Graphene
- 2. Content 2 Synthesis Advantages and disadvantages Companies in the Graphene Space Applications Conclusion 1) Introduction 2)Properties of Graphene 3) 4) 5) 6) 7) 8) Reference
- 3. INTRODUCTION • Graphene is a one-atom-thick planar sheet of carbon atoms. • Graphite itself consists of many graphene sheets stacked together. • It is an allotrope of carbon that are packed in a honeycomb crystal lattice. • Graphene is thin, transparent and flexible substance. • It is the strongest material ever discovered. 3
- 4. PROPERTIES OF GRAPHENE 4 C-C bond length is about 0.142nm. High tensile strength about 130GPa. It is 200 times stronger than steel. Graphene absorbs 2.3% of white light; i.e. it is almost transparent. It is Very light material i.e. 0.77mg/sq. m which is 1000 times lighter than same size of paper. It is Hexagonal structural element of carbon allotropes. Graphene is the highly conductive material than silver and copper.
- 5. GRAPHEN : SYNTHESIS Two basic technique used are : 1. MECHANICAL EXFOLIATION (ME) 2. CHEMICAL VAPOUR DEPOSITON(CVD) Graphene layer formed using Scotch tape(ME) 5
- 6. 1. MECHANICAL EXFOLIATION (ME): Mechanical exfoliation separates different layers of graphene from a graphite. Adhesive tape is pressed onto the graphite and peeled away when some layers stick to the surface.This tape is pressed onto the surface of the target substrate. The tape is peeled off when the layer stick to the target surface. 6
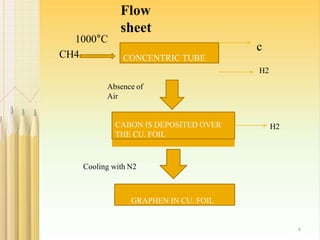
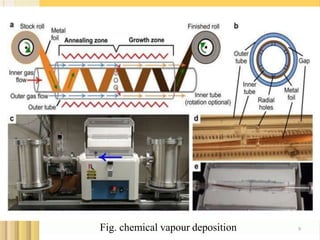
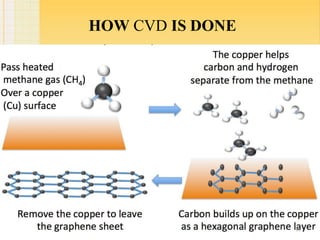
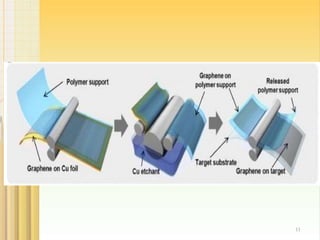
- 7. 2.CHEMICAL VAPOUR DEPOSITION : 7 Chemical vapour deposition is a well known process in which a substrate is exposed to gaseous compounds. These compounds decompose on the surface in order to grow a thinfilm . Graphene can be grown by exposing the copper foil to a gas i.e.CH4 at about 1000°C.The methane decomposes on the surface, so that the hydrogen evaporates. The carbon diffuses into cu foil. After cooling down in an atmosphere, a graphene layer grows on the surface. These graphene layers can be transferred via polymer support, which will be attached onto the top of the graphene. After etching the copper, the graphene can be stamped onto the required substrate and the polymer support gets peeled off.
- 8. 1000°C CH4 CONCENTRIC TUBE c H2 CABON IS DEPOSITED OVER THE CU. FOIL Absence of Air Cooling with N2 GRAPHEN IN CU. FOIL H2 8 Flow sheet
- 9. 9Fig. chemical vapour deposition
- 10. HOW CVD IS DONE 10
- 11. 11
- 13. ADVANTAGES 13 High heat conductivity, greater than copper and silver. No heat loss or it gives 100% output. It is transparent and lighter material. Stronger than diamond & steel. Can make very light weight parts for auto bodies. Used to make batteries that recharge faster as compare to lithium- ion battery.
- 14. DISADVANTAGES 14 Single sheet of graphene is hard to produce. Research has proven that graphene exhibits some toxic.
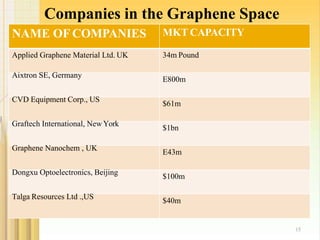
- 15. Companies in the Graphene Space NAME OF COMPANIES 15 MKT CAPACITY 34m Pound E800m $61m $1bn E43m $100m Applied Graphene Material Ltd. UK Aixtron SE, Germany CVD Equipment Corp., US Graftech International, New York Graphene Nanochem , UK Dongxu Optoelectronics, Beijing Talga Resources Ltd .,US $40m
- 16. • Solar panels • Water purifiction • Graphene battery • Super Capacitors • Automobile sector • Composites material for cars, airplanes, space shuttles, satellite. 16
- 17. 17
- 18. CONCLUSION 18 1. The quality and availability of “Synthetic graphene” will continue to improve. 2. Science must have ample access to improve graphene material & integrate it to the present day gadgets.
- 19. T 19 H A N K Y O U