Paper battery
- 1. Paper-based batteries: A review Thu H Nguyen, Arwa Fraiwan, Seokheun Choi n Bioelectronics & Microsystems Laboratory, Department of Electrical & Computer Engineering, State University of New York at Binghamton, 4400 Vestal Pkwy, Binghamton, NY 13902, USA a r t i c l e i n f o Article history: Received 31 August 2013 Received in revised form 17 October 2013 Accepted 1 November 2013 Available online 25 November 2013 Keywords: Paper battery Paper-like battery Electrochemical battery Biofuel cell Lithium-ion battery Supercapacitor Nanogenerator a b s t r a c t There is an extensively growing interest in using paper or paper-like substrates for batteries and other energy storage devices. Due to their intrinsic characteristics, paper (or paper-like) batteries show outstanding performance while retaining low cost, multifunctionality, versatility, flexibility and dispo- sability. In this overview, we review recent achievements in paper (or paper-like) batteries as well as their applications. Various types of paper power devices are discussed including electrochemical batteries, biofuel cells, lithium-ion batteries, supercapacitors, and nanogenerators. Further scientific and technological challenges in this field are also discussed. & 2013 Elsevier B.V. All rights reserved. Contents 1. Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 640 2. Paper-based batteries and energy storage devices. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 641 2.1. Electrochemical battery. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 641 2.2. Biofuel cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 644 2.2.1. Microbial fuel cells (MFCs) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 645 2.2.2. Enzymatic biofuel cells (EBFCs). . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 645 2.3. Lithium-ion batteries. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 646 2.4. Supercapacitors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 647 2.5. Nanogenerator . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 648 3. Summary and prospects . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 648 References . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 649 1. Introduction In recent years, paper has been used as a substrate for electronics instead of conventional rigid materials such as glass and silicon (Metters et al., 2013; Maxwell et al., 2013; Rolland and Mourey, 2013; Lo et al., 2013). The motivation is very clear: (i) Paper is extremely cheap and ubiquitously available; (ii) paper is combustible, so the paper-related devices can be economically disposed of by an incinerator; (iii) paper is thin, lightweight and flexible; (iv) paper is biocompatible and biodegradable and (v) paper provides high surface area for reagents to be stored (Steckl, 2013; Zhang et al., (2012); Yetisen et al., 2013; Martinez et al., 2010). Moreover, paper is attractive because it has the ability to wick fluids via capillary action. Therefore, microfluidic paper devices have a distinct advantage in that no external pumps and tubings are required to move liquid through the patterned fluidic pathways within the paper. Recently, advanced nanomater- ials and nanotechnologies have been incorporated into paper, forming “paper-like” flexible films with enhanced performance (Chen et al., 2013; Jabbour et al., 2010). The normal paper can be Contents lists available at ScienceDirect journal homepage: www.elsevier.com/locate/bios Biosensors and Bioelectronics 0956-5663/$ - see front matter & 2013 Elsevier B.V. All rights reserved. http://dx.doi.org/10.1016/j.bios.2013.11.007 n Corresponding author. Tel.: þ1 607 777 5913. E-mail address: sechoi@binghamton.edu (S. Choi). Biosensors and Bioelectronics 54 (2014) 640–649
- 2. applied as a skeleton for formation of paper-like nanocomposite films, which easily scale-up for commercial applications (Zhao and Shao, 2012). Several patterning processes on paper have also been accom- plished in a programmed manner, such as photolithography (Martinez et al., 2008), wax printing (Carrilho et al., 2009) and laser micromachining (Chitnis et al., 2011). The main objective of those patterning methods is to create hydrophobic barriers on paper that constitute the walls of capillary channels. One success- ful approach to patterning paper is based on photolithography (Martinez et al., 2008). An entire sheet of paper is impregnated with photoresist and selectively polymerized by exposing it to UV light through a transparent mask. The unexposed portion of the paper is then washed away. Another patterning method uses a commercial solid wax printer which rapidly deposits wax on paper (Carrilho et al., 2009). The paper is then heated to re-melt the wax which penetrates the paper to generate complete hydrophobic barriers. Another patterning method is based on laser treatment (Chitnis et al., 2011). Any paper with a hydrophobic surface coating, such as parchment paper, can be used for this purpose. The selective surface modification can be made by using a CO2 laser to create hydrophilic patterns on those hydrophobic papers. Equipped with a variety of techniques to pattern paper, paper electronics have also been successfully demonstrated by many groups. Whitesides et al. first demonstrated a novel biosensing system using patterned paper as a substrate, named microfluidic paper-based analytical devices (mPADs) (Martinez et al., 2007). This device was used for glucose and protein sensing in urine. Fortunato et al. fabricated flexible film field-effect transistors (FETs) using cellulose fiber paper-like materials as a dielectric layer (Fortunato et al., 2008), and (Steckl (2013) developed the electronic displays on polymer-coated paper called “e-paper”. A wide range of other electronic devices using paper substrates have been developed as well, including organic diodes (Zhang et al., 2012), MEMS sensors (Liu et al., 2011), RF antennas (Rida et al., 2007), circuit boards (Siegal et al., 2010) and capacitive touch pads (Mazzeo et al., 2012). Along with the advancement in paper electronics, paper or paper-like batteries and energy storage devices have attracted more and more interest because (i) a power source directly integrated onto paper would be preferable for easy system integration with paper electronics (Lee, 2006), and (ii) the intrinsic rough and porous surface of paper is beneficial for manipulation of electrons and ion transport across the entire structure, especially inside the electrode, for achieving high-power performance (Hu and Cui, 2012). To date, several types of paper (or paper-like) batteries and energy storage devices have been developed for various applications, such as a fluidic battery in paper-based microfluidic devices for the on-chip fluorescence assay (Thom et al., 2012), a urine-activated paper battery for biosystems (Lee, 2005), a supercapacitor integrated into photoelectrochemical lab-on-paper device (Ge et al., 2013), a paper-based microbial fuel cell for disposable diagnostic devices (Fraiwan et al., 2013a, 2013b, 2013c) and a lithium-ion paper-like battery with a high energy density (Leijonmarc,k et al., 2013). As paper (or paper-like) batteries are on the verge of entering the commercial realm, the scarcity of review articles regarding this topic creates an opportune time to summarize and examine this broad field. This review will cover the full scope of paper-based and paper-like-batteries and energy storage devices. We hope that this review will be helpful to readers who are interested in initiating work in this area as well as to researchers already working in this field who wish to learn of the progress achieved to date. 2. Paper-based batteries and energy storage devices According to the basic operating principles, we categorized the paper-based and paper-like batteries and energy storage devices as the following: (i) electrochemical batteries, (ii) biofuel cells, (iii) lithium-ion batteries, (iv) supercapacitors, and (v) nanogenerators. Table 1 summarizes their powers, electrode materials and poten- tial applications. For high-power paper electronics applications, lithium-ion batteries or supercapacitors are good power sources while mechanical nanogenerators are attractive for wearable electronics, such as with sport clothing and military uniforms. Biofuel cells or electrochemical batteries might be good candidates for paper-based mPADs or other types of small-power electronics (e.g. biosensors) which require only a couple minutes of power. In the following sections, we will discuss each type of battery in detail. 2.1. Electrochemical battery An electrochemical battery derives electrical energy from spontaneous redox reactions, and generally consists of two metals connected by a salt bridge or an ion exchange membrane. In the electrochemical batteries, species from one half-chamber lose electrons to their electrode while species from the other half- chamber obtain electrons from their electrode. The salt bridge or an ion exchange membrane is employed to provide ionic contact between two half-chambers with different electrolytes, preventing the solutions from mixing and causing unwanted side reactions. Electrochemical batteries can be fabricated on paper substrates (i) by depositing electrodes on the paper and/or (ii) by introducing electrolytes into a whole paper or hydrophilic regions patterned within the paper. One method is to use paper as an electrode. Hilder et al. (2009) reported a flexible paper-based zinc-air battery generating an Table 1 Summary of the paper-based batteries and energy storage devices. Electrochemical battery Biofuel cell Lithium-ion battery Supercapacitor Nanogenerator Operating principle Redox reaction Bio-redox reaction Liþ ion reaction Redox reaction Conversion of mechanical energy Application of paper Reservoir, electrode and/or supporter Reservoir, ion exchange membrane, electrode and/or supporter electrode and/or supporter electrode and/or supporter electrode and/or supporter Power generation μW–mW μW mW mW μW Electrode Metallic catalysts Biocatalysts (e.g. bacteria) Lithium based metal oxide materials Metal oxides or other carbon- based materials Piezoelectric or metallic materials Potential application Low power biosensors Low power biosensors High power electronics High power electronics Wearable electronics Remarks Common standard battery, rechargeable Self-sustainable, clean energy Rechargeable Rechargeable Energy harvesting from vibrations T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649 641
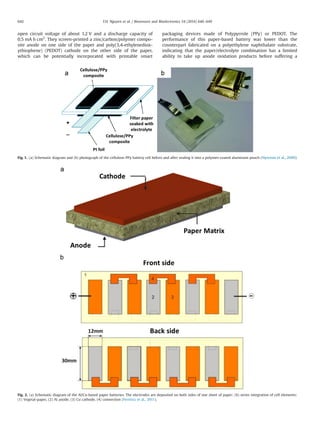
- 3. open circuit voltage of about 1.2 V and a discharge capacity of 0.5 mA h cm2 . They screen-printed a zinc/carbon/polymer compo- site anode on one side of the paper and poly(3,4-ethylenediox- ythiophene) (PEDOT) cathode on the other side of the paper, which can be potentially incorporated with printable smart packaging devices made of Polypyrrole (PPy) or PEDOT. The performance of this paper-based battery was lower than the counterpart fabricated on a polyethylene naphthalate substrate, indicating that the paper/electrolyte combination has a limited ability to take up anode oxidation products before suffering a Fig. 1. (a) Schematic diagram and (b) photograph of the cellulose-PPy battery cell before and after sealing it into a polymer-coated aluminum pouch (Nystrom et al., 2009). Fig. 2. (a) Schematic diagram of the Al/Cu-based paper batteries. The electrodes are deposited on both sides of one sheet of paper; (b) series integration of cell elements: (1) Vegetal-paper, (2) Al anode, (3) Cu cathode, (4) connection (Ferreira et al., 2011). T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649642
- 4. reduction in ionic mobility. One obvious method to improve the performance is to evenly introduce the PPy through the cellulose fibers (Nystrom et al., 2009). The cellulose-PPy conductive paper- like battery was able to be charged with currents as high as 600 mA cmÀ2 with only 6% loss in capacity over 100 subsequent charge and discharge cycles (Fig. 1). In this study, they developed highly porous cellulose substrates with several homogenous nanometer-thick layers of PPy to obtain a high-surface area exhibiting an exceptionally high ion-exchange capacity. Ferreira et al. (2010, 2011) proposed Au/Cu-based paper batteries that could be interconnected in series and recharged using water (Fig. 2). The series of integrated batteries were able to supply a voltage of about 3 V and a current ranging from 0.7 mA to 25 mA and to successfully control the ON/OFF gate state of paper transistors (Ferreira et al., 2011). The electrodes were deposited on a commercially available paper by thermal evaporation for 150–200 nm-thick layers of Cu (cathode) and Al (anode). At the same time, the paper was also used to contain the water as an electrolyte. Since the mobility of ions increases in less dense and thin papers, the current density of Al/vegetal-paper/Cu structures was about two times higher than that of Al/copy-paper/Cu structures. As has been demonstrated by several groups above, the entire region on the paper or paper-like material (combined with nanocomposites) can be used as a substrate for an anodic/cathodic electrode to be deposited on or as a membrane for the electrolyte. In this case, it is difficult for these batteries to be co-fabricated directly with other paper electronics because of the inability of creating patterns on the papers. On the other hand, the techniques of patterning microfluidic channels in paper has enabled direct incorporation of paper-based batteries into multilayer paper-based electronics for easy system integrations. Phillips et al. developed a more practical galvanic cell as low-cost power sources especially in resource limited settings (Thom et al., 2012, 2013). The Ag–Al galvanic cells were integrated directly into the microfluidic channels, which provided a direct link between a power source and an analytical function within the device (Fig. 3). The battery powered a UV LED within the paper- based device that enabled direct fluorescent measurements of the assay region (Thom et al., 2012). They also showed that multiple cells of these batteries could be connected in series and/or in parallel in a predictable way to obtain desired values of power (Thom et al., 2013) (Fig. 4). However, their operating lifetime lasted only for up to 15 min, which limited their application to single-use diagnostic tests. Moreover, the way to improve the stability of the battery and recycle the noble and heavy metals employed still remain serious challenges (Zhang et al., 2013a). Fig. 3. Galvanic cells incorporated into the microfluidic channels of the paper-based device. The cells can be connected in a series (a) or parallel (b) within the device (Thom et al., 2012). Fig. 4. Schematic diagram of a four-cell battery in which two galvanic cells are connected in parallel, and two are connected in a series. (a) Bottom view and (b) top view (Thom et al., 2013). T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649 643
- 5. To address these challenges, Wang and co-workers proposed a stable, environmentally-friendly and noble metal-free primary battery (C/FeCl3/NaCl/AlCl3/Al) (Zhang et al., 2013a) integrated with 3D origami microfluidic electrochemiluminescence (ECL) biosensing device. Fe3 þ is much more stable than Agþ , thus their design could be stocked in general conditions, and the cost would be reduced. Their battery consisted of the anodic and cathodic cells, carbon electrode, the Al anode and the salt bridge, which was activated by adding 50 μL water to the salt bridge. By assembling the power source and biosensor together in a 3D paper chip, they could develop a self-powered glucose ECL biosensing system. However, their relatively low operating voltage and current were another challenge. Unlike conventional acid-, and water-activated electrochemical batteries, another type of chemical battery, a urine-powered battery was developed (Lee, 2005; Liu and Crooks, 2012). The battery, developed by a research team in 2005, consisted of a copper (Cu) layer, copper chloride (CuCl) in the filter paper and a magnesium (Mg) layer (Lee, 2005). The whole assembly was sandwiched between two plastic layers and later laminated into a urine-activated paper battery by passing it through heated rollers at 120 1C. When a droplet of human urine was added to the battery, the urine soaked through the paper between the Mg and Cu layers, and the chemicals reacted to produce electricity. Using 0.2 mL of urine, the battery generated a voltage of around 1.5 V with a corresponding maximum power of 1.5 mW, the same as a standard AA battery, and ran for about 90 min. Other bodily fluids, such as tears, blood and semen, work just as well to activate the battery (Lee, 2006). However, their performance and cost completely depend on the electrode materials or architecture since the use of bodily fluids is limited to electrolyte. Moreover, further research is needed to integrate this battery into other paper-based devices. Another type of urine-based paper battery was developed to power both the sensing reactions and the color conversion of the electrochromic spot used for detection of glucose and H2O2 in the urine sample (Liu and Crooks, 2012) (Fig. 5). In this research, they put a power supply and a biosensor together in a single paper substrate. Two reservoirs for an urine sample were patterned on a chromatographic paper; one for the battery and the other for the sensing part. The average open circuit voltage (OCV) of the battery was about 0.94 V, and the short circuit current density was 60 μA mmÀ2 . However, since the paper was limited to storing the urine sample and the device was fabricated on Indium tin oxide (ITO) substrate, this system was not flexible nor economically disposable by incineration. In summary, electrochemical paper or paper-like batteries are well-suited for powering paper electronics since they produce enough power ranging from μW to mW and their operating principle is simple. Moreover, microfluidic devices fabricated out of paper have recently gained intense interest as promising plat- forms for conducting multiple functions simultaneously in a single paper. Particularly, the urine-activated paper battery will be an excellent power source for paper-based disposable healthcare tools for diseases such as diabetes if this battery can be fully fabricated out of paper, as it would be readily integrated into that paper-based biosensor system and ingeniously uses the fluid being tested as the power source for the device doing the testing. One concern with using electrochemical batteries is that they may not be environmentally friendly nor economically disposable because the electrode or electrolyte to be used is unstable, explosive, flammable and/or environmentally hazardous. Table 2 illustrates the list of paper-based electrochemical batteries published hence and their characteristics/performances. 2.2. Biofuel cells A biofuel cell is a bioelectrochemical device which converts biochemical energy into electrical energy by utilizing biocatalysts (Davis and Higson, 2007). The energy conversion is achieved by coupling an oxidation reaction supplying electrons at the anode with a reduction reaction utilizing electrons at the cathode. Instead of the expensive metal catalysts of electrochemical bat- teries, biofuel cells use renewable enzymes or microorganisms as the catalysts, offering a clean energy alternative to batteries and conventional fuel cells in that they can use renewable fuels such as sugar, ethanol, pyruvate and lactate to produce bioelectricity. Furthermore, biofuel cells operate under mild pH and temperature conditions compared to conventional fuel cells. Fig. 5. Schematic diagram of the device. The device consists of two major parts: a sensor and a urine-based battery, which are separated by a wax barrier. The device qualitatively detects 0.1 mM glucose and H2O2 in artificial urine samples (Liu and Crooks, 2012). Table 2 Summary of the paper-based electrochemical batteries. Hilder et al. (2009) Nystrom et al. (2009) Ferreira et al. (2010, 2011) Thom et al. (2012, 2013) Zhang et al. (2013a, 2013b) Lee (2005) Liu and Crooks (2012) Type Zinc-air Redox-active PPy Al-Cu Galvanic cells Galvanic cells Mg–CuCl ITO/metal/C Electrolyte LiCl/LiOH NaCl H2O AlCl3/AgNO3 H2O Urine Urine Type of paper/its application Photo paper /electrode supporter Filter paper/ reservoir for electrolyte Vegetal- & copy-paper/ electrode supporter & reservoir for electrolyte Filter paper/ electrodes & reservoir for electrolyte Filter paper/ electrodes & reservoir for electrolyte Filter paper /reservoir for electrolyte Filter paper /reservoir for electrolyte Anode material Zinc/carbon/ polymer Pt foil Al Al Al Mg ITO/metal/C Cathode material PEDOT Pt foil Cu Ag C Cu ITO/metal/C Device size 1 cm  2 cm 1 cm  0.5 cm 1.2 cm  3 cm $1 cm  2 cm $1 cm  1 cm 6 cm  3 cm $2 cm  1 cm Performance 1.2 V (OCV) and 0.5 mAhcm2 (discharge capacity) 6% decrease in the charge capacity when using 320 mA 3 V and 25 μA 1.3 V and 0.66 mA 1.53 V (OCV) and 4.4 mA/cm2 (short circuit current) 1.5 mW for about 90 min. 1.1 V and $30 μA/mm2 for Zn metal T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649644
- 6. The two major types of biofuel cells are microbial fuel cells and enzymatic biofuel cells (Davis and Higson, 2007; Shukla et al., 2004). Microbial fuel cells (MFCs) use microorganisms as a biocatalyst to generate electricity while enzymatic biofuel cells (EBFCs) employ enzymes to catalyze the redox reaction of the fuels (Minteer et al., 2007). MFCs typically have long lifetimes and are capable of completely oxidizing organic matter to carbon dioxide. However, they are limited by low power densities due to slow electron transport across microbial cell membranes. On the other hand, EBFCs produce orders of magnitude higher power densities and can avoid the need for a membrane separator. However, EBFCs can only partially oxidize the fuel and have limited lifetimes due to the fragile nature of the enzyme. 2.2.1. Microbial fuel cells (MFCs) MFCs are typically comprised of anodic and cathodic chambers separated by a proton exchange membrane (PEM) so that only Hþ or other cations can pass from the anode to the cathode (Yang et al., 2010). A conductive load connects the two electrodes to complete the external circuit. Microorganisms oxidize organic matter in the anodic chamber, completing respiration by transferring electrons to the anode. During this process, chemical energy is captured throughout the electron transport chain. Nicotinamide adenine dinucleotide (NADþ) and nicotinamide adenine dinucleotide dehydrogenase (NADH) function as coenzymes for the reactions, repeatedly oxidizing and reducing to synthesize adenosine triphosphate (ATP), the biolo- gical energy unit (Logan and Regan, 2006). The first paper-based MFC was developed by our group (Fraiwan et al., 2013a, 2013b, 2013c) (Fig. 6). The MFC featured (i) a paper-based proton exchange membrane by infiltrating sodium polystyrene sulfo- nate, (ii) micro-fabricated paper chambers by patterning hydrophobic barriers of photoresist and (iii) paper reservoirs for holding the anolyte and catholyte for an extended period of time. The rapid electricity generation should be noted since conventional MFCs require long start-up times (typically several days to a week), which is attributed to the accumulation and acclimation of bacteria on the anode of MFCs (Qian et al., 2009). This is probably because a hydrophilic paper reservoir rapidly absorbs the anolyte and immediately promotes the attachment of a number of bacteria cells to the anode (Fraiwan et al., 2013a, 2013b, 2013c). Our recently developed battery stack provided desired values of current and potential to power a red LED for more than 30 min without power management interface circuits (not published yet). This new paper battery is expected to be a simple, low-cost, easy-to-use power source for single-use paper-based diag- nostic devices in resource-limited settings since sewage or soiled water in a puddle can become an excellent source for extracting bioelectricity through bacterial metabolism. However, we used potas- sium ferricyanide as a catholyte, which is not suitable for actual application because it is toxic and expensive. The usage of ferricyanide was only for investigating the feasibility of developing a paper-based MFC. In the future, oxygen in ambient air can be used as an electron acceptor due to its low cost, sustainability and lack of waste products. In our research, a filter paper was micropatterned for reservoirs, fluidic channels and proton exchange membranes, which can be readily integrated with other paper electronics. 2.2.2. Enzymatic biofuel cells (EBFCs) EBFCs use redox enzymes, such as glucose oxidase, with laccase as the catalysts that facilitate the electron generation between substrates and electrode (Bullen et al., 2006). The use of enzymes allows cells to have defined reaction pathways on the anode and to overcome the limited output power generation of MFCs (Ivanov et al., 2010). Also, an enzymatic electrode can be combined with a conventional catalyst electrode to exhibit higher stability (Habrioux et al., 2009). Zhang et al. (2012) developed a novel paper-based glucose/air EBFC, which provides a potential applica- tion for a low-cost and portable power device (Fig. 7). Whatman filter paper was utilized as a substrate and hydrophilic regions were patterned within the paper. An ionic liquid functionalized carbon nanotubes (CNTs-IL) nanocomposite was used as support for stably confining the anodic biocatalyst for glucose electroox- idation and for facilitating direct electrochemistry of the cathodic biocatalyst for O2 electroreduction. The open-circuit voltage (OCV) of the BFC was 0.56 V, and the maximum power density was 13.5 μW cmÀ2 (at 0.33 V). Due to the small size of the paper-based fuel cell (1.5 cm  1.5 cm), the hydrophilic area of the paper could be filled only up to 30 μL of biofuel. However, further optimization on engineering challenges, such as target extended operational Fig. 6. (a) A photograph of the fully-assembled paper-based MFC and (b) schematic diagram of the device (Fraiwan et al., 2013a) . Fig. 7. Photographs of the fully-assembled paper-based EBFC (Zhang et al., 2012). T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649 645
- 7. stability, improved power output and a higher degree of miniatur- ization, will be required (Zhang et al., 2012). Ciniciato et al. (2012) developed bio-cathodes for application in paper-based EBFCs. They demonstrated the possibility of using different designs of air-breathing bio-cathodes and ink-based bio- cathodes. Three different air-breathing bio-cathodes were simply designed on the Toray paper with a pressed layer of teflonized carbon black and on the two different multi-walled carbon nanotube papers. For all three gas-diffusion cathodes, the enzyme was immobilized on the papers by adsorbing 15 mg bilirubin oxidase (Box) in phosphate buffer. For ink-based bio-cathodes, the biocatalyst was combined with a slurry of carbon black or alternatively with commercial graphite inks. The air-breathing bio-cathodes increased current densities up to 0.5 mA cmÀ2 while the ink-based ones enriched the variety of possible fuel cell designs. Under a load of 300 mV, a stable current density was obtained for 12 h continuous operation. However, there are still challenges for further development of long-term stability of the enzymatic bioelectrodes and efficient electron transfer between enzymes and electrode surfaces. Because of their low power generation and short-term stability, paper-based biofuel cells would be preferred for low power and/or single use applications, such as mPADs. However, biofuel cells have many advantages over other types of batteries as (i) biofuel cells are capable of generating electricity from various kinds of organic matters such as glucose, urine, biomass, wastewater, and even commercial beverages. Moreover, (ii) the cell structure is much simpler than others, (iii) the material/fabrication is cost effective and (iv) they are environmentally friendly, so they can be economically disposed of by an incinerator. Therefore, if the papers used in the biofuel cells can be more sophisticately patterned for microfluidic channels like Philip's electochemical paper batteries(Thom et al., 2012, 2013), these paper biobatteries will be the perfect power source for paper diagnostic devices. This is mainly because body fluids such as glucose can be used both for harvesting electricity and for diagnostic uses. Table 3 summarizes the paper-based biofuel cells. 2.3. Lithium-ion batteries Li-ion batteries operate by shuttling lithium ions between the anode and the cathode through an electronically insulating, ion- conductive electrolyte. The lithium-ion (Li-ion) battery becomes the most promising battery candidate for applications that espe- cially require high power and energy density. Li-ion batteries have replaced other types of batteries based on different chemistries, such as Ni–Cd and NiMH cells, especially in the field of advanced portable electronics. Recently, with substantial advancement of flexible electronics, Li-ion battery has also shown great promise as a flexible power source. The strong interest in this area has led to a number of research efforts on polymer (Koo et al., 2012) and textile-based substrates (Hu et al., 2010). Paper or paper-like Li-ion batteries have been explored as well because of (i) their intrinsic characteristics, such as large surface roughness and the porous structure for improving power generation, and (ii) the mechanical flexibility to fully realize flexible electronics. Normally, their power and current densities are significantly larger than other types of paper-based batteries preferred for powering high- power electronics. Flexible Li-ion batteries based on “paper-like” conductive sub- strates have been a major focus in this area. Recently, a single- walled carbon nanotube/polycellulose paper (SWCNT/PP) Li-ion battery was developed (Wang et al., 2012b). Polycellulose papers were dipped into the CNT ink and immersed for 10 min. Full cells using Li4Ti5O15 and LiFePO4 electrodes based on SWCNT/PPs showed a first discharge capacity of 153.3 mA h gÀ1 with Coulom- bic efficiencies of 90.6% at 0.1 C and discharge capacity of 102.6 mA h gÀ1 at high rate (10 C). Leijonmarc,k et al. (2013) developed a single-paper flexible lithium-ion battery cell. They used nano-fibrillated cellulose both as an electrode binder and as a separator material. The battery was made through a paper-making type process by sequential filtration of water dispersions contain- ing the battery components. The resulting battery cells were mechanically strong and flexible with good cycling properties with reversible capacities of 146 mA h gÀ1 LiFePO4 at C/10 and 101 mA h gÀ1 LiFePO4 at 1 C. Shao and co-workers presented a high-performance Li-ion battery based on paper-like hierarchical anatase TIO2 film electrode (Zhao and Shao, 2012). The filter papers were applied as skeletons for formation of paper-like TIO2 film since the specific surface of the papers is as low as 1.3 m2 gÀ1 . By applying such film in Li-ion batteries, good rate performance was demonstrated and the capacity was still about 100 mA h gÀ1 even at the high rate of 20 C. However, most research on paper Li-ion batteries are using “Paper-like” films incorporating various nanocomposites, which typically demand much more complicated processes with increased cost (Hu and Cui, 2012; Nyholm et al., 2011). To decrease cost and complexity, the Cui group used a com- monly available commercial paper with simple fabrication and structure on the paper (Hu et al., 2009). They reported a Li-ion battery by using a paper coated with carbon nanotube and silver nanowire film (Hu et al., 2009) (Fig. 8). Compared with plastics, paper substrates themselves dramatically improved film adhesion, greatly simplified the coating process and significantly lowered the cost. They showed that this conductive paper can be used as an excellent lightweight current collector in Li-ion batteries to replace the existing metallic chemical counterparts. This battery showed excellent cycling performance with 95% capacitance Table 3 Summary of the paper-based biofuel cells. Fraiwan et al. (2013a) Fraiwan et al. (2013b) Zhang et al. (2012) Ciniciato et al. (2012) Type MFCs MFCs EBFCs EBFCs Type of paper/its application Filter paper/reservoir for anolyte & catholyte and PEM Filter paper/reservoir for anolyte & catholyte Filter paper/reservoir for anolyte & catholyte Toroy paper/electrode supporter & reservoir for elecrolyte Anolyte/catholyte LB media/Ferricyanide LB media/Ferricyanide Glucose dehydrogenase/ bilirubin oxidase Enzyme/bilirubin oxidase Anode material Carbon clothes Multi-anode based on Carbon clothes Carbon ink printed onto the paper Pt mesh Proton exchange membrane (PEM) Chemical treated paper with Na-PSS Nafion 117 No PEM No PEM Cathode material Carbon clothes Carbon clothes Carbon ink printed onto the paper Ink-or air-based biocathode Device size 3 cm  4 cm 3 cm  4 cm 1.5 cm  1.5 cm 2 cm diameter Performance 74 μAW/cm2 and 5.5 μW/cm2 84 μAW/cm2 and 28.4 μW/cm2 13.5 μW/cm2 0.5 mA/cm2 T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649646
- 8. retention after 280 cycles and less than 0.01% of the capacity decay during the following 220 cycles. In the following year, they integrated all of the components of a lithium-ion battery into a single sheet of paper with a lamination process (Hu et al., 2010) (Fig. 9). The CNT thin films were laminated onto commercial paper, and the paper functioned as both the mechanical support and Li- ion battery membrane. Due to the intrinsic porous structure of the paper, it also functioned more effectively as a separator than commercially available separators. In summary, paper or paper-like Li-ion batteries can be an excellent power source in terms of power/current generation. Their power generation is a couple of orders of magnitude larger than those of paper electrochemical batteries or paper biofuel cells. Also, these batteries are rechargeable and durable for a long period of time. However, the Li-ion battery configuration requires many functional layers to be deposited on paper, increasing complexity of their designs and fabrication processes making them high cost. Moreover, the materials themselves are not inexpensive nor environmentally friendly and require specific disposal means. In addition, system integrations with other paper electronics necessitate an intense packaging or insulating techni- que not to be interfered from Li-based materials. Therefore, paper or paper-like Li-ion batteries are more preferably available as a flexible power source rather than as a paper power source for paper electronics. They will be more integrable into polymer- based flexible electronic applications by using roll-to-roll fabrica- tion methods. For additional reviews on paper-based energy storage devices, especially for Li-ion and supercapacitors which will be described in the next section, the reader is also directed to Hu and Cui, 2012 and Nyholm et al., 2011. 2.4. Supercapacitors Supercapacitors are energy storage devices based on electro- chemical processes which have attracted considerable attention in recent years (Snook et al., 2011; Naoi et al., 2013). Supercapacitors are typically superior to batteries in charge/discharge rate, power density, environmental impact and safety (Zhang et al., 2013b). Supercapacitors are similar to batteries in design and manufactur- ing, consisting of two electrodes, an electrolyte and a separator that electrically isolates the two electrodes. Pure physical charge accumulation can occur at the electrode/electrolyte interface (electrostatic supercapacitors) or charges can be directly stored during the charging and discharging processes (faradaic super- capactiros) (Wang et al., 2012a). The concept of paper-based supercapacitors has recently gained more interest mainly due to the possibilities of manufacturing flexible, lightweight, thin, and low cost devices (Nyholm et al., 2011; Hu and Cui, 2012; Zhang et al., 2013b). Like Li-ion batteries, “paper-like” supercapacitors are more common instead of using a commercially available paper as it is because the reinforcement of the nanocomposites with cellulose would result in a significant increase of the Young's modulus, reduced elongation at break, increased tensile strength and enhanced electron/ion transfer rates (Hu and Cui, 2012; Nyholm et al., 2011). For “paper-like” supercapacitors, highly conductive carbon- based nanomaterials/nanocomposites, such as carbon nanotubes (CNTs) and graphenes, are mainly used. Pushparaj et al. showed that all the basic components of the supercapacitor can be integrated into nanocomposite papers (Pushparaj et al., 2007). An aligned carbon nanotube (CNT) electrode and electrolyte were Fig. 9. Schematic diagram of (a) the lamination process and (b) the final paper Li-ion battery device structure (Hu et al., 2010). Fig. 8. (a) Schematic diagram of the paper-based Li-ion battery. (b) Lighting up a blue LED (Hu et al., 2009). (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.) T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649 647
- 9. embedded with nanoporous cellulose paper. This paper-based supercapacitor showed a good capacitive behavior generating a higher operating voltage (2.3 V) with the calculated specific capacitance, 22 F gÀ1 . A power density of 1.5 kW kgÀ1 was obtained at room temperature, which is within reported ranges of commercial supercapacitors and comparable to flexible devices reported (Pushparaj et al., 2007). Other CNT-based paper super- capacitors have also been developed with different fabrication processes. (Chou et al., 2008; Kang et al., 2012; Zhang et al., 2013b). MnO2 nanowires were electrodeposited onto the CNT paper by a cyclic voltammetric technique (Chou et al., 2008). This supercapacitor displayed specific capacitances as high as 167.5 F gÀ1 at a current density of 77 mA gÀ1 . After 3000 cycles, the composite paper retained more than 88% of initial capacitance. Kang et al. fabricated an all-solid-state paper supercapacitor using CNTs and ionic-liquid-based gel electrolytes showing excellent stability and flexibility (Kang et al., 2012). Zhang et al. (2013b) presented all-paper-based supercapacitors using microfibrillated cellulose and multi-walled CNTs. The specific capacitance was calculated to be 154.5 mF cmÀ2 at 20 mV sÀ1 from cyclic voltam- metry. Recently, a polyaniline-based (PANI) paper supercapacitor was introduced by Wang et al. (Yuan et al., 2012). They fabricated an all-solid-state PANI-based flexible supercapacitor on paper substrates as effective energy storage units for storing electric energy produced by a piezoelectric generator or a solar cell. This supercapacitor showed a stable areal capacitance of about 50 mF cmÀ2 with respect to the discharge current increasing from 0.1 to 2 mA cmÀ2 . Another supercapacitor based on free standing CNT/graphene and Mn3O4 nanoparticle graphene electrodes was presented, exhibiting excellent mechanical stability, greatly improved active surface areas, and enhanced ion transportation (Gao et al., 2012). The use of polymer gel electrolytes integrating with free-standing paper electrodes led to a flexible supercapacitor with high energy density and power density and excellent cycling stability. The supercapacitor generated an increased cell voltage of 1.8 V, capa- citance retention of 86% after 10,000 continuous charge/discharge cycles and an energy density of 32.7 W h kgÀ1 . As a good applica- tion of the supercapacitors, a photoelectrochemical lab-on-paper device was successfully operated with the paper-based super- capacitor for determination of adenosine triphosphate (ATP) in human serum samples (Ge et al., 2013). The paper-based super- capacitor was integrated into the device to collect and store the generated photocurrents. The stored electrical energy was released instantaneously to obtain an amplified ($13-fold) and detectable current as well as a higher sensitivity. In summary, most paper supercapacitors are based on paper- like composites of cellulose like Li-ion batteries. Although they are able to generate high current/power, their integration with other paper electronics needs further studies. 2.5. Nanogenerator Recently, energy harvesters that convert mechanical energy into electrical energy have attracted attention. Paper has emerged as their substrate materials for a wide range of flexible electronics applications since paper is thermally stable, foldable, lightweight, inexpensive and the most abundant material on earth. The first paper-based mechanical power generator was developed by Kim et al. (2011) (Fig. 10). They introduced ZnO nanorods as a piezo- electric active layer on a cellulose substrate using low-temperature aqueous solution methods. The output voltage and current density from this paper generator was 75 mV and 2 mA cmÀ2 , respectively. The current output was rarely changed, even after 10 mechanical bending cycles with a radius of curvature of 2.8 cm. Qiu et al. (2012) also reported a similar type of paper-based piezoelectric generator producing an output voltage of up to 10 mV and an output current of about 10 nA. Recently, Wang et al. developed a paper-based nanogenerator using an electrostatic effect (Zhong et al., 2013). The instantaneous output power density of the nanogenerator reached $90.6 mA cmÀ2 at a voltage of 110 V, and this illuminated 70 LEDs. The polytetrafluoroethylene (PTFE)-Ag- paper and Ag-paper were assembled to make the nanogenerator. These nanogenerators pave the way in harvesting energy based on mechanical actuation, and this study presents the significant potential applications of a paper-based, self-powered system as an energy source (Zhong et al., 2013). 3. Summary and prospects The current technologies in paper-based or paper-like batteries and energy storage devices have been summarized in this review. The successful coupling of these power devices with other paper- based electronics has been explored for the development of flexible, self-powered systems. There are several types of paper or paper-like power devices available, such as electrochemical batteries, biofuel cells, Li-ion batteries, superconductors and nanogenerators. The choice of battery type depends on the application. Small power paper-based biosensors or other phar- maceutical devices might require electrochemical batteries or biofuel cells as energy sources while other high-power electronics would prefer Lithium-ion batteries or supercapacitors. Nanogen- erators should be used to capture low-frequency mechanical energy, such as body movements, for practical applications. Since paper Li-ion batteries, supercapacitors, and nanogenerators are Fig. 10. Paper-based piezoelectric nanogenerator. (a) A SEM image of the ZnO nanorods grown on a cellulose paper and (b) schematic diagram of an integrated nanogenerator (Kim et al., 2011). T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649648
- 10. solid-state flexible power sources exploiting paper's porous, flex- ible and stretchable characteristics, the use of paper for these applications is limited to forming high-performance conductive paper-like substrates combining nanocomposites. Therefore, their applications are more preferable as a flexible power source integrated with non-paper flexible electronics. On the other hand, electrochemical batteries and biofuel cells are typically liquid-state power sources, and their working principles/device configura- tions/fabrications are simple and straightforward. Therefore, microfluidic channels patterned in the paper can be readily interfaced with other paper-based microfluidic devices especially for health care monitoring. However, challenges still exist both in performance and integration into a paper-based system. Further- more, almost none of the known methods are industrially avail- able to produce a fully functional paper-based system. In addition, most of the known methods to fabricate those power devices on paper substrates are either time-consuming or costly. Therefore, efficient fabrication and integration techniques are in urgent need of development in order to meet the practical use of these power devices for paper-based applications. References Bullen, R.A., Arnot, T.C., Lakeman, J.B., Walsh, F.C., 2006. Biosens. Bioelectron. 21, 2015–2045. Carrilho, E., Martinez, A.M., Whitesides, G.M., 2009. Anal. Chem. 81, 7091–7095. Chen, X., Zhu, H., Liu, C., Chen, Y., Weadock, N., Rubloff, G., Hu, L., 2013. J. Mater. Chem. A 1, 8201–8208. Chitnis, G., Ding, Z., Chang, C., Savran, C.A., Ziaie, B., 2011. Lab Chip 11, 1161–1165. Chou, S., Wang, J., Chew, S., Liu, H., Dou, S., 2008. Electrochem. Commun. 10, 1724–1727. Ciniciato, G.P.M.K., Lau, C., Aochrane, C., Sibbett, S.S., Gonzalez, E.R., Atanassov, P., 2012. Electrochim. Acta. 82, 208–213. Davis, F., Higson, S.P.J., 2007. Biosens. Bioelectron. 22, 1224–1235. Ferreira, I., Bras, B., Correia, N., Barquinha, P., Fortunato, E., Martins, R., 2010. J. Disp. Technol. 6, 332–335. Ferreira, I., Bras, B., Martins, J.I., Correia, N., Barquinha, P., Fortunato, E., Martins, R., 2011. Electrochem. Acta. 56, 1099–1105. Fortunato, E., Correria, N., Barquinha, P., Pereira, L., Goncalves, G., Marins, R., 2008. IEEE Electr. Device Lett. 29, 988–990. Fraiwan, A., Mukherjee, S., Sundermier, S., Lee, H.-S., Choi, S., 2013a. Biosens. Bioelectron. 49, 410–414. Fraiwan, A., Mukherjee, S., Sundermier, S., Choi, S., 2013b. In: IEEE International Conference on Micro-Electro-Mechanical-Systems (MEMS), Jan. 20–24, 2013, Taipei, Taiwan, pp. 809–812. Fraiwan A. and Choi S., Proc. IEEE Sensors, 2013c, Baltimore, Maryland, USA, pp. 1908–1911. Gao, H., Xiao, F., Ching, C.B., Duan, H., 2012. ACS Appl. Mater. Interfaces 4, 7020–7026. Ge, L., Wang, P., Ge, S., Li, N., Yu, J., Yan, M., Huang, J., 2013. Anal. Chem. 85, 3961–3970. Habrioux, A., Servat, K., Tingry, S., Kokoh, K.B., 2009. Electrochem. Commun. 11, 111–113. Hilder, M., Winther-Jensen, B., Clark, N.B., 2009. J. Power Sources. 194, 1135–1141. Hu, L., Choi, J.W., Yang, Y., Jeong, S., Mantia, F.L., Cui, L., Cui, Y., 2009. Proc. Nat. Acad. Sci. U.S.A. 106, 21490–21494. Hu, L., Wu, H., Mantia, F.L., Yang, Y., Cui, Y., 2010. ACS Nano 10, 5843–5849. Hu, L., Cui, Y., 2012. Energy Environ. Sci. 5, 6423–6435. Ivanov, I., Vidakovic-Koch, T., Sundmacher., K., 2010. Energy 3, 803–846. Jabbour, L., Gerbaldi, C., Chaussy, D., Zeno, E., Bodoardo, S., Beneventi, D., 2010. J. Mater. Chem. 20, 7344–7347. Kang, Y.J., Chung, H., Han, C., Kim, W., 2012. Nanotechnology 23, 065401. Kim, K., Lee, K.Y., Seo, J., Kumar, B., Kim, S., 2011. Small 18, 2577–2580. Koo, M., Park, K., Lee, S., Suh, M., Jeon, D., Choi, J., Kang, K., Lee, K., 2012. Nano Lett. 12, 4810–4816. Lee, K.B., 2005. J. Micromech. Microeng. 15, S210–S214. Lee, K.B., 2006. J. Micromech. Microeng. 16, 2312–2317. Leijonmarc,k, S., Cornell, A., Lindbergh, G., Wagberg, L., 2013. J. Mater. Chem. A 1, 4671–4677. Liu, X., Mwangi, M., Li, X., O'Brien, M., Whitesides, G.M., 2011. Lab Chip 11, 2189–2196. Liu, H., Crooks, R.M., 2012. Anal. Chem. 84, 2528–2532. Lo, S., Yang, S., Yao, D., Chen, J., Tu, W., Cheng, C., 2013. Lab Chip 13, 2686–2692. Logan, B.E., Regan, J.M., 2006. Environ. Sci. Technol. 40, 5172–5180. Martinez, A.W., Phillips, S.T., Butte, M.J., G.M., 2007. Angew. Chem. Int. Ed. 46, 1318–1320. Martinez, A.W., Phillips, S.T., Whitesides, G.M., 2008. Proc. Nat. Acad. Sci. U.S.A. 105, 19606–19611. Martinez, A.W., Phillips, S.T., Whitesides, G.M., 2010. Anal. Chem. 82, 3–10. Maxwell, E.J., Mazzeo, A.D., Whitesides, G.M., 2013. MRS Bull. 38, 309–314. Mazzeo, A.D., Kalb, W.B., Chan, L., Killian, M.G., Bloch, J.-F., Mazzeo, B.A., Whitesides, G.M., 2012. Adv. Mater. 24, 2850–2856. Metters, J.P., Houssein, S.M., Kampouris, D.K., Banks, C.E., 2013. Anal. Methods 5, 103–110. Minteer, S.D., Liaw, B.Y., Cooney, M.J., 2007. Curr. Opin. Biotechnol. 18, 1–7. Naoi, K., Ishimoto, S., Miyamoto, J., Naoi, W., 2013. Energy Environ. Sci. 5, 9363–9373. Nystrom, G., Razaq, A., Strømme, M., Nyholm, L., Mihranyan, A., 2009. Nano Lett. 9, 3635–3639. Nyholm, L., Nystrom, G., Mihranyan, A., Strømme, M., 2011. Adv. Mater. 23, 3751–3769. Pushparaj, V.L., Shaijumon, M.M., Kumar, A., Murugesan, S., Ci, L., Vajtai, R., Linhardt, R.J., Nalamasu, O., Ajayan, P.M., 2007. Proc. Nat. Acad. Sci. U.S.A. 104, 13574–13577. Qian, F., Baum, M., Gu, Q., Morse, D.E., 2009. Lab Chip 9, 3076–3081. Qiu, Y., Zhang, H., Hu, L., Yang, D., Wang, L., Wang, B., Ji, J., Liu, G., Liu, X., Lin, J., Li, F., Han, S., 2012. Nanoscale 4, 6568–6573. Rida, A., Yang, L., Tentzeris, M.M., 2007 In: Proceedings of the 2007 IEEE-APS Symposium Honolulu HI, 2749-2752. Rolland, J.P., Mourey, D.A., 2013. MRS Bull. 38, 299–305. Shukla, A.K., Suresh, P., Berchmans, S., Rajendran, A., 2004. Curr. Sci. 87, 455–468. Siegal, A.C., Phillips, S.T., Dickey, M.D., Lu, N., Suo, Z., Whitesides, G.M., 2010. Adv. Funct. Mater. 20, 28–35. Steckl, A.J., 2013. IEEE Spect. 50, 48–54. Snook, G.A., Kao, P., Best, A.S., 2011. J. Power Sources 196, 1–12. Thom, N.K., Yeung, K., Phillion, M.B., Phillips, S.T., 2012. Lab Chip 12, 1768–1770. Thom, N.K., Lewis, G.G., DiTucci, M.J., Phillips, S.T., 2013. RSC Adv. 3, 6888–6895. Wang, G., Zhang, L., Zhang, J., 2012a. Chem. Soc. Rev. 41, 797–828. Wang, J., Li, L., Wong, C.L., Madhavi, S., 2012b. Nanotechnology 23, 495401. Yang, Y., Sun, G., Xu, M., 2010. J. Chem. Technol. Biotechnol. 86, 625–632. Yuan, L., Xiao, X., Ding, T., Zhong, J., Zhang, X., Shen, Y., Hu, B., Huang, Y., Zhou, J., Wang, Z.L., 2012. Angew. Chem. Int. Ed. 51, 4934–4938. Yetisen, A.K, Akram, M.S., Lowe, C.R., 2013. Lab Chip 13, 2210–2251. Zhang, L., Zhou, M., Wen, D., Bai, L., Lou, B., Dong, S., 2012. Biosens. Bioelectron. 35, 155–159. Zhang, W., Zhang, X., Lu, C., Wang, Y., Deng, Y., 2012. J. Phys. Chem. C. 116, 9227–9234. Zhang, X., Li, J., Chen, C., Lou, B., Zhang, L., Wang, E., 2013a. Chem. Commun. 49, 3866–3868. Zhang, X., Lin, Z., Chen, B., Sharma, S., Wong, C., Zhang, W., Deng, Y., 2013b. J. Mater. Chem. A 1, 5835–5839. Zhao, B., Shao, Z., 2012. J. Phys. Chem. C. 116, 17440–17447. Zhong, Q., Zhong, J., Hu, B., Hu, Q., Zhou, J., Wang, Z.L., 2013. Energy Environ. Sci. 6, 1779–1784. T.H. Nguyen et al. / Biosensors and Bioelectronics 54 (2014) 640–649 649