Journal Description
Onco
Onco
is an international, peer-reviewed, open access journal on the whole field of oncotargets and cancer therapies research published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 19 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the first half of 2024).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Onco is a companion journal of Cancers.
Latest Articles
Clinical Evidence of Methods and Timing of Proper Follow-Up for Head and Neck Cancers
Onco 2024, 4(4), 275-286; https://doi.org/10.3390/onco4040020 - 29 Sep 2024
Abstract
Background: For patients with head and neck squamous cell carcinoma (HNSCC), after a single or multi-modality treatment, a specific follow-up strategy is needed, but there is no agreement between the main international societies on the proper methods and timing of follow-up. Methods: We
[...] Read more.
Background: For patients with head and neck squamous cell carcinoma (HNSCC), after a single or multi-modality treatment, a specific follow-up strategy is needed, but there is no agreement between the main international societies on the proper methods and timing of follow-up. Methods: We performed a descriptive review to evaluate the available data and compare the main guidelines, giving some practical guidance to perform effective personalized follow-up strategies. Results and Conclusions: While clinical and endoscopic follow-up alone seems to be appropriate for early-stage HNSCCs, the addition of close radiologic follow-up in locally advanced HNSCCs is still debated, as there are no data indicating that an earlier detection of recurrence correlates with increased survival, while it is mandatory in the first three-six months to define the response to treatment. For patients who have undergone conservative surgery or have major pathological risk factors, the incidence of locoregional recurrence is higher, and locoregional radiologic follow-up (magnetic resonance imaging is preferred to computed tomography) should be considered. Positron emission tomography may be useful in cases of suspected locoregional persistence of disease, differentiating it from post-irradiation outcomes. Distant radiological follow-up can be considered in the detection of the second primary in cases of specific risk factors and for virus-related tumors. For the latter, the use of circulating DNA should always be considered. A brain scan is not recommended without specific symptoms. For all patients who do not fall into the above categories, clinical and endoscopic follow-up should be proposed, reserving radiological investigations only at the onset of symptoms.
Full article
Open AccessReview
Exploring the Potential of Epiregulin and Amphiregulin as Prognostic, Predictive, and Therapeutic Targets in Colorectal Cancer
by
Cara Guernsey-Biddle, Peyton High and Kendra S. Carmon
Onco 2024, 4(4), 257-274; https://doi.org/10.3390/onco4040019 - 26 Sep 2024
Abstract
►▼
Show Figures
The epidermal growth factor receptor (EGFR) plays a critical role in regulating essential cellular processes that are frequently hijacked to promote cancer. In colorectal cancer (CRC) in particular, the EGFR signaling pathway is frequently hyperactivated via receptor and/or ligand overexpression and downstream oncogenic
[...] Read more.
The epidermal growth factor receptor (EGFR) plays a critical role in regulating essential cellular processes that are frequently hijacked to promote cancer. In colorectal cancer (CRC) in particular, the EGFR signaling pathway is frequently hyperactivated via receptor and/or ligand overexpression and downstream oncogenic mutations. Current EGFR-targeted therapies for metastatic CRC (mCRC) include the mAbs cetuximab and panitumumab. However, intrinsic and acquired resistance to EGFR-targeted mAbs are commonly observed. Thus, additional biomarkers are necessary to better understand patient sensitivity to EGFR-targeted therapies. Furthermore, therapeutic targeting of alternative EGFR pathway components may serve as one mechanism to overcome EGFR-targeted mAb resistance. In this review, we discuss the mounting evidence supporting EGFR ligands epiregulin (EREG) and amphiregulin (AREG), which are overexpressed in CRC with potential key roles in tumor progression, as predictive biomarkers for EGFR-targeted therapy sensitivity, as well as mediators of therapy resistance, though further studies are necessary to validate the prognostic roles and mechanisms by which these ligands contribute to resistance. Additionally, we review recent advances towards therapeutic targeting of EREG and AREG in cancer through the development and use of EREG- and AREG-targeted mAbs as well as antibody–drug conjugates (ADCs). We conclude with a discussion on the roadblocks to clinical implementation of EREG and AREG as biomarkers, as well as approaches to enhance the efficacy of current EREG- and AREG-targeted strategies.
Full article

Figure 1
Open AccessArticle
Association of JAK2 Haplotype GGCC_46/1 with the Response to Onco-Drug in MPNs Patients Positive for JAK2V617F Mutation
by
Michela Perrone, Sara Sergio, Amalia Tarantino, Giuseppina Loglisci, Rosella Matera, Davide Seripa, Michele Maffia and Nicola Di Renzo
Onco 2024, 4(3), 241-256; https://doi.org/10.3390/onco4030018 - 21 Sep 2024
Abstract
►▼
Show Figures
Background: JAK2 V617F is a somatic mutation associated with myeloproliferative neoplasms (MPNs) including polycythemia vera (PV), essential thrombocythemia (ET), and primary myelofibrosis (PMF). In MPNs, this mutation is associated with the germline GGCC (46/1) haplotype. Several studies associated JAK2 haplotype GGCC_46/1 with some
[...] Read more.
Background: JAK2 V617F is a somatic mutation associated with myeloproliferative neoplasms (MPNs) including polycythemia vera (PV), essential thrombocythemia (ET), and primary myelofibrosis (PMF). In MPNs, this mutation is associated with the germline GGCC (46/1) haplotype. Several studies associated JAK2 haplotype GGCC_46/1 with some MPNs clinical parameters, but not one explore the link between JAK2 haplotype GGCC_46/1 and onco-drug resistance. Thus, we assessed for the JAK2 46/1 haplotype’s correlation with therapy response in JAK2 V617F-positive patients. Methods: Patients with MPN, selected by the Hematology Laboratory of “V. Fazzi” Hospital (LE), were analyzed with RLFP-PCR assay with rs10974944 SNP. Results: Results show how the majority of patients had PV (63%) or PMF (61%) and that 58% of patients who developed drug resistance had the C/G genotype, while only 11% had the G/G allele. While no direct correlation between JAK2 46/1 haplotype variants and drug resistance was found, the G/G allele was associated with disease progression to myelofibrosis and certain resistance-related clinical parameters (p = 0.002449, odds ratio = 3.701209). Conclusions: Although other analyses are required, due to the narrow cardinality of sample, our findings suggest how the G/G allele could be useful for MPNs diagnosis and for the prediction of the disease outcome.
Full article

Figure 1
Open AccessReview
State of the Art on CAR T-Cell Therapies for Onco-Haematological Disorders and Other Conditions
by
Jose Alejandro Madrigal and José C. Crispín
Onco 2024, 4(3), 232-240; https://doi.org/10.3390/onco4030017 - 8 Sep 2024
Abstract
►▼
Show Figures
The use of chimeric antigen receptors (CAR T-cells) for the treatment of patients with malignant haematological diseases has become a well-established application for conditions such as refractory or relapsed B-cell acute lymphoblastic leukaemia (B-ALL), B-cell lymphomas (BCL), and multiple myeloma (MM). Nearly 35,000
[...] Read more.
The use of chimeric antigen receptors (CAR T-cells) for the treatment of patients with malignant haematological diseases has become a well-established application for conditions such as refractory or relapsed B-cell acute lymphoblastic leukaemia (B-ALL), B-cell lymphomas (BCL), and multiple myeloma (MM). Nearly 35,000 patients have received autologous CAR T-cells for the treatment of these conditions only in the USA. Since their approval by the Food and Drug Administration (FDA) in 2017, over 1200 clinical trials have been initiated globally and there are at least 10 different CAR T-cells with approval by different regulatory agencies around the globe. In the USA, the FDA has approved six commercial CAR T-cells that are widely distributed worldwide. At the time of writing, several clinical trials have been performed in patients with solid tumours such as glioblastoma, renal and pancreatic cancer, as well as in patients with autoimmune conditions such as systemic lupus erythematosus (SLE), idiopathic inflammatory myositis (IIM), and systemic sclerosis (SS). There are also several studies showing the potential benefit of CAR T-cells for other non-malignant diseases such as asthma and even fungal infections. In this review, without pretending to cover all current areas of treatments with CAR T-cells, we offer a brief summary of some of the most relevant aspects of the use of CAR T-cells for some of these conditions.
Full article

Figure 1
Open AccessReview
Targeting the Hippo- Yes-Associated Protein/Transcriptional Coactivator with PDZ-Binding Motif Signaling Pathway in Primary Liver Cancer Therapy
by
Yina Wang and Liangyou Rui
Onco 2024, 4(3), 217-231; https://doi.org/10.3390/onco4030016 - 22 Aug 2024
Abstract
►▼
Show Figures
Liver cancer imposes a pervasive global health challenge, ranking among the most prevalent cancers worldwide. Its prevalence and mortality rates are on a concerning upward trajectory and exacerbated by the dearth of efficacious treatment options. The Hippo signaling pathway, originally discovered in Drosophila,
[...] Read more.
Liver cancer imposes a pervasive global health challenge, ranking among the most prevalent cancers worldwide. Its prevalence and mortality rates are on a concerning upward trajectory and exacerbated by the dearth of efficacious treatment options. The Hippo signaling pathway, originally discovered in Drosophila, comprises the following four core components: MST1/2, WW45, MOB1A/B, and LATS1/2. This pathway regulates the cellular localization of the transcriptional coactivator Yes-associated protein/transcriptional coactivator with PDZ-binding motif (YAP/TAZ) through a series of enzymatic reactions. The Hippo-YAP/TAZ pathway maintains a balance between cell proliferation and apoptosis, regulates tissue and organ sizes, and stabilizes the internal environment. Abnormalities of any genes within the Hippo signaling pathway, such as deletion or mutation, disturb the delicate balance between cell proliferation and apoptosis, creating a favorable condition for tumor initiation and progression. Mutations or epigenetic alterations in the Hippo signaling pathway components can lead to its inactivation. Consequently, YAP/TAZ becomes overexpressed and activated, promoting excessive cell proliferation and inhibiting apoptosis. This dysregulation is closely associated with the development of liver cancer. This review discusses the pivotal role of the Hippo signaling pathway in the pathogenesis and progression of liver cancer. By elucidating its mechanisms, we aim to offer new insights into potential therapeutic targets for effectively combating liver cancer.
Full article

Figure 1
Open AccessArticle
Impact of Physical Exercise on Quality of Life, Self-Esteem, and Depression in Breast Cancer Survivors: A Pilot Study
by
Eduarda Maria Rocha Teles de Castro Coelho, Helena Isabel Azevedo Mendes, Carla Afonso Varajidás and Sandra Celina Fernandes Fonseca
Onco 2024, 4(3), 207-216; https://doi.org/10.3390/onco4030015 - 22 Aug 2024
Abstract
Controlled study designs usually report that physical exercise improves the health of women living with breast cancer. However, many of these women are not sufficiently active to experience the benefits of exercise. The main objective was to analyze the effect of a physical
[...] Read more.
Controlled study designs usually report that physical exercise improves the health of women living with breast cancer. However, many of these women are not sufficiently active to experience the benefits of exercise. The main objective was to analyze the effect of a physical exercise program on quality of life, self-esteem, and depression in breast cancer survivors. Thirteen participants (46.54 ± 6.31 years old) completed the exercise intervention. Three patient-reported questionnaires were used: Supplementary Questionnaire Breast Cancer Module (QLQ-BR23), Beck Depression Inventory (BDI), and Rosenberg Self-Esteem Scale (RSES). All participants had significantly improved self-esteem (p = 0.004). Although there were no statistically significant changes in depression, there was a notable decrease in scores (6.39 ± 4.75 vs. 5.00 ± 4.75; p = 0.080). Regarding quality of life, significant improvements were observed in “future perspectives” (p = 0.047) and “arm symptoms” (p = 0.015). No significant changes were noted in the other variables. Our results suggest that physical exercise is an effective strategy that positively affects breast cancer survivors’ quality of life and self-esteem. The results reinforce the need for community-based exercise programs for breast cancer survivors. Healthcare professionals should promote physical exercise to improve health outcomes before, during, and after treatment.
Full article
(This article belongs to the Special Issue The Evolving Landscape of Contemporary Cancer Therapies)
►▼
Show Figures

Figure 1
Open AccessArticle
Predicting Resistance to Immunotherapy in Melanoma, Glioblastoma, Renal, Stomach and Bladder Cancers by Machine Learning on Immune Profiles
by
Guillaume Mestrallet
Onco 2024, 4(3), 192-206; https://doi.org/10.3390/onco4030014 - 20 Aug 2024
Abstract
►▼
Show Figures
Strategies for tackling cancer involve surgery, radiotherapy, chemotherapy, and immune checkpoint inhibitors (ICB). However, the effectiveness of ICB remains constrained, prompting the need for a proactive strategy to foresee treatment responses and resistances. This study undertook an analysis across diverse cancer patient cohorts
[...] Read more.
Strategies for tackling cancer involve surgery, radiotherapy, chemotherapy, and immune checkpoint inhibitors (ICB). However, the effectiveness of ICB remains constrained, prompting the need for a proactive strategy to foresee treatment responses and resistances. This study undertook an analysis across diverse cancer patient cohorts (including melanoma, clear cell renal carcinoma, glioblastoma, bladder, and stomach cancers) subjected to various immune checkpoint blockade treatments. Surprisingly, our findings unveiled that over 38% of patients demonstrated resistance and persistent disease progression despite undergoing ICB intervention. To unravel the intricacies of resistance, we scrutinized the immune profiles of cancer patients experiencing ongoing disease progression and resistance post-ICB therapy. These profiles delineated multifaceted defects, including compromised macrophage, monocyte, and T cell responses, impaired antigen presentation, aberrant regulatory T cell (Tregs) responses, and an elevated expression of immunosuppressive and G protein-coupled receptor molecules (TGFB1, IL2RA, IL1B, EDNRB, ADORA2A, SELP, and CD276). Building upon these insights into resistance profiles, we harnessed machine learning algorithms to construct models predicting the response and resistance to ICB and developed the accompanying software. While previous work on glioblastoma with only one type of algorithm had an accuracy of 0.82, we managed to develop 20 models that provided estimates of future events of resistance or response in five cancer types, with accuracies ranging between 0.79 and 1, based on their distinct immune characteristics. In conclusion, our approach advocates for the personalized application of immunotherapy in cancer patients based on patient-specific attributes and computational models.
Full article

Figure 1
Open AccessArticle
A Systems Biology Analysis of Chronic Lymphocytic Leukemia
by
Giulia Pozzati, Jinrui Zhou, Hananel Hazan, Giannoula Lakka Klement, Hava T. Siegelmann, Jack A. Tuszynski and Edward A. Rietman
Onco 2024, 4(3), 163-191; https://doi.org/10.3390/onco4030013 - 6 Aug 2024
Abstract
►▼
Show Figures
Whole-genome sequencing has revealed that TP53, NOTCH1, ATM, SF3B1, BIRC3, ABL, NXF1, BCR, and ZAP70 are often mutated in CLL, but not consistently across all CLL patients. This paper employs a statistical thermodynamics approach in combination with the systems biology of the CLL
[...] Read more.
Whole-genome sequencing has revealed that TP53, NOTCH1, ATM, SF3B1, BIRC3, ABL, NXF1, BCR, and ZAP70 are often mutated in CLL, but not consistently across all CLL patients. This paper employs a statistical thermodynamics approach in combination with the systems biology of the CLL protein–protein interaction networks to identify the most significant participant proteins in the cancerous transformation. Betti number (a topology of complexity) estimates highlight a protein hierarchy, primarily in the Wnt pathway known for aberrant CLL activation. These individually identified proteins suggest a network-targeted strategy over single-target drug development. The findings advocate for a multi-target inhibition approach, limited to several key proteins to minimize side effects, thereby providing a foundation for designing therapies. This study emphasizes a shift towards a comprehensive, multi-scale analysis to enhance personalized treatment strategies for CLL, which could be experimentally validated using siRNA or small-molecule inhibitors. The result is not just the identification of these proteins but their rank-order, offering a potent signal amplification in the context of the 20,000 proteins produced by the human body, thus providing a strategic basis for therapeutic intervention in CLL, underscoring the necessity for a more holistic, cellular, chromosomal, and genome-wide study to develop tailored treatments for CLL patients.
Full article

Figure 1
Open AccessArticle
A Transformative Technology Linking Patient’s mRNA Expression Profile to Anticancer Drug Efficacy
by
Chen Yeh, Shu-Ti Lin and Hung-Chih Lai
Onco 2024, 4(3), 143-162; https://doi.org/10.3390/onco4030012 - 14 Jul 2024
Cited by 1
Abstract
►▼
Show Figures
As precision medicine such as targeted therapy and immunotherapy often have limited accessibility, low response rate, and evolved resistance, it is urgent to develop simple, low-cost, and quick-turnaround personalized diagnostic technologies for drug response prediction with high sensitivity, speed, and accuracy. The major
[...] Read more.
As precision medicine such as targeted therapy and immunotherapy often have limited accessibility, low response rate, and evolved resistance, it is urgent to develop simple, low-cost, and quick-turnaround personalized diagnostic technologies for drug response prediction with high sensitivity, speed, and accuracy. The major challenges of drug response prediction strategies employing digital database modeling are the scarcity of labeled clinical data, applicability only to a few classes of drugs, and losing the resolution at the individual patient level. Although these challenges have been partially addressed by large-scale cancer cell line datasets and more patient-relevant cell-based systems, the integration of different data types and data translation from pre-clinical to clinical utilities are still far-fetched. To overcome the current limitations of precision medicine with a clinically proven drug response prediction assay, we have developed an innovative and proprietary technology based on in vitro patient testing and in silico data analytics. First, a patient-derived gene expression signature was established via the transcriptomic profiling of cell-free mRNA (cfmRNA) from the patient’s blood. Second, a gene-to-drug data fusion and overlaying mechanism to transfer data were performed. Finally, a semi-supervised method was used for the database searching, matching, annotation, and ranking of drug efficacies from a pool of ~700 approved, investigational, or clinical trial drug candidates. A personalized drug response report can be delivered to inform clinical decisions within a week. The PGA (patient-derived gene expression-informed anticancer drug efficacy) test has significantly improved patient outcomes when compared to the treatment plans without PGA support. The implementation of PGA, which combines patient-unique cfmRNA fingerprints with drug mapping power, has the potential to identify treatment options when patients are no longer responding to therapy and when standard-of-care is exhausted.
Full article

Figure 1
Open AccessArticle
Revisiting the Role of PD-L1 Overexpression in Prognosis and Clinicopathological Features in Patients with Oral Squamous Cell Carcinoma
by
Fernando Leporace-Jiménez, Isabel Portillo-Hernandez, Justino Jiménez-Almonacid, Ignacio Zubillaga Rodriguez, María Mejía-Nieto, Pablo Caballero Pedrero and Gregorio Sanchez Aniceto
Onco 2024, 4(3), 131-142; https://doi.org/10.3390/onco4030011 - 12 Jul 2024
Abstract
Background: PD1 and its ligand PD-L1 are related to prognosis in many solid tumors; however, their role in oral squamous cell carcinoma (OSCC) remains unclear. Methods: A retrospective monocentric study including all patients with OSCC diagnosed and treated between January 2020 and May
[...] Read more.
Background: PD1 and its ligand PD-L1 are related to prognosis in many solid tumors; however, their role in oral squamous cell carcinoma (OSCC) remains unclear. Methods: A retrospective monocentric study including all patients with OSCC diagnosed and treated between January 2020 and May 2022 was performed. PD-L1 expression was assessed per a combined positive score (CPS), considering a CPS of > or equal to 1 as positive (1–20 indicating “low expression” and ≥20 indicating “high”). A descriptive analysis of the patient cohort and tumors was performed, including tumor size, stage, lymph node involvement, recurrence, and survival. Results: In total, 65 patients (65 tumors) were analyzed. A total of 66.15% of the tumors were in advanced stages (III-IV), of which 97.67% expressed PD-L1+, compared with 71.42% in the early stages (I–II). T4 tumors expressed PD-L1 in 100% of cases, compared with 54% in T1 tumors. A total of 50.79% of the tumors showed lymph node involvement (pN+), with 100% of the pN+ showing PD-L1+. The prevalence of pN+ was 59.38% vs. 40.63% for high vs. low PD-L1 expression, respectively. Patients’ follow-ups ranged from 2 to 34.5 months. No significant difference was seen between overall survival (OS) and PD-L1 +/− (CPS ≥ 1 vs. CPS < 1) or high (CPS ≥ 20) and low (CPS < 20) PD-L1 expression (p < 0.97 and 0.64, respectively). Conclusions: The method used to measure PD-L1 (a laboratory test with Dako 22C3 anti-PD-L1 primary antibodies) was reliable and accurate, with a correlation coefficient between PD-L1 expression in the biopsy and the surgical piece of 0.83 (p < 0.0001). A CPS of ≥1 was observed in large tumors (p < 0.001) and was correlated with that of lymph node metastases (p < 0.004). Further analysis of PD-L1 expression in OSCC and studies to determine its relevance in tumor biology and prognosis is needed.
Full article
(This article belongs to the Topic Individualized Molecular Mechanisms and Treatment in Tumor Metastasis)
►▼
Show Figures

Figure 1
Open AccessReview
The World of Immunotherapy Needs More Than PD-1/PD-L1—Two of the New Kids on the Block: LAG-3 and TIGIT
by
João Martins Gama, Paulo Teixeira and Rui Caetano Oliveira
Onco 2024, 4(3), 116-130; https://doi.org/10.3390/onco4030010 - 1 Jul 2024
Abstract
Immunotherapy has paved the way for the development of solid tumor new treatments in the last decade. The approval of immune checkpoint inhibitors such as anti PD-1/PD-L1 provided a revolution with optimal results. However, a considerable proportion of patients experience adverse therapeutic effects,
[...] Read more.
Immunotherapy has paved the way for the development of solid tumor new treatments in the last decade. The approval of immune checkpoint inhibitors such as anti PD-1/PD-L1 provided a revolution with optimal results. However, a considerable proportion of patients experience adverse therapeutic effects, and up to 50% may develop secondary resistance in the first three to five years. This has prompted the need for identifying new targets for immunotherapy that have good tolerance and biosafety and, of course, good tumoral response, either alone or in combination. Two of these new targets are the Lymphocyte-activation gene 3 (LAG-3) and the T cell immunoglobulin and ITIM domain (TIGIT). They are responsible for several interactions with the immune system, prompting an immunosuppressive phenotype in the tumor microenvironment. Both LAG-3 and TIGIT can be druggable, alone or in combination with anti-PD-1/PD-L1, with rather safe profiles making them attractive. In this review, we highlight some of the immune mechanisms of TIGIT and LAG-3 and their detection by immunohistochemistry, providing some insight into their use in the clinical setting.
Full article
(This article belongs to the Topic Individualized Molecular Mechanisms and Treatment in Tumor Metastasis)
►▼
Show Figures

Figure 1
Open AccessReview
The Prognostic Role of Prognostic Nutritional Index and Controlling Nutritional Status in Predicting Survival in Older Adults with Oncological Disease: A Systematic Review
by
Ana Filipa Ferreira, Tatiana Fernandes, Maria do Carmo Carvalho and Helena Soares Loureiro
Onco 2024, 4(2), 101-115; https://doi.org/10.3390/onco4020009 - 2 Jun 2024
Abstract
►▼
Show Figures
The increase in new cancer diagnoses in the elderly calls for new, accessible, and easy-to-use prognostic tools that contribute to lowering the burden of the disease. Recognising the importance of inflammation and nutritional status in the progression of the disease, the purpose of
[...] Read more.
The increase in new cancer diagnoses in the elderly calls for new, accessible, and easy-to-use prognostic tools that contribute to lowering the burden of the disease. Recognising the importance of inflammation and nutritional status in the progression of the disease, the purpose of this systematic review was to synthesise the evidence on the prognostic role of Prognostic Nutritional Index (PNI) and Controlling Nutritional Status (CONUT) in predicting survival of older adult cancer patients. A comprehensive search was conducted in PubMed and Web of Science Core Collection databases until 22 February 2024. The articles included in this review (n = 38) examined the relationships of PNI and CONUT with survival outcomes in elderly cancer patients. Despite high heterogeneity between the studies, most concluded that low PNI values are associated with poor overall survival (OS), particularly in gastric cancer patients. Most studies did not find an association between PNI and cancer-specific survival, progression-free survival, disease-free survival, recurrence-free survival, and mortality. Results regarding the prognostic role of CONUT in predicting survival were inconclusive. This study suggests that PNI could be used to predict OS in elderly cancer patients, while more studies are needed to assess the prognostic role of CONUT.
Full article

Figure 1
Open AccessReview
Can Tumour Antigens Act as Biomarkers for the Early Detection of Non-Small Cell Lung Cancer?
by
Eithar Mohamed, Daniel Fletcher, Simon Hart and Barbara-ann Guinn
Onco 2024, 4(2), 87-100; https://doi.org/10.3390/onco4020008 - 31 May 2024
Abstract
Lung cancer (LC) is one of the leading causes of cancer-related deaths. Pulmonary nodules are one of the risk factors, and their discovery rate has been increasing due to enhanced performance of chest CT scans, but more than 90% are non-malignant, causing unnecessary
[...] Read more.
Lung cancer (LC) is one of the leading causes of cancer-related deaths. Pulmonary nodules are one of the risk factors, and their discovery rate has been increasing due to enhanced performance of chest CT scans, but more than 90% are non-malignant, causing unnecessary stress to patients and costs to healthcare providers. Early diagnosis of LC is associated with a 5-year survival rate of up to 75% following surgical resection, but LC is often diagnosed late due to a lack of symptoms and poor 5-year survival rates as low as 10%. The cost of LC diagnosis is high, with 40% of it associated with benign lesions, which are difficult to differentiate from malignant lesions. Tumour-associated antigens (TAAs) may provide one way in which LC could be diagnosed early using minimally-invasive techniques, under their association with immune responses and specificity for disease. Here we discuss the potential of cancer-testis antigens (CTAs) to act as non-invasive biomarkers for the early detection of non-small cell lung cancer.
Full article
(This article belongs to the Special Issue Progress in Vaccination against Cancer - 2023 (PIVAC-23))
►▼
Show Figures

Graphical abstract
Open AccessArticle
Inhibitory Effects of Metformin for Pancreatic Neuroendocrine Neoplasms: Experimental Study on Mitochondrial Function
by
Shogo Maruzen, Seiichi Munesue, Mitsuyoshi Okazaki, Satoshi Takada, Shinichi Nakanuma, Isamu Makino, Linxiang Gong, Susumu Kohno, Chiaki Takahashi, Hidehiro Tajima, Yasuhiko Yamamoto and Shintaro Yagi
Onco 2024, 4(2), 77-86; https://doi.org/10.3390/onco4020007 - 27 Apr 2024
Abstract
►▼
Show Figures
Although pancreatic neuroendocrine neoplasms (panNENs) are much less common and have a better prognosis than exocrine pancreatic cancers, their recurrence rate is not low, even in Grade 1 (World Health Organization classification) panNEN. Recently, there have been several reports that the progression-free survival
[...] Read more.
Although pancreatic neuroendocrine neoplasms (panNENs) are much less common and have a better prognosis than exocrine pancreatic cancers, their recurrence rate is not low, even in Grade 1 (World Health Organization classification) panNEN. Recently, there have been several reports that the progression-free survival in patients with unresectable panNEN could be improved by an antidiabetic drug, metformin, with the co-treatment of everolimus or a somatostatin analog. In this study, we aimed to evaluate the effects of metformin on cell metabolism and viability using the panNEN cell line, QGP-1, and RIN-m in culture. We observed an inhibitory effect of metformin on QGP-1 cell proliferation in a dose-dependent manner. Metformin was found to decrease the oxygen consumption rate in QGP-1 and RIN-m cells after metformin 48 h treatment and immediately after exposure. Cell proliferation was suppressed after metformin treatment. Phosphorylated adenosine monophosphate-activated protein kinase (AMPK) expression was increased, and cyclin D1 expression was decreased in RIN-m cells 24 h after metformin treatment by Western blotting in a dose-dependent manner. In conclusion, suppressive mitochondrial respiration and AMPK activation by metformin are, thus, suggested to inhibit panNEN cell viability and cell survival.
Full article
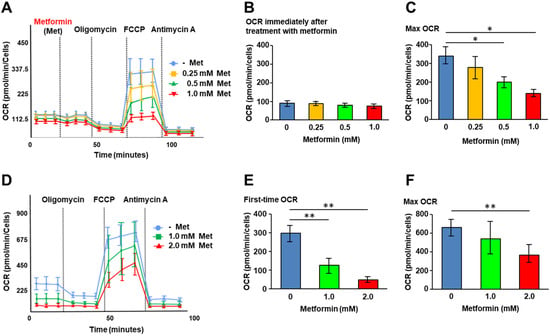
Figure 1
Open AccessCommentary
How Reliable Are Predictions of CD8+ T Cell Epitope Recognition? Lessons for Cancer
by
Alexander A. Lehmann, Paul V. Lehmann and Stephen Todryk
Onco 2024, 4(2), 68-76; https://doi.org/10.3390/onco4020006 - 17 Apr 2024
Abstract
Synthetic peptides derived from antigen sequences are essential reagents for the detection of CD8+ cytotoxic T lymphocytes (CTLs), in assays such as ELISPOT/ImmunoSpot®. Indeed, the combination of peptides and ImmunoSpot® has been widely used for immune monitoring in numerous
[...] Read more.
Synthetic peptides derived from antigen sequences are essential reagents for the detection of CD8+ cytotoxic T lymphocytes (CTLs), in assays such as ELISPOT/ImmunoSpot®. Indeed, the combination of peptides and ImmunoSpot® has been widely used for immune monitoring in numerous vaccine trials. Target antigens in pathogens or cancers may be large in size and multiple in number, often seemingly necessitating in silico peptide epitope predictions using algorithms and programs for certain HLA alleles to narrow down the numbers of required peptides. In this commentary, we discuss our data in the context of immune responses to viral and cancer antigens, concluding that systematic high-throughput immune monitoring of CD8+ T cells will provide more reliable insights on the host’s response to cancer than the reliance on select CD8+ T cell epitopes, no matter whether these are in silico predicted or even if they had been empirically established. We show the feasibility of large scale, high-throughput systematic CD8+ T cell epitope testing towards this goal.
Full article
(This article belongs to the Special Issue Progress in Vaccination against Cancer - 2023 (PIVAC-23))
►▼
Show Figures
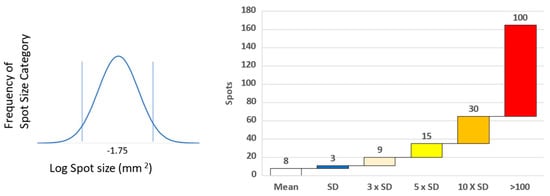
Figure 1
Open AccessArticle
Deficiency in DNA Damage Repair Proteins Promotes Prostate Cancer Cell Migration through Oxidative Stress
by
Philippa Lantwin, Adam Kaczorowski, Cathleen Nientiedt, Constantin Schwab, Martina Kirchner, Viktoria Schütz, Magdalena Görtz, Markus Hohenfellner, Anette Duensing, Albrecht Stenzinger and Stefan Duensing
Onco 2024, 4(2), 56-67; https://doi.org/10.3390/onco4020005 - 28 Mar 2024
Abstract
►▼
Show Figures
Introduction: DNA damage repair gene deficiency defines a subgroup of prostate cancer patients with early metastatic progression and unfavorable disease outcome. Whether deficiency in DNA damage repair genes directly promotes metastatic dissemination is not completely understood. Methods: The migratory behavior of prostate cancer
[...] Read more.
Introduction: DNA damage repair gene deficiency defines a subgroup of prostate cancer patients with early metastatic progression and unfavorable disease outcome. Whether deficiency in DNA damage repair genes directly promotes metastatic dissemination is not completely understood. Methods: The migratory behavior of prostate cancer cells was analyzed after siRNA-mediated knockdown of DNA damage repair and checkpoint proteins, including BRCA2, ATM, and others, using transwell migration assays, scratch assays and staining for F-actin to ascertain cell circularity. Cells deficient in BRCA2 or ATM were tested for oxidative stress by measuring reactive oxygen species (ROS). The effects of ROS inhibition on cell migration were analyzed using the antioxidant N-acetylcysteine (NAC). The correlation between BRCA2 deficiency and oxidative stress was ascertained via immunohistochemistry for methylglyoxal (MG)-modified proteins in 15 genetically defined primary prostate cancers. Results: Prostate cancer cells showed a significantly increased migratory activity after the knockdown of BRCA2 or ATM. There was a significant increase in ROS production in LNCaP cells after BRCA2 knockdown and in PC-3 cells after BRCA2 or ATM knockdown. Remarkably, the ROS scavenger NAC abolished the enhanced motility of prostate cancer cells after the knockdown of BRCA2 or ATM. Primary prostate cancers harboring genetic alterations in BRCA2 showed a significant increase in MG-modified proteins, indicating enhanced oxidative stress in vivo. Conclusions: Our results indicate that DNA damage repair gene deficiency may contribute to the metastatic dissemination of prostate cancer through enhanced tumor cell migration involving oxidative stress.
Full article
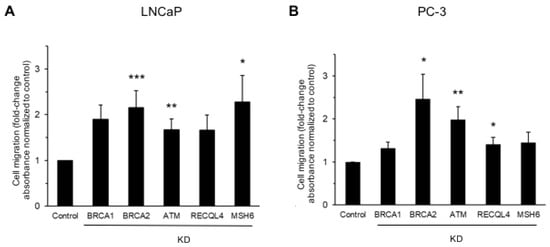
Figure 1
Open AccessReview
Dendritic Cell Immunotherapy for Ovarian Cancer: An Overview of Our Achievements
by
Jiřina Bartůňková
Onco 2024, 4(1), 46-55; https://doi.org/10.3390/onco4010004 - 21 Mar 2024
Abstract
Epithelial ovarian carcinoma (EOC) is the fifth leading cause of cancer-related death in women, largely reflecting the early dissemination of this malignant disease to the peritoneum. Due to its immunological features, EOC has poor response to immune checkpoint inhibitors (ICIs), including a limited
[...] Read more.
Epithelial ovarian carcinoma (EOC) is the fifth leading cause of cancer-related death in women, largely reflecting the early dissemination of this malignant disease to the peritoneum. Due to its immunological features, EOC has poor response to immune checkpoint inhibitors (ICIs), including a limited tumor mutational burden (TMB), poor infiltration by immune cells, and active immunosuppression. Thus, novel strategies are needed to overcome the frequent lack of pre-existing immunity in patients with EOC. We developed and tested an autologous dendritic cell (DC)-based vaccine (DCVAC), which has recently been shown to be safe and to significantly improve progression-free survival (PFS) in two independent randomized phase II clinical trials enrolling patients with EOC (SOV01, NCT02107937; SOV02, NCT02107950). In addition, our exploratory data analyses suggest that the clinical benefits of the DCVAC were more pronounced in patients with EOC with lower-than-median TMBs and reduced CD8+ T cell infiltration. Thus, the DC-based vaccine stands out as a promising clinical tool to jumpstart anticancer immunity in patients with immunologically “cold” EOC. Our findings underscore the need for personalized immunotherapy and the clinical relevance of potential tumor-related biomarkers within the immunotherapy field. Additional clinical trials are needed to address these strategies as well as the potential value of the TMB and immune infiltration at baseline as biomarkers for guiding the clinical management of EOC.
Full article
(This article belongs to the Special Issue Progress in Vaccination against Cancer - 2023 (PIVAC-23))
►▼
Show Figures
Figure 1
Open AccessCommentary
When Therapy-Induced Cancer Cell Apoptosis Fuels Tumor Relapse
by
Razmik Mirzayans
Onco 2024, 4(1), 37-45; https://doi.org/10.3390/onco4010003 - 4 Feb 2024
Cited by 1
Abstract
Most therapeutic strategies for solid tumor malignancies are designed based on the hypothesis that cancer cells evade apoptosis to exhibit therapy resistance. This is somewhat surprising given that clinical studies published since the 1990s have demonstrated that increased apoptosis in solid tumors is
[...] Read more.
Most therapeutic strategies for solid tumor malignancies are designed based on the hypothesis that cancer cells evade apoptosis to exhibit therapy resistance. This is somewhat surprising given that clinical studies published since the 1990s have demonstrated that increased apoptosis in solid tumors is associated with cancer aggressiveness and poor clinical outcome. This is consistent with more recent reports demonstrating non-canonical (pro-survival) roles for apoptotic caspases, including caspase 3, as well as the ability of cancer cells to recover from late stages of apoptosis via a process called anastasis. These activities are essential for the normal development and maintenance of a healthy organism, but they also enable malignant cells (including cancer stem cells) to resist anticancer treatment and potentially contribute to clinical dormancy (minimal residual disease). Like apoptosis, therapy-induced cancer cell dormancy (durable proliferation arrest reflecting various manifestations of genome chaos) is also not obligatorily a permanent cell fate. However, as briefly discussed herein, compelling pre-clinical studies suggest that (reversible) dormancy might be the “lesser evil” compared to treacherous apoptosis.
Full article
(This article belongs to the Special Issue Targeting of Tumor Dormancy Pathway)
►▼
Show Figures
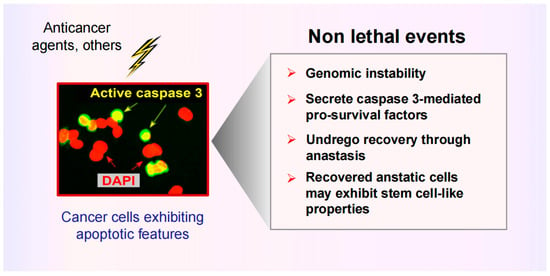
Figure 1
Open AccessReview
Significance of PET/CT Imaging in Myeloma Assessment: Exploring Novel Applications beyond Osteolytic Lesion Detection and Treatment Response
by
Mahdi Zirakchian Zadeh
Onco 2024, 4(1), 15-36; https://doi.org/10.3390/onco4010002 - 9 Jan 2024
Abstract
►▼
Show Figures
In multiple myeloma (MM), specific cytokines produced by plasma cells disrupt the equilibrium between osteoblasts and osteoclasts. As a result, MM patients experience an increase in osteoclast activity and a decrease in osteoblast activity. This disparity is fundamental to the development of myeloma
[...] Read more.
In multiple myeloma (MM), specific cytokines produced by plasma cells disrupt the equilibrium between osteoblasts and osteoclasts. As a result, MM patients experience an increase in osteoclast activity and a decrease in osteoblast activity. This disparity is fundamental to the development of myeloma bone disease. Lytic lesions, which are a feature of MM, can result in pathologic fractures and excruciating pain. For many years, whole-body X-ray radiography has been the standard imaging method for identifying lytic lesions. However, its sensitivity is limited because it can only detect lesions once the bone mass has been reduced by 30% to 50%. Hence, utilizing advanced and sensitive imaging modalities, such as positron emission tomography (PET) fused with computed tomography (CT), is crucial for the early detection of osteolytic lesions. Among radiotracers used in PET imaging, 1⁸F-fluorodeoxyglucose ([18F]FDG) is the most commonly employed in the field of oncology. Currently, most guidelines include [18F]FDG PET/CT in the assessment of myeloma patients, particularly for detecting osteolytic lesions, evaluating treatment response, and assessing extramedullary and residual disease. Nonetheless, in recent years, new applications of PET/CT for evaluating myeloma have been investigated. These include assessing aspects such as bone turnover, dual-time-point imaging (early and delayed scans), the impact of chemotherapy on the brain (commonly known as ‘chemo brain’), innovative PET radiotracers, and the use of artificial intelligence technology. This article aims to provide a comprehensive review of both conventional and innovative uses of PET/CT in evaluating multiple myeloma.
Full article
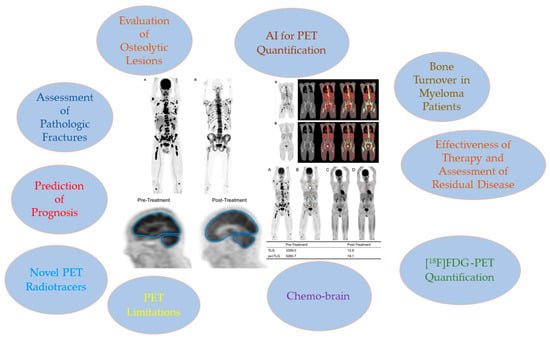
Figure 1
Open AccessSystematic Review
Predictors of Complete Pathological Response with Chemoimmunotherapy in Triple-Negative Breast Cancer: A Meta-Analysis
by
Arya Mariam Roy, Supritha Chintamaneni, Sabah Alaklabi, Hassan Awada, Kristopher Attwood and Shipra Gandhi
Onco 2024, 4(1), 1-14; https://doi.org/10.3390/onco4010001 - 28 Dec 2023
Cited by 1
Abstract
►▼
Show Figures
Background: Multiple randomized controlled trials (RCTs) have investigated the impact of adding checkpoint inhibitors to neoadjuvant chemotherapy for triple-negative breast cancer (TNBC) patients. However, there is a lack of biomarkers that can help identify patients who would benefit from combination therapy. Our research
[...] Read more.
Background: Multiple randomized controlled trials (RCTs) have investigated the impact of adding checkpoint inhibitors to neoadjuvant chemotherapy for triple-negative breast cancer (TNBC) patients. However, there is a lack of biomarkers that can help identify patients who would benefit from combination therapy. Our research identifies response predictors and assesses the effectiveness of adding immunotherapy to neoadjuvant chemotherapy for TNBC patients. Methods: We identified eligible RCTs by searching PubMed, Cochrane CENTRAL, Embase, and oncological meetings. For this meta-analysis, we obtained odds ratios using the standard random effects model. To assess the heterogeneity of the study outcomes, the I2 statistic was obtained. Potential bias was assessed using a funnel plot and the corresponding Egger’s test. Results: In total, 1637 patients with TNBC were included from five RCTs. Neoadjuvant chemoimmunotherapy significantly improved pCR when compared to neoadjuvant chemotherapy alone. In the subgroup analysis, neoadjuvant chemoimmunotherapy showed higher pCR rates in both Programmed death-ligand 1 (PD-L1)-positive and PD-L1-negative TNBC patients. An Eastern Cooperative Oncology Group (ECOG) performance score (PS) of 0 correlated with increased pCRs (OR = 1.9, p < 0.001) in neoadjuvant chemoimmunotherapy vs. neoadjuvant chemotherapy, but no benefit was observed for patients with ECOG PS 1. Nodal positivity was significantly associated with pCR (OR = 2.52, p < 0.001), while neoadjuvant chemoimmunotherapy did not benefit patients with negative lymph nodes. Conclusions: Checkpoint inhibition and neoadjuvant chemotherapy significantly increased pCRs in TNBC patients, regardless of their PDL-1 status. Additional checkpoint inhibitors improved pCR rates, mainly for patients with ECOG PS 0 and lymph node-positive disease.
Full article
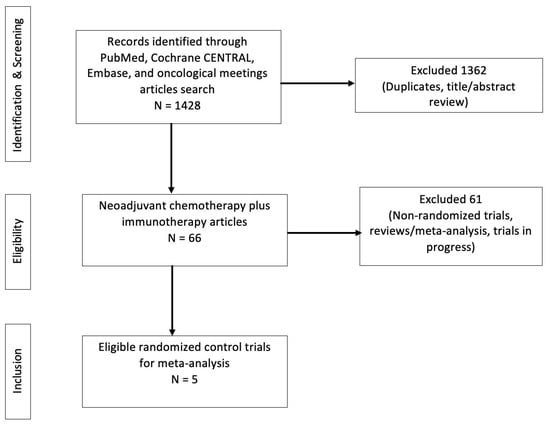
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biology, Cancers, Current Oncology, Diseases, Onco
miRNAs in Pathophysiology of Disease
Topic Editors: Francesca Orso, Roberto Coppo, Federico VirgaDeadline: 12 November 2024
Topic in
Cancers, Current Oncology, JCM, JPM, Onco
Targeting Tumor Microvasculature of Malignant Metastatic Tumor
Topic Editors: Prabhu Thirusangu, Vigneshwaran Vellingiri, Prabhakar ThippegowdaDeadline: 25 November 2024
Topic in
Biomedicines, Cancers, Cells, IJMS, Onco
Advances in Colorectal Cancer Therapy
Topic Editors: Niki Christou, Mireille Verdier, Fabrice LallouéDeadline: 20 December 2024
Topic in
Cancers, Current Oncology, Healthcare, Nursing Reports, Onco
Life of Cancer Survivor
Topic Editors: Nuno Lunet, Natália Araújo, Samantha MoraisDeadline: 24 December 2024

Conferences
Special Issues
Special Issue in
Onco
Targeting of Tumor Dormancy Pathway
Guest Editors: Athanassios Kotsinas, Constantin N. BaxevanisDeadline: 31 October 2024
Special Issue in
Onco
The Evolving Landscape of Contemporary Cancer Therapies
Guest Editor: Constantin N. BaxevanisDeadline: 15 November 2024






