LETTERS TO THE EDITOR
Chlorambucil plus rituximab as front-line therapy for
elderly and/or unfit chronic lymphocytic leukemia
patients: correlation with biologically-based risk
stratification
First-line treatment for young/fit patients with chronic
lymphocytic leukemia (CLL) is the combination of fludarabine, cyclophosphamide and rituximab (FCR), which
has improved these patients’ progression-free survival
and overall survival,1 but is poorly tolerated by elderly
patients or patients with comorbidities.2 Such patients
have been historically treated with chlorambucil, which
is well tolerated but does not improve survival.3 To
improve outcomes, chlorambucil has been combined
with anti-CD20 monoclonal antibodies. Three prospective studies4-6 and one retrospective7 one investigated the
combination of chlorambucil with rituximab (Chl-R) as
front-line treatment for elderly CLL patients or for
younger patients unsuitable for fludarabine-based therapies. Overall response rates ranging from 66% to 84%
have been reported, with complete response rates of 826% and progression-free survival from 16.3 to 34.7
months.
In the present GIMEMA study we conducted a retrospective analysis of Chl-R used as front-line treatment in
elderly (≥65 years) and/or unfit [Cumulative Illness
Rating Scale (CIRS) score >6, calculated before treatment]8 CLL patients treated in 15 different Italian hematology centers. The primary aim was to establish the efficacy and safety of Chl-R and to investigate whether certain CLL subsets could benefit more from this combination. All patients with a minimum follow-up of 12
months and with an Eastern Cooperative Oncology
Group (ECOG) score ≤2, treated between 2009 and 2011,
were enrolled; their data were collected by treating
physicians and inserted into the GIMEMA electronic
database.
The treatment schedule differed between centers. The
majority of patients (72/102) were treated according to
schedules previously reported by Foà et al.5 and Laurenti
et al.;7 the remaining patients were treated as reported by
Goede et al.6 or with local protocols. Primary endpoints
included the overall and complete response rates, evaluated according to the revised International Workshop
CLL 2008 criteria9, with the exception of a few patients
for whom a computed tomography scan and/or bone
marrow biopsy were not available. Secondary endpoints
included progression-free survival, time to retreatment,
overall survival, and toxicity evaluated according to
Common Terminology Criteria for Adverse Events version 4. Responses and outcome were correlated with
clinical and biological parameters. For subgroup analyses,
patients were classified as high-risk (patients with
del17p), intermediate-risk (patients with unmutated
IGHV and/or del11q) and low-risk (patients with mutated IGHV without del11q).10-12
Non-parametric tests were carried out for comparisons
and logistic regression was performed to adjust for the
effect of clinical and biological factors on the overall
response rate. Overall response rates were stratified
according to the immunophenotypic profile, fluorescence
in situ hybridization-based cytogenetic evaluation, IGHV
status, ECOG score and age. Survival distributions were
estimated using the Kaplan-Meier product limit estimator. Differences in overall survival, progression-free survival and time to retreatment were evaluated using the
log-rank test in univariate analyses and the Cox regression model in the adjusted analysis, after assessment of
proportionality of hazards. All tests were two-sided,
accepting P<0.05 as indicating a statistically significant
difference and confidence intervals (CI) were calculated
at a 95% level. All analyses were performed using SAS
software (release 9.4).
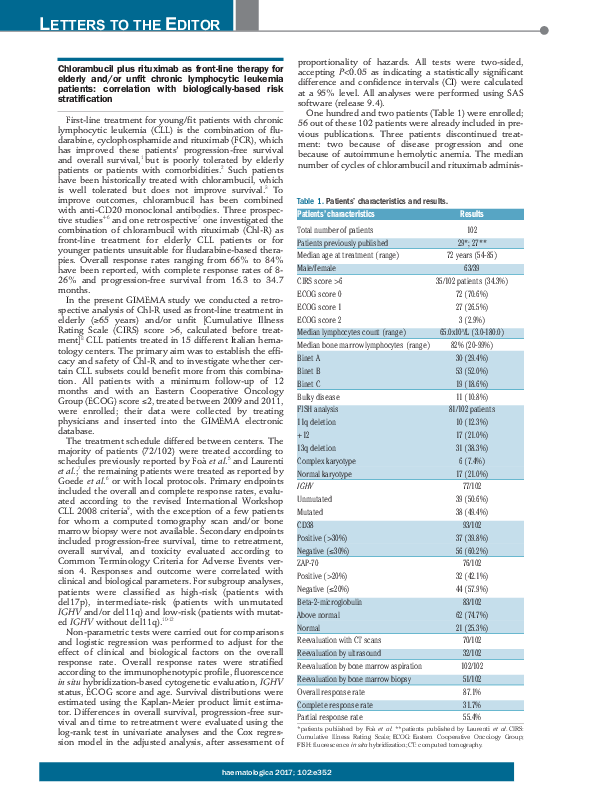
One hundred and two patients (Table 1) were enrolled;
56 out of these 102 patients were already included in previous publications. Three patients discontinued treatment: two because of disease progression and one
because of autoimmune hemolytic anemia. The median
number of cycles of chlorambucil and rituximab adminis-
Table 1. Patients’ characteristics and results.
Patients’ characteristics
Results
Total number of patients
102
Patients previously published
29*; 27**
Median age at treatment (range)
72 years (54-85)
Male/female
63/39
CIRS score >6
35/102 patients (34.3%)
ECOG score 0
72 (70.6%)
ECOG score 1
27 (26.5%)
ECOG score 2
3 (2.9%)
Median lymphocytes count (range)
65.0x109/L (3.0-180.0)
Median bone marrow lymphocytes (range)
82% (20-99%)
Binet A
30 (29.4%)
Binet B
53 (52.0%)
Binet C
19 (18.6%)
Bulky disease
11 (10.8%)
FISH analysis
81/102 patients
11q deletion
10 (12.3%)
+12
17 (21.0%)
13q deletion
31 (38.3%)
Complex karyotype
6 (7.4%)
Normal karyotype
17 (21.0%)
IGHV
77/102
Unmutated
39 (50.6%)
Mutated
38 (49.4%)
CD38
93/102
Positive (>30%)
37 (39.8%)
Negative (≤30%)
56 (60.2%)
ZAP-70
76/102
Positive (>20%)
32 (42.1%)
Negative (≤20%)
44 (57.9%)
Beta-2-microglobulin
83/102
Above normal
62 (74.7%)
Normal
21 (25.3%)
Reevaluation with CT scans
70/102
Reevaluation by ultrasound
32/102
Reevaluation by bone marrow aspiration
102/102
Reevaluation by bone marrow biopsy
51/102
Overall response rate
87.1%
Complete response rate
31.7%
Partial response rate
55.4%
*patients published by Foà et al. **patients published by Laurenti et al. CIRS:
Cumulative Illness Rating Scale; ECOG: Eastern Cooperative Oncology Group;
FISH: fluorescence in situ hybridization; CT: computed tomography.
haematologica 2017; 102:e352
�LETTERS TO THE EDITOR
tered was 8 (range, 2-12) and 6 (range, 1-9), respectively.
The median total dose of chlorambucil administered during treatment was 600 mg per patient (median dose 90
mg each cycle) and the median dose of rituximab was
4200 mg per patient (median dose 700 mg each cycle).
The dose of chlorambucil was reduced in 19% of
patients, while rituximab was decreased in only 5% of
patients.
On an intention-to-treat basis, the overall response rate
was 87.1%. Thirty-two patients (31.7%) obtained a clinical complete response and 56 patients (55.4%) a partial
response (Table 1). No statistically significant differences
in overall response rate were noted based on the variables analyzed (Table 2).
The median progression-free survival was reached at a
median time of 43.7 months (Figure 1A). Among 58
patients who experienced progression, 35 (60.3%)
received a second line of treatment after a median time of
72.3 months. Among all investigated clinical and biological characteristics, only an ECOG score of 2 (versus
ECOG score of 0-1) and IGHV unmutated status (versus
IGHV mutated) were bad prognosticators at univariate
analysis for progression-free survival and time to retreatment.
The median follow-up was 54 months, during which
the median overall survival was not reached. Estimated
survival rates after 48 and 60 months were 86.1% (95%
CI: 79.4-93.5) and 81.2% (95% CI: 72.4-91.2), respectively (Figure 1B). ECOG score of 2 (versus ECOG score of
0-1), CIRS score >6 (versus CIRS score ≤6) and normal
karyotype (versus +12 and del13q) showed negative
impacts on overall survival at univariate analysis.
The 72 patients with available IGHV and fluorescence
in situ hybridization data were classified as intermediaterisk (IGHV unmutated and/or del11q: 36 patients) and
low-risk (IGHV mutated without del11q: 36 patients);
there were no high-risk patients (del17p). The majority of
low-risk patients (54.9%) and only 18.7% of intermediate-risk patients remained free from progression 60
months after treatment; low-risk patients showed a significantly better progression-free survival than intermediate-risk patients: 65.8 months versus 35.2 months
(P=0.0116) (Figure 1C). A trend towards a better overall
survival was observed, but this has so far not reached sta-
Table 2. Time-dependent results.
Patients’ characteristics
ECOG score (n=102)
0 (n=72)
1 (n=27)
2 (n=3)
Age (n=102)
<70 (n=42)
>70 (n=60)
CIRS score (n=102)
1-6 (n=67)
≥7 (n=35)
FISH (n=81)
del 13q (n=31)
+12 (n=17)
del 11q (n=10)
complex karyotype(n=6)
normal (n=17)
IGVH mutation status (n=77)
Mutated (n=38)
Unmutated (n=39)
Bulky disease (n=100)
no (n=89)
yes (n=11)
CD38 (n=93)
<30% (n=56)
>30% (n=37)
ZAP-70 (n=76)
<20% (n=44)
>20% (n=32)
ORR (%)
88.73
85.19
66.67
P
value
0.38
PFS
(60 months
estimate)
40.42
33.43
0.00
P
value
<.0001
TTR
(60 months
estimate)
58.70
51.05
0.00
P
value
<.0001
OS
(60 months
estimate)
88.57
72.34
0.00
P
value
<.0001
88.10
86.44
1
35.25
40.87
0.5608
54.52
57.40
0.5771
90.41
73.04
0.2578n=
89.55
82.35
0.23
39.68
35.71
0.0460
56.84
54.53
0.1597
85.70
73.55
0.0124
83.87
94.12
90
100
82.35
0.82
40.60
52.28
20.00
33.33
nr
0.5079
66.74
53.57
37.50
44.44
nr
0.4587
87.10
94.12
90
83.3
58.72
0.0801
92.11
89.74
1
54.04
18.70
0.0335
74.22
32.15
0.0099
87.25
75.16
0.3348
88.64
81.82
0.62
40.03
18.18
0.1151
59.20
22.73
0.0151
83.15
71.59
0.1379
85.71
91.89
0.52
44.36
26.62
0.9033
60.41
46.54
0.8645
77.28
86.89
0.3756
88.64
87.50
1
37.67
18.75
0.3317
56.72
33.91
0.4958
81.10
73.47
0.5720
ORR: overall response rate; PFS: progression-free survival; TTT: time to retreatment; OS: overall survival; nr: not reached; ECOG: Eastern Cooperative Oncology Group;
CIRS: Cumulative Illness Rating Scale; FISH: fluorescence in situ hybridization.
haematologica 2017; 102:e353
�LETTERS TO THE EDITOR
A
B
C
D
Figure 1. Progression-free survival
and overall survival. (A) Progressionfree survival (PFS) in the study population. (B) Overall survival (OS) in the
study population. (C) PFS in low-risk
and intermediate-risk patients. (D) OS
in low-risk and intermediate-risk
patients.
tistical significance (Figure 1D).
Forty neutropenic events were reported in 33 patients
(32.3%), while anemia and thrombocytopenia were
recorded in only three patients. Grade 3-4 neutropenia
was reported in 14 patients (13.7%), seven were treated
with granulocyte colony-stimulating factor. Two grade 3
pulmonary infections were recorded during the treatment. One patient discontinued treatment because of
autoimmune hemolytic anemia and one patient experienced a grade 3 thrombocytopenia. No patient was
admitted to hospital, except for the patient with autoimmune hemolytic anemia. Forty episodes of non-hematologic toxicity, mainly grade 1-2, were reported, most
involving the respiratory and/or gastrointestinal tracts.
Grade 3-4 non-hematologic toxicities were recorded in
only ten patients (9.8%). Mild (grade 1-2) infusion-related reactions were reported in 30% of patients, in most
cases during the first administration of rituximab. Fifteen
patients died during follow-up: six from progressive disease or Richter transformation, six from myocardial
infarction or other cardio-pulmonary complications, and
three due to a solid tumor (glioblastoma, colon or thyroid
cancer) 26, 19 and 14 months after the end of treatment,
respectively.
The data presented support the recently reported findings from prospective clinical trials that Chl-R is well tolerated and effective in elderly/unfit CLL patients.4-7 When
comparing our results to those of these studies, we wish
to underline that our study is observational and characterized by less stringent measures for data collection and
absence of central review.
Our results are slightly different from those reported by
Hillmen et al.4 and Foà et al.5 with regards to both overall
response rate (87.1% versus 84% and 82.4%, respectively) and complete response rate (31.7% versus 10% and
19%, respectively). These differences could be explained
by the absence of del17p cases and a lower number of
patients (51%) harboring unmutated IGHV genes in our
series; in fact, in the studies by Hillmen et al.4 and Foà et
al.,5 del17p was present in 3% and 6% of cases, and
unmutated IGHV in 59% and 58% of patients, respectively. The higher complete response rate could also be
due to the fact that 31% of our patients were evaluated
by ultrasound and 50% by bone marrow biopsy, which
does not enable identification of nodular partial response,
whereas all the patients in the studies by Hillmen et al.4
and Foà et al.5 were evaluated by computed tomography
scan.
In the CLL11 GCLLSG trial, Goede et al.6 observed an
overall response rate of 65.9% and a complete response
rate of 8.3%; these less favorable results are possibly due
to the lower cumulative dose of chlorambucil (median
dose 400 mg) or to differences in the populations studied
(all patients were unfit and 7% of them carried del17p).
These differences could also explain the longer median
progression-free survival observed in our series compared
to those in other studies (43.7 months versus 23.5 months
in the study by Hillmen et al.,4 34.7 months in that by
Foà et al.,5 and 16.3 months in the one by Goede et al.6).
The Chl-R regimen was well tolerated in all studies,
with dose reductions or interruptions being recorded in
only 24% of patients in our series and in 25% of patients
in the series of Hillmen et al.4 and Foà et al.5 Grade 3-4
neutropenia occurred in 13.7% of our patients and in
19.6%, 28% and 41% of patients in the series reported
by Foà et al.,5 Goede et al.,6 and Hillmen et al.,4 respective-
haematologica 2017; 102:e354
�LETTERS TO THE EDITOR
ly. The greater incidence of neutropenia reported by
Goede et al. and Hillmen et al. could be related to the
greater number of unfit patients6 or the higher chlorambucil dose.4 Serious non-hematologic adverse events
were rare in all studies; we observed no grade 3-4 infusion-related reactions, similarly to the other series.4-7
A recent study by Rossi et al.12 showed that CLL
patients harboring mutated IGHV genes but neither
del11q or del17p have a very low risk and can experience
durable remissions after front-line FCR. The impact of
IGHV status in patients treated first-line with FCR has
been confirmed in two other studies.13,14 Subgroup analysis of the patients investigated in our study showed similar results. These data suggest that the Chl-R protocol is
particularly effective for the treatment of elderly or unfit
CLL patients with this highly favorable biologicallybased prognostic profile.
In conclusion, treatment of elderly or unfit CLL
patients with the Chl-R regimen is associated with low
toxicity, high overall response rate and durable progression-free survival. Particularly good results are achieved
in CLL patients with a mutated IGHV profile not carrying
del17p or del11q, suggesting that in this low-risk subset
Chl-R could represent a particularly promising therapeutic option, in view of its safety, efficacy and low treatment costs.
Luca Laurenti,1 Idanna Innocenti,1 Francesco Autore,1
Stefania Ciolli,2 Francesca Romana Mauro,3 Donato Mannina,4
Giovanni Del Poeta,5 Giovanni D’Arena,6 Massimo Massaia,7
Marta Coscia,8 Stefano Molica,9 Gabriele Pozzato,10
Dimitar G. Efremov,11 Barbara Vannata,1 Roberto Marasca,12
Pietro Galieni,13 Antonio Cuneo,14 Sonia Orlando,15
Alfonso Piciocchi,15 Riccardo Boncompagni,2 Donatella Vincelli,16
Anna Marina Liberati,17 Filomena Russo18 and Robin Foa’3
1
Università Cattolica del Sacro Cuore, Roma; 2Policlinico di Careggi,
Firenze; 3Ematologia, Policlinico Umberto 1, Università "Sapienza",
Roma; 4A.O.U. Riuniti "Papardo Piemonte", Messina; 5Ospedale S.
Eugenio, Roma; 6Hematology and Stem Cell Transplantation Unit,
IRCCS Centro di Riferimento Oncologico della Basilicata, Rionero in
Vulture; 7Ospedale Mauriziano e Università di Torino; 8A.O. Città
della Salute e della Scienza S. Giovanni Battista, Torino; 9Azienda
Ospedaliera Pugliese Ciaccio, Catanzaro; 10Ospedale Maggiore,
Trieste; 11ICGEB, Trieste; 12Centro Oncologico Modenese, Modena;
13
P.O. "C. e G. Mazzoni", Ascoli; 14A.O. - Arcispedale S. Anna,
Ferrara; 15Fondazione GIMEMA; 16A.O. "Bianchi-MelacrinoMorelli", Reggio Calabria; 17Azienda Ospedaliera - S. Maria Di
Terni, Terni and 18Ematologia CTMO Università degli Studi di Parma,
Italy
Funding: this study was partly supported by the Italian Association
for Cancer Research (AIRC) Special Program Molecular Clinical
Oncology, 5 x 1000, MCO1007 (to RF).
Correspondence: luca.laurenti@unicatt.it
doi:10.3324/haematol.2016.156901
Information on authorship, contributions, and financial & other disclosures was provided by the authors and is available with the online version
of this article at www.haematologica.org.
References
1. Hallek M, Fischer K, Fingerle-Rowson G, et al. Addition of rituximab
to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet.
2010;376(9747):1164–1174.
2. Eichhorst B, Goede V, Hallek M. Treatment of elderly patients with
chronic lymphocytic leukemia. Leuk Lymphoma. 2009;50(2):171178.
3. Eichhorst BF, Busch R, Stilgenbauer S, et al. First-line therapy with
fludarabine compared with chlorambucil does not result in a major
benefit for elderly patients with advanced chronic lymphocytic
leukemia. Blood. 2009;114(16):3382-3391.
4. Hillmen P, Gribben JG, Follows GA, et al. Rituximab plus chlorambucil as first-line treatment for chronic lymphocytic leukemia: final
analysis of an open-label phase II study. J Clin Oncol. 2014;
32(12):1236-1241.
5. Foà R, Del Giudice I, Cuneo A, et al. Chlorambucil plus rituximab
with or without maintenance rituximab as first-line treatment for
elderly chronic lymphocytic leukemia patients. Am J Hematol. 2014;
89(5):480-486.
6. Goede V, Fischer K, Bosch F, et al. Updated survival analysis from the
CLL11 study: obinutuzumab versus rituximab in chemoimmunotherapy-treated patients with chronic lymphocytic leukemia.
Abstract ASH 2015. Blood. 2015;126:1733.
7. Laurenti L, Vannata B, Innocenti I, et al. Chlorambucil plus rituximab
as front-line therapy in elderly/unfit patients affected by B-cell chronic lymphocytic leukemia: results of a single-centre experience.
Mediterr J Hematol Infect Dis. 2013;5(1):e2013031.
8. Miller MD, Paradis CF, Houck PR, et al. Rating chronic medical illness burden in geropsychiatric practice and research: application of
the Cumulative Illness Rating Scale. Psychiatry Res. 1992;41(3):237248.
9. Hallek M, Cheson BD, Catovsky D, et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the
International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines.
Blood. 2008;111(12):5446-5456.
10. Döhner H, Stilgenbauer S, Benner A, et al. Genomic aberrations and
survival in chronic lymphocytic leukemia. N Engl J Med. 2000;
343(26):1910-1916.
11. Hamblin TJ, Davis Z, Gardiner A, et al. Unmutated Ig V(H) genes are
associated with a more aggressive form of chronic lymphocytic
leukemia. Blood. 1999;94(6):1848-1854.
12. Rossi D, Terzi-di-Bergamo L, De Paoli L, et al. Molecular prediction
of durable remission after first-line fludarabine - cyclophosphamide rituximab in chronic lymphocytic leukemia. Blood. 2015;
126(16):1921-1924.
13. Fischer K, Bahlo J, Fink AM, et al. Long-term remissions after FCR
chemoimmunotherapy in previously untreated patients with CLL:
updated results of the CLL8 trial. Blood. 2016;127(2):208-215.
14. Jain N, O'Brien S. Targeted therapies for CLL: Practical issues with
the changing treatment paradigm. Blood Rev. 2016;30(3):233-244.
haematologica 2017; 102:e355
�
 Donato Mannina
Donato Mannina