Acids-Bases-Salts @Kidegalize Network_077203774 (1).pdf
- 1. ACIDS, BASES & SALTS ACIDS, BASES & SALTS Charles Kidega Kidegalize Network +256(0)702816081 es.gulu1910@gmail.com
- 2. Acids An acid is a compound which when dissolved in water produces hydrogen ions as the only positively charged ions e.g. sulphuric acid (H2SO4), hydrochloric acid(HCl), nitric acid(HNO3), phosphoric acid (H3PO4) and carbonic acid (H2CO3).
- 3. Basicity of an acid Basicity of an acid is the number of hydrogen ions produced by one molecule of an acid when dissolved in water. • Monobasic acids: acids that produces only one hydrogen ions when dissolved in water e.g. nitric acid and hydrochloric acid.
- 4. • Dibasic acids: acids that produces two hydrogen ions when dissolved in water e.g. sulphuric acid, carbonic acid, oxalic acid. • Tribasic acids: acids that produces three hydrogen ions when dissolved in water e.g. phosphoric acid.
- 5. Types of acids • Strong acids: are acids which ionize completely when dissolved in water e.g. hydrochloric acid, nitric acid and sulphuric acid. HCl(aq) H+(aq) + Cl-(aq) HNO3(aq) H+(aq) + NO3 -(aq) H2SO4(aq) 2H+(aq) + SO4 2-(aq)
- 6. • Weak acids: are acids which ionize partially when dissolved in water e.g. carbonic acid, ethanoic acid, sulphurous acid.
- 7. Remember: Solution containing a small amount of acid dissolved in water is dilute whereas solution containing large amount of acid dissolved in water is concentrated.
- 8. Properties of acids • Acids have a sour taste. • Acids are soluble in water. • Acids are corrosive. • Acids turn blue litmus paper red/ pink. • Acids have pH less than 7.
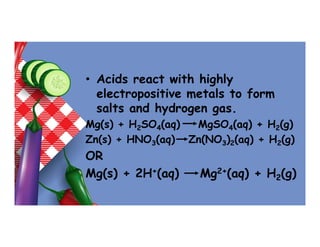
- 9. • Acids react with highly electropositive metals to form salts and hydrogen gas. Mg(s) + H2SO4(aq) MgSO4(aq) + H2(g) Zn(s) + HNO3(aq) Zn(NO3)2(aq) + H2(g) OR Mg(s) + 2H+(aq) Mg2+(aq) + H2(g)
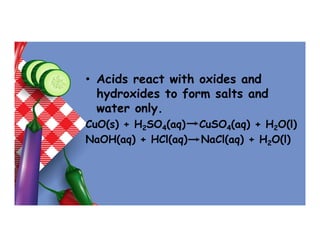
- 10. • Acids react with oxides and hydroxides to form salts and water only. CuO(s) + H2SO4(aq) CuSO4(aq) + H2O(l) NaOH(aq) + HCl(aq) NaCl(aq) + H2O(l)
- 11. • Acids react with carbonates and hydrogencarbonates to form salts, water and carbon dioxide. CuCO3(s) + 2HNO3(aq) Cu(NO3)2(aq) + H2O(l) + CO2(g) Observation: Bubbles of a colourless gas observed.
- 12. • Acids react with sulphites and hydrogensulphite to form salts, water and sulphur dioxide. 2H+(aq) + SO3 2-(aq) H2O(l) + SO2(g) H+(aq) + HSO3 -(aq) H2O(l) + SO2(g) Observation: Bubbles of a colourless gas observed.
- 13. Bases and alkalis A base is an oxide or hydroxide of a metal that reacts with an acid to form salt and water only. An alkali is a compound which when dissolved in water forms hydroxide ions, OH- as the only negatively charged ions e.g. sodium oxide, potassium oxide, sodium hydroxide,
- 14. Potassium hydroxide, ammonium hydroxide and calcium hydroxide. Alkalis are soluble bases. There are two types of alkalis: • Strong alkalis: are alkalis which completely ionized in solution e.g. sodium hydroxide and potassium hydroxide.
- 15. • Weak alkalis: are alkalis which partially ionizes in solution e.g. ammonium hydroxide. Remember: Bases are proton acceptors whereas acids are proton donators.
- 16. Properties of alkalis • Alkalis have bitter taste. • They turn red litmus paper blue. • Alkalis reacts with acids (neutralization reaction) to form salts and water only. NaOH(aq) + HCl(aq) NaCl(aq) + H2O(l) • Alkalis liberates ammonia gas when heated with ammonium salts
- 17. NaOH(aq) + NH4Cl(s) NaCl(aq) + NH3(g) + H2O(l) • Alkalis react with Ca2+, Mg2+, Zn2+, Al3+, Pb2+ ions to form white precipitate, Cu2+ forms blue precipitate, Fe2+ forms dirty green precipitate and Fe3+ forms brown precipitate.
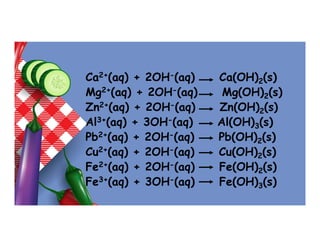
- 18. Ca2+(aq) + 2OH-(aq) Ca(OH)2(s) Mg2+(aq) + 2OH-(aq) Mg(OH)2(s) Zn2+(aq) + 2OH-(aq) Zn(OH)2(s) Al3+(aq) + 3OH-(aq) Al(OH)3(s) Pb2+(aq) + 2OH-(aq) Pb(OH)2(s) Cu2+(aq) + 2OH-(aq) Cu(OH)2(s) Fe2+(aq) + 2OH-(aq) Fe(OH)2(s) Fe3+(aq) + 3OH-(aq) Fe(OH)3(s)
- 19. Remember: Zn(OH)2 is soluble in both excess ammonia solution and sodium hydroxide solution whereas Al(OH)3 and Pb(OH)2 are soluble in excess sodium hydroxide solution only. A colourless solution is formed. Cu(OH)2 is soluble in excess ammonia forming a deep blue solution.
- 20. Zn(OH)2(s) + 2OH-(aq) Zn(OH)4 2-(aq) Zincate ions Zn(OH)2(s) + 4NH3(aq) Zn(NH3)4 2+(aq) + 2OH-(aq) Tetraamminezinc ions Al(OH)3(s) + OH-(aq) Al(OH)4 -(aq) Aluminate ions Pb(OH)2(s) + 2OH-(aq) Pb(OH)4 2-(aq) Plumbate ions Cu(OH)2(s) + 4NH3(aq) Cu(NH3)4 2+(aq) + 2OH-(aq) Tetraamminecopper(II) ions
- 21. • Sodium hydroxide and potassium hydroxide are deliquescent. Both absorbs water from the atmosphere to form solutions and thus absorbs carbon dioxide from the atmosphere to form sodium carbonate and potassium carbonate (both are white crystalline solids).
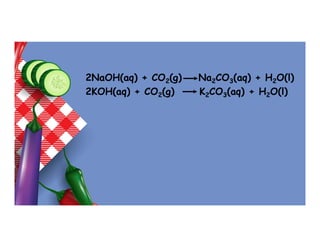
- 22. 2NaOH(aq) + CO2(g) Na2CO3(aq) + H2O(l) 2KOH(aq) + CO2(g) K2CO3(aq) + H2O(l)
- 23. pH of solutions The pH of a solution is a measure of the number of hydrogen ions in that solution. The lower the pH number, the more hydrogen ions there are, and thus the more acidic the solution. Strong acids have low pH values whereas strong bases have high pH values and neutral solutions show pH = 7.
- 24. Indicators Indicators are substances which changes colour according to the pH of the medium in which it is added. Common indicators are: Indicator Colour in acid Colour in alkaline Phenolphthalein Colourless Pink Methyl orange Red Yellow Litmus paper Red Blue
- 25. Salts A salt is a compound formed when either all or part of the ionisable hydrogen ions of an acid is replaced by a metallic ion or ammonium ion. There are two types of salts: • Normal salts: are salts formed when all the ionisable hydrogen ions of an acid is replaced by a metallic ion or ammonium ion e.g. sodium
- 26. chloride, calcium nitrate, zinc sulphate, ammonium chloride etc. • Acid salts: are salts formed when part of the ionisable hydrogen ions of an acid is replaced by a metallic ion or ammonium ion e.g. sodium hydrogensulphate, magnesium hydrogencarbonate etc.
- 27. Remember: Acid salts are formed by dibasic acids like H2SO4, H2CO3, H2SO3 or by tribasic acids like H3PO4.
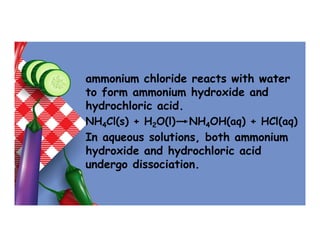
- 28. Nature of salts in water • Acid salts dissolve in water to form acidic solutions because hydrogen ion is produced. NaHCO3(aq) Na+(aq) + H+(aq) + CO3 2-(aq) • Normal salts dissolve in water to form neutral solutions. However, ammonium chloride/ ammonium nitrate dissolve in water to form acidic solution. This is because
- 29. ammonium chloride reacts with water to form ammonium hydroxide and hydrochloric acid. NH4Cl(s) + H2O(l) NH4OH(aq) + HCl(aq) In aqueous solutions, both ammonium hydroxide and hydrochloric acid undergo dissociation.
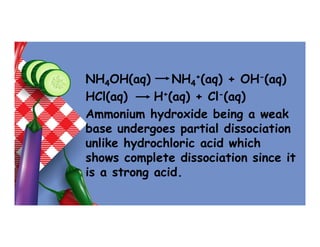
- 30. NH4OH(aq) NH4 +(aq) + OH-(aq) HCl(aq) H+(aq) + Cl-(aq) Ammonium hydroxide being a weak base undergoes partial dissociation unlike hydrochloric acid which shows complete dissociation since it is a strong acid.
- 31. This results into a solution of ammonium chloride having more hydrogen ions compared to the hydroxide ions formed by the weak base. So the solution is overall acidic.
- 32. Also sodium carbonate dissolves in water to form an alkaline solution. This is because sodium carbonate reacts with water to form sodium hydroxide and carbonic acid. Na2CO3(s) + 2H2O(l) 2NaOH(aq) + H2CO3(aq) In aqueous solutions, both sodium hydroxide and carbonic acid undergo dissociation.
- 33. NaOH(aq) Na+(aq) + OH-(aq) H2CO3(aq) 2H+(aq) + CO3 2-(aq) Carbonic acid being a weak acid undergoes partial dissociation unlike sodium hydroxide which shows complete dissociation since it is a strong base.
- 34. This results into a solution of sodium carbonate having more hydroxide ions compared to the hydrogen ions formed by the weak acid. So the solution is overall alkaline.
- 35. Water of crystallisation Water of crystallization is the definite amount of water with which some substances combine chemically to form crystals from their solutions. A salt without water of crystallization is called anhydrous salt whereas a hydrated salt is a
- 36. salt that contains the water of crystallization e.g. sodium carbonate – 10 – water, Na2CO3.10H2O; copper(II) sulphate – 5 – water, CuSO4.5H2O; iron(II) sulphate – 7 – water, FeSO4.7H2O
- 37. Exposure of salts to air Salts behave differently when exposed to air. Some lose their water of crystallization to the atmosphere and others absorb water vapour from the atmosphere. • A deliquescent substance absorbs water from the atmosphere and dissolves in it to form a solution
- 38. e.g. iron(III) chloride, potassium hydroxide, sodium hydroxide, zinc chloride, calcium chloride. • A hygroscopic substance absorbs water from the water e.g. all deliquescent substances, calcium oxide, concentrated sulphuric acid.
- 39. Remember: Deliquescent and hygroscopic substances are used as drying agents because of their ability to absorb water from the surrounding.
- 40. • Efflorescent substance loses water to the atmosphere e.g. sodium carbonate-10-water, copper(II) sulphate-5-water, sodium sulphate-10-water, iron(II) sulphate-7-water.
- 41. Preparation of soluble salts Soluble salts of copper, lead, iron, magnesium, aluminium and zinc are prepared by the general steps: Add the metal(Mg, Zn, Fe), metal oxide, metal carbonate or metal hydroxide to an appropriate acid until when the solid is in excess. Heat if necessary.
- 42. Filter off the excess solid. Saturate the filtrate by heating to evaporate off most of the water. Cool the saturated solution until when the crystals form. Filter off the crystals. Wash the crystals with distilled
- 43. water to remove any soluble impurities. Dry the crystals using filter papers.
- 44. Example (UNEB 2003/P2/14(a)) Describe how a dry sample of copper(II) sulphate may be prepared from copper(II) oxide. Possible Answer To warm dilute sulphuric acid in a beaker, copper(II) oxide is added little at a time while stirring the
- 45. mixture until in excess. Copper(II) sulphate is produced according to the equation: CuO(s) + H2SO4(aq) CuSO4(aq) + H2O(l) The excess copper(II) oxide is filtered off and the filtrate is saturated by heating.
- 46. The saturated solution is left to cool until when the crystals form. The crystals are filtered off, washed with distilled water to remove any soluble impurities and dried by pressing between filter papers.
- 47. Preparation of insoluble salts Insoluble salts are prepared by precipitation/ double decomposition. Precipitation involves reacting two soluble salts to form soluble salt and an insoluble salt. The insoluble salt is obtained as a residue after filtering. The residue is washed with distilled
- 48. water to remove any soluble impurities and dried by pressing between filter papers. Example (UNEB 2006/P2/12(c)) Briefly describe how a pure sample of calcium carbonate can be prepared.(Diagrams not required)
- 49. Possible Answer To a solution of calcium chloride in a beaker, aqueous sodium carbonate is added a little at a time while stirring the mixture until precipitation stops. Calcium carbonate is formed according to the equation:
- 50. Ca2+(aq) + CO3 2-(aq) CaCO3(s) Filter out the calcium carbonate from the mixture. Wash the residue with distilled water and dry it using filter paper.
- 51. Preparing salts by neutralization The salts of potassium, sodium and ammonium are prepared by neutralization. Neutralization is where an acid is reacted with an alkali to form a salt and water only.
- 52. Example 1(UNEB 1992/P2/13(c)) Describe how a dry sample of sodium chloride can be prepared in the laboratory. Possible Answer Titrate a known volume(V1cm3) of a standard sodium hydroxide solution with a standard hydrochloric acid
- 53. using phenolphthalein indicator. Sodium chloride is formed according to the equation: NaOH(aq) + HCl(aq) NaCl(aq) + H2O(l) Note the volume(V2cm3) of the acid at the end point. To a fresh volume(V1cm3) of the sodium hydroxide, add a volume of hydrochloric acid equal to V2cm3 and stir.
- 54. Heat the mixture to saturation point. Allow the saturated solution to cool in order for the crystals to form. Filter, wash with distilled water and dry them between filter papers.
- 55. Example 2(UNEB 2008/P2/13(b)) Outline how a pure dry sample of sodium hydrogensulphate can be prepared in the laboratory (No equation or diagram required). Possible Answer Titrate a known volume(V1cm3) of a standard sodium hydroxide solution with a standard sulphuric acid
- 56. using phenolphthalein indicator. Note the volume(V2cm3) of the acid at the end point. To a fresh volume(V1cm3) of the sodium hydroxide, add a volume of sulphuric acid equal to V2cm3 and stir.
- 57. Heat the mixture to saturation point. Allow the saturated solution to cool in order for the crystals to form. Filter, wash with distilled water and dry them between filter papers.
- 58. Direct synthesis Direct synthesis can be used to prepare both soluble and insoluble salts e.g. • Iron(III) chloride is formed when dry chlorine is passed over heated iron metal. 2Fe(s) + 3Cl2(g) 2FeCl3(s)
- 60. • Zinc sulphide, copper(II) sulphide and iron(II) sulphide are formed when a mixture of the metal and sulphur is heated. Zn(s) + S(s) ZnS(s) Cu(s) + S(s) CuS(s) Fe(s) + S(s) FeS(s)
- 61. Effect of heat on carbonates • Ammonium carbonate decomposes on heating to form ammonia gas, carbon dioxide and water. (NH4)2CO3(s) NH3(g) + CO2(g) + H2O(l) • Sodium carbonate and potassium carbonate do not decompose on heating.
- 62. • Magnesium carbonate and calcium carbonate decomposes to form respective metal oxide and carbon dioxide. MgCO3(s) MgO(s) + CO2(g) CaCO3(s) CaO(s) + CO2(g)
- 63. Observation: White solid forms white residue. • Zinc carbonate decomposes to form zinc oxide and carbon dioxide. ZnCO3(s) ZnO(s) + CO2(g) Observation: White powder turns to yellow residue when hot and white on cooling.
- 64. • Lead(II) carbonate decomposes to form lead(II) oxide and carbon dioxide. PbCO3(s) PbO(s) + CO2(g) Observation: White solid turned to reddish-brown residue when hot and yellow on cooling.
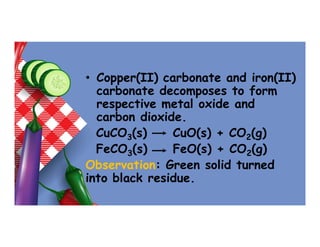
- 65. • Copper(II) carbonate and iron(II) carbonate decomposes to form respective metal oxide and carbon dioxide. CuCO3(s) CuO(s) + CO2(g) FeCO3(s) FeO(s) + CO2(g) Observation: Green solid turned into black residue.
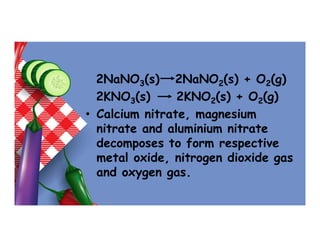
- 66. Effect of heat on nitrates • Ammonium nitrate decomposes to form dinitrogen oxide and water. NH4NO3(s) N2O(g) + 2H2O(l) • Sodium nitrate and potassium nitrate decomposes to form respective metal nitrite and oxygen gas.
- 67. 2NaNO3(s) 2NaNO2(s) + O2(g) 2KNO3(s) 2KNO2(s) + O2(g) • Calcium nitrate, magnesium nitrate and aluminium nitrate decomposes to form respective metal oxide, nitrogen dioxide gas and oxygen gas.
- 68. 2Ca(NO3)2(s) 2CaO(s) + 4NO2(g) + O2(g) 2Mg(NO3)2(s) 2MgO(s) + 4NO2(g) + O2(g) 4Al(NO3)2(s) 2Al2O3(s) + 12NO2(g) + 3O2(g) Observation: White solid forms white residue and brown gas given off. • Lead(II) nitrate decomposes with cracking sound to form lead(II) oxide, nitrogen dioxide
- 69. and oxygen gas. 2Pb(NO3)2(s) 2PbO(s) + 4NO2(g) + O2(g) Observation: White solid turned to reddish-brown residue when hot and yellow on cooling and brown gas given off. • Zinc nitrate decomposes on heating to form zinc oxide,
- 70. nitrogen dioxide and oxygen gas. 2Zn(NO3)2(s) 2ZnO(s) + 4NO2(g) + O2(g) Observation: White solid turned to yellow residue when hot and white on cooling and brown gas given off.
- 71. • Copper(II) nitrate decomposes on heating to form copper(II) oxide, nitrogen dioxide and oxygen gas. 2Cu(NO3)2(s) 2CuO(s) + 4NO2(g) + O2(g) Observation: Blue solid turned to black residue and brown gas given off.
- 72. • Iron(II) nitrate decomposes on heating to form iron(II) oxide, nitrogen dioxide and oxygen gas. 2Fe(NO3)2(s) 2FeO(s) + 4NO2(g) + O2(g) Observation: Green solid turned to black residue and brown gas given off.
- 73. • Mercury(II) nitrate and silver nitrate decomposes to form respective metal, nitrogen dioxide gas and oxygen gas. 2AgNO3(s) 2Ag(s) + 2NO2(g) + O2(g) Hg(NO3)2(s) Hg(s) + 2NO2(g) + O2(g)
- 74. Effect of heat on sulphates • Ammonium sulphate decomposes to form ammonia gas and sulphuric acid (NH4)2SO4(s) 2NH3(g) + H2SO4(aq) • Potassium sulphate, sodium sulphate and magnesium sulphate are stable and do not decompose on heating.
- 75. • Copper(II) sulphate, zinc sulphate and lead(II) sulphate decomposes to form metal oxides and sulphur trioxide. CuSO4(s) CuO(s) + SO3(g) ZnSO4(s) ZnO(s) + SO3(g) PbSO4(s) PbO(s) + SO3(g)
- 76. • Iron(II) sulphate decomposes on strong heating to form iron(III) oxide, sulphur dioxide and sulphur trioxide. 2FeSO4(s) Fe2O3(s) + SO3(g) + SO2(g) Observation: White solid turned to reddish-brown residue.
- 77. Remember: When hydrated iron(II) sulphate (green solid) is gently heated, anhydrous iron(II) sulphate (white powder) is formed and on strong heating, iron(III) oxide, sulphur dioxide and sulphur trioxide are formed whereas
- 78. hydrated copper(II) sulphate (blue solid) forms anhydrous copper(II) sulphate (white powder) on gentle heating, then copper(II) oxide and sulphur trioxide on strong heating.
- 79. Effect of heat on hydroxides • Sodium hydroxide and potassium hydroxide are stable and do not decompose on heating. • When zinc hydroxide is strongly heated, the white solid turns yellow when hot and white on cooling. Zn(OH)2(s) ZnO(s) + H2O(l)
- 80. • When lead(II) hydroxide is strongly heated, the white solid turns reddish-brown on heating and yellow on cooling. Pb(OH)2(s) PbO(s) + H2O(l)
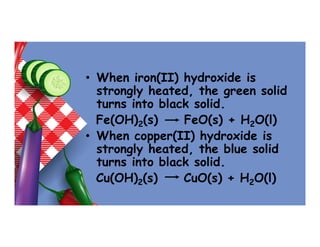
- 81. • When iron(II) hydroxide is strongly heated, the green solid turns into black solid. Fe(OH)2(s) FeO(s) + H2O(l) • When copper(II) hydroxide is strongly heated, the blue solid turns into black solid. Cu(OH)2(s) CuO(s) + H2O(l)
- 82. Solubility of salts Solubility of a salt is the mass of salt(solute) required to saturate 100g of water(solvent) at a particular temperature. Solubility of most salts increase with increase in temperature. • Solute is a substance that dissolves in a solvent to form a solution.
- 83. • A solution is a uniform mixture of a solute and a solvent. • A saturated solution is one that contains the maximum amount of a solute in a given amount of a solvent at a particular temperature.
- 84. • Supersaturated solution is one which has dissolved more solute than it can normally hold in the presence of undissolved solute at a particular temperature.
- 85. Determination of solubility of a salt Prepare a saturated solution, measure and record its temperature. Weigh an empty dish and transfer the saturated solution into the dish. Reweigh the dish to determine the mass of the dish and the saturated solution. Evaporate the solution to dryness and weigh the dish again to determine the mass of the salt and the dish.
- 86. Results Mass of empty = xg Mass of dish and saturated solution = yg Mass of dish and salt = zg Mass of salt = (z – x)g Mass of water = (y – z)g
- 87. Treatment of results (y – z)g of H2O dissolve (z – x)g of salt 100g of H2O dissolve (z − x) (y − z) × 100g of salt Solubility of the salt is (z − x) (y − z) × 100g per 100g of water.
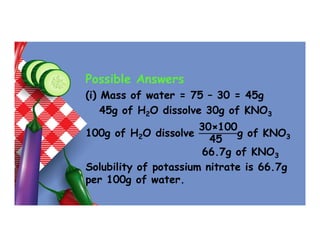
- 88. Example 1 (UNEB 1987/P2/11(b)) 75g of a saturated solution contains 30g of salt. Calculate (i) the solubility of the salt. (ii) the percentage of the salt in the saturated solution.
- 89. Possible Answers (i) Mass of water = 75 – 30 = 45g 45g of H2O dissolve 30g of KNO3 100g of H2O dissolve 30×100 45 g of KNO3 66.7g of KNO3 Solubility of potassium nitrate is 66.7g per 100g of water.
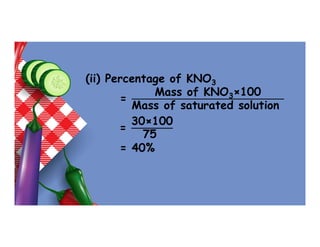
- 90. (ii) Percentage of KNO3 = Mass of KNO3×100 Mass of saturated solution = 30×100 75 = 40%
- 91. Example 2 (UNEB 1997/P1/6) The solubility of salt W is 35g per 100cm3 of water at 20oC. The mass of W in 40cm3 of water at the same temperature is A. 7.0g B. 14.0g C. 87.5g D. 114.3g
- 92. Possible Answers 100cm3 of H2O dissolve 35g of W 40cm3 of H2O dissolve 35 × 40 100 g of W = 14g of W Correct option is B
- 93. Example 3 200g of water at 55oC are saturated by 150g of sodium chloride. (a) Calculate the solubility of sodium chloride at 55oC. (b) If the solubility of sodium chloride at 30oC is 30g per 100g of water. Calculate the mass of
- 94. mass of sodium chloride crystals formed when the saturated solution at 55oC is cooled to 30oC. Possible Answers (a) 200g of H2O dissolve 150g of NaCl 100g of H2O dissolve 150 × 100 200 g of NaCl = 75g of NaCl
- 95. (b) 100g of H2O dissolve 30g of NaCl 200g of H2O dissolve 30 × 200 100 g of NaCl = 60g of NaCl Mass of crystals formed = 150 – 60 = 90g
- 96. Solubility curves Solubility curve is a graph that shows how the solubility of a salt varies with temperature. It is used to determine • solubility of a salt at various temperature, • temperature at which a certain mass of salt dissolved in water,
- 97. • the mass of salt obtained by cooling the solution from higher (T2) to lower(T1) temperature i.e. Mass of crystals = solubility at T2 – solubility at T1 Remember: The marking points for a graph are: Axes, scales, shape and sharpness.
- 98. Example (UNEB 2006/P2/14) (b) The solubilities of potassium chloride and potassium nitrate at certain temperatures are shown in the table below. Temperature(oC) 0 11 15 30 40 50 57 Solubility of KCl/ 100g of water 27.9 31.0 32.0 36.6 40.0 43.0 45.0 Solubility of KNO3 / 100g of water 14 21.5 25 43 63 84 102
- 99. (i) Plot on the same axes, a graph of solubility against temperature for the solubilities of potassium chloride and potassium nitrate. (ii) State which one of the two salts has a solubility which increases less rapidly with increase in temperature.
- 100. (iii) Determine the temperature at which the solubilities of the two salts are equal. (c) A saturated solution of potassium nitrate at 30oC was cooled to 5oC. Calculate the number of moles of potassium nitrate crystal formed.
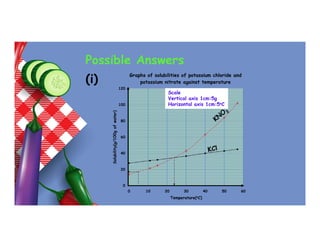
- 101. Possible Answers (i) 0 20 40 60 80 100 120 0 10 20 30 40 50 60 Solubility(g/100g of water) Temperature(oC) Graphs of solubilities of potassium chloride and potassium nitrate against temperature Scale Vertical axis 1cm:5g Horizontal axis 1cm:5oC
- 102. (ii) Potassium chloride (iii) 24oC (intersection) (c) Solubility at 30oC = 43 Solubility at 5oC = 16 Mass of KNO3 crystals formed = 43 – 16 = 27 RFM of KNO3 = 39 + 14 + 48 = 101 Moles of KNO3 formed = 27 101 = 0.267
- 103. Knowledge Check Qn.1(UNEB 1999/P1/39) An acid X2SO4 ionises as X2SO4(aq) 2X+(aq) + SO4 2-(aq) The basicity of the acid is A. 1 B. 2 C. 3 D. 4
- 104. Qn.2(UNEB 1999/P1/4) The solubility of copper(II) sulphate at 30oC is 25g per 100g of water. The mass of copper(II) sulphate that would crystallise if a solution containing 50g of copper(II) sulphate in 100g of water at 60oC is cooled to 30oC is A. 12.5g B. 25.0g C. 50.0g D. 75.0g
- 105. Qn.3(UNEB 2009/P1/32) Which one of the following is true about bases? A. are soluble in water. B. are hydroxides. C. neutralise acids. D. are oxides.
- 106. Qn.4(UNEB 1994/P1/14) Which one of the following acids can react with a base to produce an acid salt? A. Nitric acid. B. Ethanoic acid. C. Sulphuric acid. D. Hydrochloric acid.
- 107. Qn.5(UNEB 2005/P1/28) Which one of the following is an acid salt? A. CaSO4 B. NH4Cl C. KNO3 D. NaHCO3
- 108. Qn.6(UNEB 2005/P1/14) Which one of the following nitrates will produce nitrogen dioxide when strongly heated? A. Potassium nitrate. B. Sodium nitrate. C. Zinc nitrate. D. Ammonium nitrate.
- 109. Qn.7(UNEB 2006/P1/12) When heated strongly, lead (II) nitrate leaves a solid residue whose colour is A. reddish brown (hot), grey (cold). B. yellow (hot), white cold. C. reddish brown (hot), yellow (cold) D. reddish brown (hot), white (cold)
- 110. Qn.8(UNEB 2006/P1/36) The substance which does not produce carbon dioxide when heated strongly is A. Calcium carbonate B. Sodium carbonate C. Potassium hydrogencarbonate D. sodium hydrogencarbonate
- 111. Qn.9(UNEB 1987/P1/15) 10g of a saturated sodium chloride solution was evaporated and 6g of solid sodium chloride was left. The solubility of sodium chloride is A. 6 100 10 B. 6 100 4 C. 6 100 16 D. 10 100 16
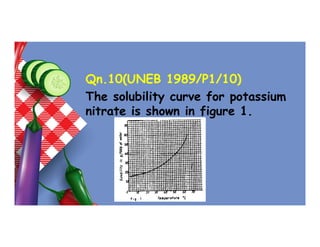
- 112. Qn.10(UNEB 1989/P1/10) The solubility curve for potassium nitrate is shown in figure 1.
- 113. The mass of potassium nitrate which would dissolve in 25g of water at 30oC is A. 0.6g B. 1.2g C. 6.0g D. 12.0g
- 114. Qn.11(UNEB 2002/P2/2) (a) Define the terms: (i) a normal salt (ii) an acid salt (b) Give one example of (i) a normal salt (ii) an acid salt
- 115. Qn.12(UNEB 1995/P2/13(a)) Sulphuric acid is a strong dibasic acid. (i) Explain the terms strong acid and basicity. (ii) Write an equation to show how sulphuric acid ionizes in water.
- 116. Qn.13(UNEB 1991/P2/12) (a) Describe how you would prepare pure crystals of lead(II) nitrate in the laboratory starting from lead(II) oxide. Write an equation for the reaction that takes place. (b)State what happens when lead(II) nitrate is strongly heated.
- 117. (c) State what is observed if ammonia solution in gradually added to a solution of lead(II) nitrate until the alkali is in excess. Write an equation for the reaction that takes place.
- 118. Qn.14(UNEB 2004/P2/3) A mixture containing copper(II) sulphate and copper(II) carbonate was shaken with excess water and filtered. (a) Identify the residue. (b) The dry residue was heated strongly. (i) State what was observed. (ii) Write an equation for the reaction.
- 119. (c) (i) Name a reagent that can be used to identify the anion in the filtrate. (ii) Write an ionic equation for the anion and the reagent you have named in (c)(i).
- 120. Qn.15(UNEB 1994/P2/11) (a) Copper(II) carbonate was heated strongly until there was no further change. (i) State what was observed. (ii) Write an equation for the reaction. (iii) Name one reagent which can be used to identify the gaseous product.
- 121. (b) Excess dilute sulphuric acid was added to the residue in (a) and the mixture warmed. (i) State what was observed. (ii) Write an equation for the reaction.
- 122. (c) To the product in (b) was added dilute sodium hydroxide solution dropwise until in excess. (i) State what was observed. (ii) Write a equation for the reaction.
- 123. Qn.16(UNEB 1991/P2/9) Copper(II) sulphate-5-water decomposes when heated. (a) State what would be observed when copper(II) sulphate-5-water is strongly heated. (b) Write an equation for the reaction. (c) Name one reagent that can be used to convert the residue back to copper(II) sulphate.
- 124. Qn.17(UNEB 2018/P2/9) (a) When a sample of copper(II) nitrate was strongly heated, a reddish brown gas was evolved. (i) Identify the gas. (ii) Write the formula of the residue.
- 125. (b) A sample of copper(II) nitrate contaminated with zinc nitrate was dissolved in water and the solution was treated with excess sodium hydroxide solution and then filtered. Identify the cation in the (i) Filtrate (ii) Residue
- 126. (c) The residue from (b) was strongly heated. (i) State what was observed. (ii) Write equation for the reaction that took place.
- 127. Qn.18(UNEB 2014/P2/7) (a) When a nitrate of a metal Y was heated strongly, brown fumes were observed together with a solid residue which was reddish brown when hot and yellow when cooled. (i) Identify Y. (ii) Write equation for the reaction that took place.
- 128. (b) The residue from (a) was heated with dilute nitric acid. Write equation for the reaction that took place. (c) To the product in (b), dilute sodium hydroxide was added drop wise until there was not further change. State what was observed.
- 129. Qn.19(UNEB 1997/P2/12) (a) Describe briefly how copper (II) sulphate crystals can be prepared from copper(II) oxide. (b) What would be observed if (i) sodium hydroxide solution was gradually added to a solution of copper(II) sulphate until the alkali was in excess?
- 130. Write the equation for the reaction that took place. (ii) hydrated crystals of copper sulphate were heated strongly?
- 131. Qn.20(UNEB 1991/P2/2) 2.5g of zinc carbonate was heated strongly until there was no further change. (a) State what was observed. (b) Write an equation for the reaction.
- 132. Qn.21(UNEB 1994/P2/10) The table below shows the solubilities of a salt P in water at different temperatures. Temperature (oC) 10 20 30 40 50 60 Solubility (g/100g of water) 18 20 24 30 38 50
- 133. (a) Plot a graph of solubility of P against temperature. (b) Use your graph to determine the solubility of P at 25oC. (c) Calculate the mass of P that would dissolve in 45g of water at 25oC.
- 134. Qn.22(UNEB 1995/P2/12) (a) Explain what is meant by the term saturated solution. (b) Describe how the solubility of potassium chloride can be determined in the laboratory.
- 135. (c) The table below shows the solubilities of potassium chloride and potassium nitrate at various temperatures. Temperature (oC) 0 20 40 60 Solubility of potassium chloride(g) 28.2 33.5 38.8 44.7 Solubility of potassium chloride(g) 12.9 31.8 61.2 108.2
- 136. (i) On the same axes, plot graphs of solubilities of potassium chloride and potassium against temperature. (ii) Determine the temperature at which the concentrations of the two salts are equal.
- 137. (iii)Which of the two salts dissolves more rapidly with increase in temperature? (iv)State what would happen if a saturated solution of potassium chloride at 40oC was cooled to 30oC.
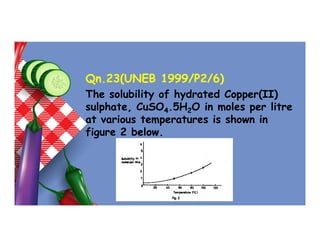
- 138. Qn.23(UNEB 1999/P2/6) The solubility of hydrated Copper(II) sulphate, CuSO4.5H2O in moles per litre at various temperatures is shown in figure 2 below.
- 139. (a) Determine the solubility of hydrated copper(II) sulphate at 80oC. (b) Calculate the solubility of hydrated copper(II) sulphate in g/100g of water at 80oC. (H = 1, 0 = 16, S = 32, Cu = 64)