Meciadanol
Appearance
| |
| Names | |
|---|---|
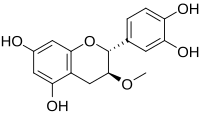
| IUPAC name
3-Methoxyflavan-3′,4′,5,7-tetrol
| |
| Systematic IUPAC name
(2R,3S)-2-(3,4-Dihydroxyphenyl)-3-methoxy-3,4-dihydro-2H-1-benzopyran-5,7-diol | |
| Other names
3-O-Methylcatechin
Meciadanolum 3',4',5,7-Tetrahydroxy-3-methoxyflavan (2R,3S)-2-(3,4-Dihydroxyphenyl)-3-methoxy-5,7-chromandiol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.059.719 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H16O6 | |
| Molar mass | 304.298 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Meciadanol is a synthetic O-methylated flavanol. It is the 3-O-methylation of catechin.
It inhibits histidine decarboxylase in rats.[1]
