37,38), dispersal capability (relative wing length, ratio of membranous to leathery wing area, front tibia shape, hind tibia shape31, relative length of the hind femur39), resource utilization in feeding (relative rostrum length40,41,42,43, shape of the front femur44), habitat utilization (body size, relative length of the scutellum), and orientation ability (relative antenna length, relative eye size45). In addition to traits with demonstrated functionality, we incorporated several predictive traits, species with a greater proportion of membranous wing area are expected to exhibit enhanced dispersal capabilities32,33; anticipating the shape of the front tibia and hind tibia are related to dispersal capabilities, with species possessing broader tibiae exhibiting stronger dispersal abilities31; and significant morphological variation in the scutellum across different species of Heteroptera, its relative length is predicted to be associated with habitat utilization46,47,48. For more details see Table 2.
The 17 morphological traits were calculated from the 34 morphometric measurements (see Table 1). The detailed calculation formulas can be found in the “Description” column of Table 2. All computations were conducted using Microsoft Excel software.
Data Records
This dataset is a species-level data set of 34 morphometric measures from 4346 specimens of 307 species of Heteroptera. For 299 species (about 97%), at least five individuals were measured for each species. The comprehensive dataset is constituted by three discrete files: two of which are formatted in ‘.csv’ (Unicode UTF-8), and one in PDF format. All files are accessible via figshare49. Rows denote unique species records, while columns correspond to the variables provided.
HeteropteraMorphometricTraits_1
This file encompasses morphometric measurements conducted on both male and female specimens across 307 species within the Heteroptera, comprising 34 distinct morphological measurements. The file presents the mean, minimum, and maximum values of these measurements for the collective specimens of each species, as well as for individual female and male specimens, with all dimensions denoted in millimeters. Additionally, the dataset includes information on the number of individuals measured, as well as taxonomic data at the order and family levels.
HeteropteraMorphometricTraits_2
The morphological traits in the file, designated as HeteropteraMorphometricTraits_2 were derived through calculations based on 34 morphometric measurements (see Table 1) taken from specimens of 307 species within the Heteroptera. For each species, average, minimum, and maximum values were computed for the total specimens measured, as well as separately for female and male individuals, with all measurements presented in millimeters. Additionally, the file encompasses information on the number of individuals, as well as their respective orders and families.
HeteropteraFigures
This study presents a total of 310 photos of all measured species for verification purposes and to ensure the future usability of the data.
Technical Validation
All data contained within this dataset were provided by the authors. Field methods followed a standardized protocol (Supplementary Tables S1 and S2). To ensure comparability, future data reuse and synthesis, both the primary measurement methods and classification schemes for the traits were consistent with those of Gossner et al.30. Species identification was cross-checked by different taxonomic experts and at random by barcoding. Although the measurement of morphological traits involves a minimal degree of subjective intervention, the measurement of all trait data was conducted by the same individual to minimize subjective bias.
This study examined whether data for each trait contained abnormal values or outliers. In most instances, these issues were addressed by inspecting images of the species with attached scales and the specimens themselves. All specimens are stored in the Institute of Highland Forest Science, Chinese Academy of Forestry.
Usage Notes
The information allocated on the Figshare repository is a static version of the data last reviewed on June 2024, further updates and refinements planned for release within the same Figshare project, but the DOI could be different. A report record will be also included to control further updates. Anticipated future expansions include the incorporation of ecological trait dataset and site-specific information. Traits can be univocally linked to each species included in the latter datasets (or others) using the species Latin name.
Code availability
No custom code was used to generate or process the data in this study.
References
Mcgill, B. J., Enquist, B. J., Weiher, E. & Westoby, M. Rebuilding community ecology from functional traits. Trends in Ecology & Evolution 21, 178–185 (2006).
Webb, C. T., Hoeting, J. A., Ames, G. M., Pyne, M. I. & LeRoy Poff, N. A structured and dynamic framework to advance traits-based theory and prediction in ecology. Ecology Letters 13, 267–283 (2010).
Munoz, F. et al. The ecological causes of functional distinctiveness in communities. Ecology Letters 26, 1452–1465 (2023).
Luck, G. W., Lavorel, S., McIntyre, S. & Lumb, K. Improving the application of vertebrate trait-based frameworks to the study of ecosystem services. Journal of Animal Ecology 81, 1065–1076 (2012).
He, N. et al. Ecosystem Traits Linking Functional Traits to Macroecology. Trends in Ecology & Evolution 34, 200–210 (2019).
Loewen, C. J. G., Jackson, D. A. & Gilbert, B. Biodiversity patterns diverge along geographic temperature gradients. Global Change Biology 29, 603–617 (2023).
Kardol, P., Fanin, N. & Wardle, D. A. Long-term effects of species loss on community properties across contrasting ecosystems. Nature 557, 710–713 (2018).
Rosas-Ramos, N., Asís, J. D., Goula, M., Ballester-Torres, I. & Baños-Picón, L. Linear Landscape Elements and Heteropteran Assemblages within Mediterranean Vineyard Agroecosystems. Sustainability 14, 12435 (2022).
Gross, N. et al. Functional trait diversity maximizes ecosystem multifunctionality. Nat Ecol Evol 1 (2017).
Fontana, S., Rasmann, S., de Bello, F., Pomati, F. & Moretti, M. Reconciling trait based perspectives along a trait‐integration continuum. Ecology 102, e03472 (2021).
Mammola, S. et al. A trait database and updated checklist for European subterranean spiders. Sci Data 9, 236 (2022).
Phillips, H. R. P. et al. Global data on earthworm abundance, biomass, diversity and corresponding environmental properties. Sci Data 8, 136 (2021).
Lam‐Gordillo, O., Baring, R. & Dittmann, S. Establishing the South Australian Macrobenthic Traits (SAMT) database: A trait classification for functional assessments. Ecology and Evolution 10, 14372–14387 (2020).
Lam-Gordillo, O., Lohrer, A. M., Hewitt, J. & Dittmann, S. NZTD - The New Zealand Trait Database for shallow-water marine benthic invertebrates. Sci Data 10, 502 (2023).
Wang, H. et al. The China plant trait database version 2. Sci Data 9, 769 (2022).
Church, S. H., Donoughe, S., de Medeiros, B. A. S. & Extavour, C. G. A dataset of egg size and shape from more than 6,700 insect species. Sci Data 6, 104 (2019).
Borges, R. C., Padovani, K., Imperatriz-Fonseca, V. L. & Giannini, T. C. A dataset of multi-functional ecological traits of Brazilian bees. Sci Data 7, 120 (2020).
Middleton-Welling, J. et al. A new comprehensive trait database of European and Maghreb butterflies, Papilionoidea. Sci Data 7, 351 (2020).
Rajpurohit, S., Zhao, X. & Schmidt, P. S. A resource on latitudinal and altitudinal clines of ecologically relevant phenotypes of the Indian Drosophila. Sci Data 4, 170066 (2017).
Gossner, M. M. et al. A summary of eight traits of Coleoptera, Hemiptera, Orthoptera and Araneae, occurring in grasslands in Germany. Sci Data 2, 150013 (2015).
Simons, N. K., Weisser, W. W. & Gossner, M. M. Multi-taxa approach shows consistent shifts in arthropod functional traits along grassland land-use intensity gradient. Ecology 15-0616.1, https://doi.org/10.1890/15-0616.1 (2015).
Osorio-Canadas, S., Flores-Hernández, N., Sánchez-Ortiz, T. & Valiente-Banuet, A. Changes in bee functional traits at community and intraspecific levels along an elevational gradient in a Mexical-type scrubland. Oecologia 200, 145–158 (2022).
Schuh, R. T. & Weirauch, C. True Bugs of the World (Hemiptera: Heteroptera): Classification and Natural History. (Siri Scientific Press, 2020).
Andrew, N. R. & Hughes, L. Diversity and assemblage structure of phytophagous Hemiptera along a latitudinal gradient: predicting the potential impacts of climate change. Global Ecology and Biogeography 14, 249–262 (2005).
Jiang, K. et al. Diversity and conservation of endemic true bugs for four family groups in China. Diversity and Distributions 28, 2824–2837 (2022).
Torma, A. et al. Species and functional diversity of arthropod assemblages (Araneae, Carabidae, Heteroptera and Orthoptera) in grazed and mown salt grasslands. Agriculture, Ecosystems & Environment 273, 70–79 (2019).
Dorman, S. J., Schürch, R., Huseth, A. S. & Taylor, S. V. Landscape and climatic effects driving spatiotemporal abundance of Lygus lineolaris (Hemiptera: Miridae) in cotton agroecosystems. Agriculture, Ecosystems & Environment 295, 106910 (2020).
Seibold, S. et al. Arthropod decline in grasslands and forests is associated with landscape-level drivers. Nature 574, 671–674 (2019).
Torma, A., Gallé, R. & Bozsó, M. Effects of habitat and landscape characteristics on the arthropod assemblages (Araneae, Orthoptera, Heteroptera) of sand grassland remnants in Southern Hungary. Agriculture, Ecosystems & Environment 196, 42–50 (2014).
Gossner, M. M., Simons, N. K., Höck, L. & Weisser, W. W. Morphometric measures of Heteroptera sampled in grasslands across three regions of Germany: Ecological Archives E096‐102. Ecology 96, 1154–1154 (2015).
Zhao, X. et al. Petal-shaped femoral lobes facilitate gliding in orchid mantises. Current Biology 34, 183–189.e4 (2024).
Reid, H. E. et al. Wing flexibility reduces the energetic requirements of insect flight. Bioinspir. Biomim. 14, 056007 (2019).
Sappington, T. W. & Burks, C. S. Patterns of Flight Behavior and Capacity of Unmated Navel Orangeworm (Lepidoptera: Pyralidae) Adults Related to Age, Gender, and Wing Size. Environ Entomol 43, 696–705 (2014).
Chen, J. et al. Global land cover mapping at 30m resolution: A POK-based operational approach. ISPRS Journal of Photogrammetry and Remote Sensing 103, 7–27 (2015).
Chen, J., Ban, Y. & Li, S. Open access to Earth land-cover map. Nature 514, 434–434 (2014).
Wang, J. Y. Taxonomic study on the subfamily Ectrichodiinae (Hemiptera: Reduviidae) from China and reginos around China. (China Agricultural University, 2016).
Siemann, E. N., Tilman, D. & Haarstad, J. N. Abundance, diversity and body size: patterns from a grassland arthropod community. Journal of Animal Ecology 68, 824–835 (1999).
Friess, N., Gossner, M. M., Weisser, W. W., Brandl, R. & Brändle, M. Habitat availability drives the distribution–abundance relationship in phytophagous true bugs in managed grasslands. Ecology 98, 2561–2573 (2017).
Arnold, P. A., Cassey, P. & White, C. R. Functional traits in red flour beetles: the dispersal phenotype is associated with leg length but not body size nor metabolic rate. Functional Ecology 31, 653–661 (2017).
Imms, A. D., Richards, O. W. & W, R. O. Imms’ General Textbook of Entomology. Tenth Edition, Vol 1. Structure, Physiology and Development. (Chapman and Hall, 1977).
Imms, A. D., Richards, O. W. & Davies, R. G. IMMS’ General Textbook of Entomology: Volume I: Structure, Physiology and Development. (Springer Science & Business Media, 2012).
Cohen, A. C. Plant Feeding by Predatory Heteroptera: Evolutionary and Adaptational Aspects of Trophic Switching. in Zoophytophagous Heteroptera: Implications for Life History and Integrated Pest Management vol. none 1 (Entomological Society of America, 1996).
Roitberg, B. D. et al. The cost of being an omnivore: mandible wear from plant feeding in a true bug. Naturwissenschaften 92, 431–434 (2005).
Sweet, M. H. The biology and ecology of the Rhyparochrominae of New England (Heteroptera: Lygaeidae). (University of Connecticut, 1963).
Talarico, F., Romeo, M., Massolo, A., Brandmayr, P. & Zetto, T. Morphometry and eye morphology in three species of Carabus (Coleoptera: Carabidae) in relation to habitat demands. J Zoological System 45, 33–38 (2007).
Rivas, N. et al. Morphology and morphometry of the scutellum of six species in the genus Meccus (Hemiptera: Triatominae). Journal of Vector Ecology 39, 14–20 (2014).
Martínez, L. C., Plata—Rueda, A., Zanuncio, J. C., Leite, G. & Serrão, J. E. Morphology and Morphometry ofDemotispa neivai(Coleoptera: Chrysomelidae) Adults. Annals of the Entomological Society of America 106, 164–169 (2013).
Schwertner, C. F., Albuquerque, G. S. & Grazia, J. Descrição dos Estágios Imaturos de Acrosternum (Chinavia) ubicum Rolston (Heteroptera: Pentatomidae) e Efeito do Alimento no Tamanho e Coloração das Ninfas. Neotrop. Entomol. 31, 571–579 (2002).
Gao, S. et al. A Morphological traits dataset of Heteroptera sampled in biodiversity priority areas of southwest China. figshare https://doi.org/10.6084/m9.figshare.25376458.v1 (2024).
Acknowledgements
This project supported by the biodiversity investigation, observation and assessment program (2019–2023) of Ministry of Ecology and Environment of China. We thank Qing Wang, Chunying Tang, Meixian Zheng, Di Yan, Xiaoyu Yu, Jinyu Yue, Longkun Yang, Zhiwen Lin, and Siming Wang for their assistance in insect collection, as well as Zilan Li, Jiami Duan, Zhengmei Shi, and Shoucheng Bao for their help with measuring morphometric traits. We thank zhuo chen from the China Agricultural University for identifying Heteroptera species.
Author information
Authors and Affiliations
Contributions
S.G. and Y.C. jointly conceived the research idea. S.G. and W.Y. were responsible for revising, proofreading, and validating all datasets, compiling the data, and drafting the initial version of the manuscript. All authors collectively gathered and/or measured the traits data. Y.C. and Q.L. secured funding and organized the fieldwork. Every author contributed to the final manuscript version and approved the final draft for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, S., Yu, W., Tian, T. et al. A morphological traits dataset of Heteroptera sampled in biodiversity priority areas of Southwest China. Sci Data 11, 694 (2024). https://doi.org/10.1038/s41597-024-03556-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-024-03556-x
- Springer Nature Limited