Journal Description
Reactions
Reactions
is an international, peer-reviewed, open access journal on reaction chemistry and engineering published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within ESCI (Web of Science), Scopus, EBSCO, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 25.3 days after submission; acceptance to publication is undertaken in 4.8 days (median values for papers published in this journal in the first half of 2024).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Impact Factor:
2.2 (2023);
5-Year Impact Factor:
2.1 (2023)
Latest Articles
One-Pot Syntheses of [c2]Daisy-Chain Rotaxane Networks via Thiol-Ene Reaction and Its Application to Gel Electrolyte for Secondary Battery
Reactions 2024, 5(4), 800-811; https://doi.org/10.3390/reactions5040041 - 16 Oct 2024
Abstract
►
Show Figures
A new topological material, the [c2]daisy-chain rotaxane network, was successfully synthesized via a thiol-ene reaction between a [c2]daisy-chain rotaxane, which consists of a host–guest compound (H–G compound) where a crown ether and a secondary ammonium salt are linked, and a multi-branched thiol compound.
[...] Read more.
A new topological material, the [c2]daisy-chain rotaxane network, was successfully synthesized via a thiol-ene reaction between a [c2]daisy-chain rotaxane, which consists of a host–guest compound (H–G compound) where a crown ether and a secondary ammonium salt are linked, and a multi-branched thiol compound. The resulting network polymer exhibited higher compressive strength compared to one without the [c2]daisy-chain rotaxane. Additionally, the neutralized [c2]daisy-chain rotaxane network, in which the ammonium salt was neutralized and there was no interaction with the crown ether, showed increased rigidity compared to its state before neutralization. Furthermore, a gel electrolyte was prepared by impregnating the [c2]daisy-chain rotaxane network with an organic electrolyte containing dissolved lithium salts, and its ionic conductivity was investigated. As a result, high ionic conductivity was achieved despite the high polymer content.
Full article
Open AccessArticle
Revisiting Atmospheric Oxidation Kinetics of Nitrogen Oxides: The Use of Low-Cost Electrochemical Sensors to Measure Reaction Kinetics
by
Steven M. Owen, Lachlan H. Yee and Damien T. Maher
Reactions 2024, 5(4), 789-799; https://doi.org/10.3390/reactions5040040 - 15 Oct 2024
Abstract
►▼
Show Figures
The high cost of equipment is a significant entry barrier to research for smaller organisations in developing solutions to air pollution problems. Low-cost electrochemical sensors have shown sensitivity at parts-per-billion by volume (ppbV) mixing ratios but are subject to variations due to changing
[...] Read more.
The high cost of equipment is a significant entry barrier to research for smaller organisations in developing solutions to air pollution problems. Low-cost electrochemical sensors have shown sensitivity at parts-per-billion by volume (ppbV) mixing ratios but are subject to variations due to changing environmental conditions, particularly temperature. We have previously demonstrated that under isothermal/isohume conditions such as those found in kinetic studies, very stable electrochemical responses occur. In this paper, we demonstrate the utility of a low-cost IoT-based sensor system that employs four-electrode electrochemical sensors under isothermal/isohume conditions for studying the kinetics of the atmospheric oxidation of nitrogen oxides. The results suggest that reproducible results for NO and NO2 kinetics can be achieved. The method produced oxidation rates of 7.95 × 103 L2 mol−2 s−1 (±1.3%), for NO and 7.99 × 10−4 s−1 (±2.1%) for NO2. This study suggests that the oxidation kinetics of nitrogen oxides can be assessed with low-cost sensors, which can support a wide range of industrial applications, such as designing biocatalytic coatings for air pollution remediation.
Full article

Figure 1
Open AccessArticle
The Second Protonation in the Bio-Catalytic Cycles of the Enzymes Cytochrome P450 and Superoxide Reductase
by
Tudor Spataru, Lisa Maria Dascalu, Andreea Moraru and Mariana Moraru
Reactions 2024, 5(4), 778-788; https://doi.org/10.3390/reactions5040039 - 14 Oct 2024
Abstract
►▼
Show Figures
The enzymes Cytochrome P450 and Superoxide Reductase, which have a similar coordination center [FeN4S], begin their biochemical cycles similarly. They absorb an oxygen molecule, add two electrons, and link a hydrogen atom to the distal oxygen atom of the product obtained, creating the
[...] Read more.
The enzymes Cytochrome P450 and Superoxide Reductase, which have a similar coordination center [FeN4S], begin their biochemical cycles similarly. They absorb an oxygen molecule, add two electrons, and link a hydrogen atom to the distal oxygen atom of the product obtained, creating the so-called Compound 0 in the case of the first enzyme. However, the bio-catalytic processes of these two enzymes continue in different ways. In the bio-catalytic cycle of Cytochrome P450, the enzyme binds another proton to the distal oxygen atom, producing a water molecule and Compound 1. In contrast, in the bio-catalytic cycle of the Superoxide Reductase, the enzyme binds a proton to the proximal oxygen atom, producing a hydrogen peroxide molecule, which later decomposes into oxygen and water. The MCSCF method in the CASSCF form was used to study the difference in Cytochrome P450 and Superoxide Reductase’s bio-catalytic cycles. The results of these enzymes’ hydroperoxo adduct models’ geometric optimization showed that, in fact, all their properties, including their spin states, the wave functions in their active zones, and the Fe-N, Fe-S, and Fe-O bond lengths, are different. The Fe-N, Fe-S, and Fe-O chemical bond lengths are much longer in the case of the second enzyme compared to the chemical bond lengths in the case of the first enzyme, reflecting a spin value equal to 5/2 in the second case and a spin value equal to 1/2 in the first. A decisive role in the difference in their bio-catalytic cycles is played by the fact that the first bonded hydrogen atom is linked to the distal oxygen atom in the side position in the case of Compound 0 and the up position in the case of the hydroperoxo adduct of the enzyme Superoxide Reductase, protecting the distal oxygen atom from possible interaction with the substrate. The second protonation to Compound 0 at the distal oxygen atom in the case of Cytochrome P450’s bio-catalytic cycle and the second protonation at the proximal oxygen atom in the case of the hydroperoxo adduct of Superoxide Reductase’s bio-catalytic cycle depend on the proton transfer through the Asp251 channel in the first case and on the transferal of H+ from the substrate to the water molecule and the proximal oxygen in the second case.
Full article

Figure 1
Open AccessArticle
Production of Sugars and Ethanol from Acid–Alkaline-Pretreated Agave sisalana Residue
by
Habila Yusuf Thomas, José Demétrio Nery Cavalcante, Willame Marinho dos Santos, Domingos Fabiano de Santana Souza, Carlos Eduardo de Araújo Padilha, Rômulo Simões Cezar Menezes, Rafael Barros de Souza and Emmanuel Damilano Dutra
Reactions 2024, 5(4), 765-777; https://doi.org/10.3390/reactions5040038 - 14 Oct 2024
Abstract
Drylands in Brazil have been exploring sisal (Agave sisalana) as an essential source of income. However, the solid residues generated because of this activity still need suitable destinations; therefore, research has been carried out to transform them into added-value products. Therefore,
[...] Read more.
Drylands in Brazil have been exploring sisal (Agave sisalana) as an essential source of income. However, the solid residues generated because of this activity still need suitable destinations; therefore, research has been carried out to transform them into added-value products. Therefore, the present study evaluated the potential of sisal or agave solid residue as a precursor feedstock for second-generation ethanol production. Acid and acid–alkaline pretreatments were carried out on sisal residues to enrich the biomass with cellulose and maximize enzymatic digestibility. Second-generation ethanol production was carried out using Semi-simultaneous saccharification and fermentation (SSSF). Regardless of catalyst dosage and incubation time, oxalic acid pretreatments generated samples with a similar chemical composition to those pretreated with sulfuric acid. However, samples pretreated with oxalic acid showed lower enzymatic digestibility. Samples pretreated with oxalic acid and sodium hydroxide obtained 14.28 g/L of glucose and cellulose conversion of 79.1% (at 5% solids), while 21.49 g/L glucose and 91.2% of cellulose conversion were obtained in the hydrolysis of pretreated samples with sulfuric acid and sodium hydroxide combined pretreatments. The pretreatment sequence efficiently reduced cellulase dosage from 20 to 10 FPU/g without compromising sugar release. SSSF achieved maximum production of 40 g/L ethanol and 43% ethanol conversion using 30% solids and gradually adding biomass and cellulases.
Full article
(This article belongs to the Special Issue Waste Biorefinery Technologies for Accelerating Sustainable Energy Processes)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Theoretical Study of the Halogen Concentration Effect on the 1,3-Butadiene Polymerization Catalyzed by the Neodymium-Based Ziegler–Natta System
by
Alexey N. Masliy, Ildar G. Akhmetov, Andrey M. Kuznetsov and Ilsiya M. Davletbaeva
Reactions 2024, 5(4), 753-764; https://doi.org/10.3390/reactions5040037 - 7 Oct 2024
Abstract
►▼
Show Figures
In this work, an attempt is made to theoretically substantiate the experimentally known facts of the influence of halogen concentration on the catalytic properties of the neodymium-based Ziegler–Natta system. Based on the structural and thermochemical data obtained using modern methods of quantum chemistry,
[...] Read more.
In this work, an attempt is made to theoretically substantiate the experimentally known facts of the influence of halogen concentration on the catalytic properties of the neodymium-based Ziegler–Natta system. Based on the structural and thermochemical data obtained using modern methods of quantum chemistry, the process of the 1,3-butadiene cis-1,4-polymerization under the model active centers of the neodymium Ziegler–Natta catalysts with different contents of chloride ions was studied. Results are presented that explain the increase in the cis-stereospecificity and activity of the polymerization system with an increase in the content of the chloride ions in the neodymium catalytic system. Reasons were established for the decrease in the concentration of active centers relative to the introduced Nd(III) with an excess of chloride ions and the occurrence of the anti-syn isomerization as a source of the formation of the trans-1,4-structures in the cis-1,4-polybutadiene.
Full article

Figure 1
Open AccessArticle
Overview of the Catalytic Liquefaction of Waste Plastics Process Development, Operation and Product Quality
by
Jumluck Srinakruang, Haruki Tani and Kaoru Fujimoto
Reactions 2024, 5(4), 740-752; https://doi.org/10.3390/reactions5040036 - 4 Oct 2024
Abstract
►▼
Show Figures
Waste plastics were successfully decomposed with a solid acid catalyst to make oil. Spent FCC (Fluid Catalytic Cracking) catalyst was used for this process. The operation of this process was conducted in a horizontal agitated laboratory reactor with an inner volume of 1
[...] Read more.
Waste plastics were successfully decomposed with a solid acid catalyst to make oil. Spent FCC (Fluid Catalytic Cracking) catalyst was used for this process. The operation of this process was conducted in a horizontal agitated laboratory reactor with an inner volume of 1 L with 0.1 kg/h to the capacity of 80 kg/h of the demonstrate plant conducted by continuously feeding a plastic flake or molded cube to the heated powder catalyst bed at around 400–450 °C. The yield of oil was as high as 70 to 85%, depending on the type of plastic. The processing of a variety of waste plastics from home waste, industrial waste and even marine plastics could be processed to obtain oil with a low freezing point and a high heating value. The product was mainly composed of iso-paraffins, olefins and aromatics. The effective in situ dichlorination was attained from the waste plastic containing PVC. A small amount of PET in the plastic was converted to methyl benzene during the cracking operation.
Full article

Figure 1
Open AccessReview
Synthesis, Photocatalytic and Bio Activity of ZnO-TiO2 Nanocomposites: A Review Study
by
Fulvia Pinzari
Reactions 2024, 5(4), 680-739; https://doi.org/10.3390/reactions5040035 - 2 Oct 2024
Abstract
Zinc oxide and titanium dioxide are materials with strong photocatalytic and antimicrobial activity. This activity is greater when the material is in nanocrystalline form. It has been seen that these properties are also present in the ZnO-TiO2 nanocomposite material, and the extent
[...] Read more.
Zinc oxide and titanium dioxide are materials with strong photocatalytic and antimicrobial activity. This activity is greater when the material is in nanocrystalline form. It has been seen that these properties are also present in the ZnO-TiO2 nanocomposite material, and the extent depends on multiple factors, such as crystallinity, structural composition, crystallite size, and morphology. These structural properties can be varied by acting on the synthesis of the material, obtaining a wide variety of composites: random nanoparticles, nanorods, nanowires, nanotubes, nanofibers, tetrapods, core–shell, hollow spheres, inverse opal structures (IOSs), hierarchical structures, and films. When an interface between nanocrystallites of the two oxides is created, the composite system manages to have photocatalytic activity greater than that of the two separate oxides, and in certain circumstances, even greater than P25. The antimicrobial activity results also improved for the composite system compared to the two separate oxides. These two aspects make these materials interesting in various fields, such as wastewater and air treatment, energy devices, solar filters, and pharmaceutical products and in the context of the restoration of monumental cultural assets, in which their use has a preventive purpose in the formation of biofilms. In this review we analyse the synthesis techniques of ZnO-TiO2 nanocomposites, correlating them to the shape obtained, as well as the photocatalytic and antimicrobial activity. It is also illustrated how ZnO-TiO2 nanocomposites can have a less negative impact on toxicity for humans and the environment compared to the more toxic ZnO nanoparticles or ZnO.
Full article
(This article belongs to the Special Issue Nanoparticles: Synthesis, Properties, and Applications)
►▼
Show Figures

Scheme 1
Open AccessArticle
Investigation of Acetoin Biosynthesis by Bacillus subtilis ACA-DC 1176 Growing on Crude Glycerol in Flask and Bioreactor Trials
by
Dimitris Karayannis, Eleni Moraiti, Ourania Kalantzi and Seraphim Papanikolaou
Reactions 2024, 5(4), 664-679; https://doi.org/10.3390/reactions5040034 - 1 Oct 2024
Abstract
Acetoin biosynthesis by two Bacillus subtilis strains valorising crude glycerol was thoroughly explored within a pre-defined range of culture conditions and systems. B. subtilis ACA-DC 1176 stood out for its higher efficiency in acetoin production, prompting an investigation into the potential for enhanced
[...] Read more.
Acetoin biosynthesis by two Bacillus subtilis strains valorising crude glycerol was thoroughly explored within a pre-defined range of culture conditions and systems. B. subtilis ACA-DC 1176 stood out for its higher efficiency in acetoin production, prompting an investigation into the potential for enhanced productivity through the evaluation of diverse culture conditions and media compositions. The primary by-products of the biodiesel and corn industries, namely crude glycerol and corn steep liquor, respectively, were successfully employed as the principal carbon and nitrogen sources of the newly developed low-cost culture medium. Furthermore, the results of the various feeding strategies that were tested indicated that the conversion of 2,3-butanediol (BDO) to acetoin occurred exclusively when the concentration of glycerol was below approximately 5 g/L. This seemed to be necessary for the production of NADH, which is essential for maintaining cellular processes. Following the complete depletion of glycerol, acetic acid increased and became the predominant metabolite, while both acetoin and BDO decreased, presumably resulting in ATP generation. This is likely a mechanism employed by the cell to generate energy in the absence of a carbon source. In the fed-batch bioreactor culture, the kinetics of metabolites differed, as there was no conversion of BDO to acetoin at the final depletion of glycerol. At volumetric mass transfer coefficient (kLa) levels exceeding approximately 70 1/h, the production of acetoin was favoured over that of BDO, with the highest observed acetoin/BDO ratio reaching 4.29 g/g. Conversely, at kLa values below approximately 60 1/h, the titres of acetoin and BDO were found to be nearly equal. The final concentrations of acetoin and BDO reached 36.0 g/L and 25.5 g/L, respectively, resulting in a total yield of both (acetoin + BDO) per glycerol consumption of 0.40 g/g. To the best of our knowledge, this is the first study to focus on acetoin production from crude glycerol fermentative valorisation. The study presents new findings regarding the parameters influencing the level of BDO conversion to acetoin. However, further research is required in order to gain a comprehensive understanding of the underlying phenomena and metabolic pathways involved.
Full article
(This article belongs to the Special Issue Feature Papers in Reactions in 2024)
►▼
Show Figures

Figure 1
Open AccessArticle
Synthesis of Nanostructured Mg2Ni for Hydrogen Storage by Mechanical Alloying via High-Pressure Torsion
by
Edgar Ignacio López Gómez, Joaquín Gonzalez, Jorge M. Cubero-Sesin and Jacques Huot
Reactions 2024, 5(4), 651-663; https://doi.org/10.3390/reactions5040033 - 24 Sep 2024
Abstract
Mg2Ni is a highly promising candidate for solid-state hydrogen storage due to its high storage capacity. However, its synthesis is challenging due to the high melting point of Ni (1455 °C) and the boiling point of Mg (1090 °C). In this
[...] Read more.
Mg2Ni is a highly promising candidate for solid-state hydrogen storage due to its high storage capacity. However, its synthesis is challenging due to the high melting point of Ni (1455 °C) and the boiling point of Mg (1090 °C). In this study, elemental powder mixtures of Mg and 30 at% Ni were processed by high-pressure torsion (HPT) to synthesize the Mg2Ni intermetallic compound through mechanical methods. The formation of 11 wt% of Mg2Ni after 50 turns of HPT was confirmed by X-ray diffraction (XRD), scanning electron microscopy (SEM), and energy-dispersive spectroscopy (EDS), reaching a maximum of 59 wt% after 100 turns. Rietveld refinement confirmed a nanocrystalline size for the Mg2Ni phase synthesized via HPT. Hydrogenation tests showed that the Mg-Ni synthesized by HPT can absorb hydrogen at 350 °C even after several weeks of air exposure. Furthermore, a maximum absorption capacity of 3.8 wt% was reached after 20 h of hydrogen exposure for the sample with 100 turns. This capacity is close to the theoretical capacity of 3.9 wt% for this composition. The results confirm that combining HPT with subsequent heat treatment is an efficient strategy to increase the Mg2Ni fraction after HPT processing.
Full article
(This article belongs to the Special Issue Hydrogen Production and Storage, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
Enhancing Laccase Production by Trametes hirsuta GMA-01 Using Response Surface Methodology and Orange Waste: A Novel Breakthrough in Sugarcane Bagasse Saccharification and Synthetic Dye Decolorization
by
Guilherme Guimarães Ortolan, Alex Graça Contato, Guilherme Mauro Aranha, Jose Carlos Santos Salgado, Robson Carlos Alnoch and Maria de Lourdes Teixeira de Moraes Polizeli
Reactions 2024, 5(3), 635-650; https://doi.org/10.3390/reactions5030032 - 19 Sep 2024
Abstract
Trametes hirsuta GMA-01 was cultivated in a culture medium supplemented with orange waste, starch, wheat bran, yeast extract, and salts. The fungus produced several holoenzymes, but the laccase levels were surprisingly high. Given the highlighted applicability of laccases in various biotechnological areas with
[...] Read more.
Trametes hirsuta GMA-01 was cultivated in a culture medium supplemented with orange waste, starch, wheat bran, yeast extract, and salts. The fungus produced several holoenzymes, but the laccase levels were surprisingly high. Given the highlighted applicability of laccases in various biotechnological areas with minimal environmental impact, we provided a strategy to increase its production using response surface methodology. The immobilization of laccase into ionic supports (CM-cellulose, DEAE-agarose, DEAE-cellulose, DEAE-Sephacel, MANAE-agarose, MANAE-cellulose, and PEI-agarose) was found to be efficient and recuperative, showcasing the technical prowess of research. The crude extract laccase (CE) and CM-cellulose-immobilized crude extract (ICE) showed optimum activity in acidic conditions (pH 3.0) and at 70 °C for the CE and 60 °C for the ICE. The ICE significantly increased thermostability at 60 °C for the crude extract, which retained 21.6% residual activity after 240 min. The CE and ICE were successfully applied to sugarcane bagasse hydrolysis, showing 13.83 ± 0.02 µmol mL−1 reducing sugars after 48 h. Furthermore, the CE was tested for dye decolorization, achieving 96.6%, 71.9%, and 70.8% decolorization for bromocresol green, bromophenol blue, and orcein, respectively (0.05% (w/v) concentration). The properties and versatility of T. hirsuta GMA-01 laccase in different biotechnological purposes are interesting and notable, opening several potential applications and providing valuable insights into the future of biotechnological development.
Full article
(This article belongs to the Special Issue Waste Biorefinery Technologies for Accelerating Sustainable Energy Processes)
►▼
Show Figures

Figure 1
Open AccessArticle
The Influence of the Structure of Organochlorine Compounds on Their Decomposition Process in a Dielectric Barrier Discharge
by
Bogdan Ulejczyk
Reactions 2024, 5(3), 623-634; https://doi.org/10.3390/reactions5030031 - 11 Sep 2024
Abstract
►▼
Show Figures
The decomposition efficiency of C2HCl3 and CHCl3 in a barrier discharge is very different, even though these compounds differ little in chemical composition. In both compounds, there are three chlorine atoms and one hydrogen atom. The difference between them
[...] Read more.
The decomposition efficiency of C2HCl3 and CHCl3 in a barrier discharge is very different, even though these compounds differ little in chemical composition. In both compounds, there are three chlorine atoms and one hydrogen atom. The difference between them is the presence of one carbon atom in CHCl3 and two carbon atoms connected by a double bond in C2HCl3 and the higher polarizability of C2HCl3. The polarizability of C2HCl3 is 10.21 Å3 and that of CHCl3 is 8.39 Å3. As a result of these differences, the C2HCl3 conversion was two to three times higher than the CHCl3 conversion. The main product of CHCl3 decomposition containing chlorine was ClO2, while Cl2, COCl2, HCl, CCl4, and Cl− were formed in smaller amounts. The main products of C2HCl3 decomposition, which contain chlorine, were COCl2, HCl, and Cl−. CCl4 was not formed. Cl2 and ClO2 were formed in smaller amounts. Pathways of C2HCl3 and CHCl3 decomposition are shown in this paper. The process was carried out at low power (0.2–0.8 W) in air. The gas flow was 10 L/h, and the concentration of the decomposed compound was 0.4%. The volume of the gas space of the reactor (plasma zone) was 27 cm3.
Full article

Figure 1
Open AccessArticle
Impact of Inverse Manganese Promotion on Silica-Supported Cobalt Catalysts for Long-Chain Hydrocarbons via Fischer–Tropsch Synthesis
by
Ntebogang Thibanyane, Joshua Gorimbo and Yali Yao
Reactions 2024, 5(3), 607-622; https://doi.org/10.3390/reactions5030030 - 9 Sep 2024
Abstract
One of the challenges in Fischer–Tropsch synthesis (FTS) is the high reduction temperatures, which cause sintering and the formation of silicates. These lead to pore blockages and the coverage of active metals, particularly in conventional catalyst promotion. To address the challenge, this article
[...] Read more.
One of the challenges in Fischer–Tropsch synthesis (FTS) is the high reduction temperatures, which cause sintering and the formation of silicates. These lead to pore blockages and the coverage of active metals, particularly in conventional catalyst promotion. To address the challenge, this article investigates the effects of the preparation method, specifically the inverse promotion of SiO2-supported Co catalysts with manganese (Mn), and their reduction in H2 for FTS. The catalysts were prepared using stepwise incipient wetness impregnation of a cobalt nitrate precursor into a promoted silica support. The properties of the catalysts were characterized using XRD, XPS, TPR, and BET techniques. The structure–performance relationship of the inversely promoted catalysts in FTS was studied using a fixed-bed reactor to obtain the best performing catalysts for heavy hydrocarbons (C5+). XRD and XPS results indicated that Co3O4 is the dominant cobalt phase in oxidized catalysts. It was found that with increase in Mn loading, the reduction temperature increased in the following sequence 10%Co/SiO2 < 10%Co/0.25%Mn-SiO2 < 10%Co/0.5%Mn-SiO2 < 10%Co/3.0%Mn-SiO2. The catalyst with the lowest Mn loading, 10%Co/0.25%Mn-SiO2, exhibited higher C5+ selectivity, which can be attributed to less MSI and higher reducibility. This catalyst showed the lowest CH4 selectivity possibly due to lower H2 uptake and higher CO chemisorption.
Full article
(This article belongs to the Special Issue Fischer-Tropsch Synthesis: Bridging Carbon Sustainability)
►▼
Show Figures

Figure 1
Open AccessArticle
Zirconium Complexes Bearing Methyl/tButyl Salicylate and Their Catalytic Activity on ε-Caprolactone
by
Gamze Uzun and Asgar Kayan
Reactions 2024, 5(3), 594-606; https://doi.org/10.3390/reactions5030029 - 2 Sep 2024
Abstract
►▼
Show Figures
In this study, methyl/tbutyl salicylate-bearing zirconium complexes (C1–C8) were prepared by the reaction of zirconium (IV) propoxide/butoxide with salicylic acid, 3-methylsalicylic acid, 4-methylsalicylic acid, and 3,5-di-tert-butylsalicylic acid in alcohols, respectively. All these complexes (C1
[...] Read more.
In this study, methyl/tbutyl salicylate-bearing zirconium complexes (C1–C8) were prepared by the reaction of zirconium (IV) propoxide/butoxide with salicylic acid, 3-methylsalicylic acid, 4-methylsalicylic acid, and 3,5-di-tert-butylsalicylic acid in alcohols, respectively. All these complexes (C1–C8) were characterized by 1H NMR, 13C NMR, FTIR, mass spectroscopy (MS), elemental, and thermogravimetric analyses (TGA). These complexes were utilized as catalysts in the ring-opening polymerization (ROP) of Ɛ-caprolactone and were very effective. Polycaprolactone (PCL) was characterized by 1H-NMR, 13C NMR, and gel permeation chromatography (GPC). In this study, perhaps for the first time, the effects of electron-donating substituents (Me and tBu) on Ɛ-caprolactone polymerization reactions on salicylate ligands linked to zirconium atoms were investigated.
Full article

Figure 1
Open AccessCommunication
Biodiesel Production from Edible Oil Using Heteropoly Acid Catalysts at Room Temperature
by
Noah L. Fioravante, Guoqiang Cao and Nan Yi
Reactions 2024, 5(3), 587-593; https://doi.org/10.3390/reactions5030028 - 1 Sep 2024
Abstract
►▼
Show Figures
Edible oils are one of the renewable sources that enable the possibility of producing biodiesel sustainably. The transesterification of canola oil with methanol using cesium-modified phosphotungstic acid (Cs2.5H0.5PW12O40) as a heterogeneous catalyst was studied. Reaction
[...] Read more.
Edible oils are one of the renewable sources that enable the possibility of producing biodiesel sustainably. The transesterification of canola oil with methanol using cesium-modified phosphotungstic acid (Cs2.5H0.5PW12O40) as a heterogeneous catalyst was studied. Reaction conditions, specifically reaction time, catalyst loading, and the ratio of methanol to canola oil, were systematically explored. The canola oil conversion reached 55% at room temperature after 24 h. The reusability tests showed that the conversion of canola oil to biodiesel was maintained.
Full article

Graphical abstract
Open AccessArticle
C2-Symmetric Amino Acid Amide-Derived Organocatalysts
by
Zahraa S. Al-Taie, Simon J. Coles, Aileen Congreve, Dylan Ford, Lucy Green, Peter N. Horton, Leigh F. Jones, Pippa Kett, Rolf Kraehenbuehl, Patrick J. Murphy, Graham J. Tizzard, Niles B. Willmore and Oliver T. Wright
Reactions 2024, 5(3), 567-586; https://doi.org/10.3390/reactions5030027 - 24 Aug 2024
Abstract
►▼
Show Figures
N-alkylated C2-symmetric amino acid amide derivatives were shown to catalyse the Michael addition of 2-hydroxy-1,4-napthoquinone to β-nitrostyrene, achieving a maximum ee of 44%. The corresponding trifluoroacetic acid salts also catalysed the aldol reaction between 4-nitrobenzaldehyde and hydroxyacetone, leading to the
[...] Read more.
N-alkylated C2-symmetric amino acid amide derivatives were shown to catalyse the Michael addition of 2-hydroxy-1,4-napthoquinone to β-nitrostyrene, achieving a maximum ee of 44%. The corresponding trifluoroacetic acid salts also catalysed the aldol reaction between 4-nitrobenzaldehyde and hydroxyacetone, leading to the formation of predominantly syn-aldol products in up to 55% ee. Aspects of the solvent dependence of the aldol reaction and the H-bonding of the catalyst were investigated.
Full article

Figure 1
Open AccessReview
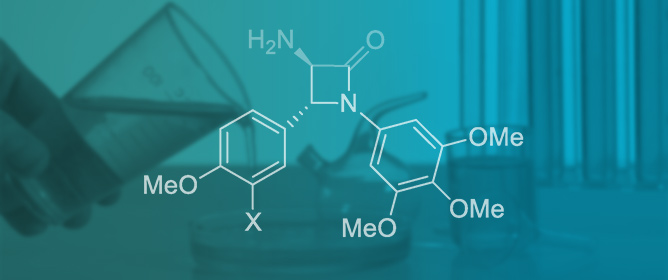
Synthesis of 2-Azetidinones via Cycloaddition Approaches: An Update
by
Franca Maria Cordero, Donatella Giomi and Fabrizio Machetti
Reactions 2024, 5(3), 492-566; https://doi.org/10.3390/reactions5030026 - 16 Aug 2024
Abstract
The present review is a comprehensive update of the synthesis of monocyclic β-lactams via cycloaddition reactions. According to the IUPAC definition of cycloaddition, both elementary and stepwise processes (formal cycloadditions) have been considered. The years 2019–2022 are covered by the cited literature. The
[...] Read more.
The present review is a comprehensive update of the synthesis of monocyclic β-lactams via cycloaddition reactions. According to the IUPAC definition of cycloaddition, both elementary and stepwise processes (formal cycloadditions) have been considered. The years 2019–2022 are covered by the cited literature. The focus of the review is on synthetic aspects with emphasis on the structural scope, reaction conditions, mechanistic aspects, and selectivity results. Selected significant data related to biological activities and synthetic applications are also highlighted.
Full article
(This article belongs to the Special Issue Cycloaddition Reactions at the Beginning of the Third Millennium)
►▼
Show Figures

Figure 1
Open AccessArticle
Autocatalytic Acetylation of Crude Glycerol Using Acetic Acid: A Kinetic Model
by
Federico M. Perez, Francisco Pompeo, Gerardo F. Santori and Martín N. Gatti
Reactions 2024, 5(3), 472-491; https://doi.org/10.3390/reactions5030025 - 9 Aug 2024
Abstract
►▼
Show Figures
The aim of this work was to develop a kinetic model based on the power law to describe the evolution of glycerol conversion and product distribution in the crude glycerol (G) acetylation reaction with acetic acid (AA) without the use of a catalyst.
[...] Read more.
The aim of this work was to develop a kinetic model based on the power law to describe the evolution of glycerol conversion and product distribution in the crude glycerol (G) acetylation reaction with acetic acid (AA) without the use of a catalyst. For this purpose, experimental tests were carried out with analytical glycerol under different reaction conditions (T = 80–160 °C, AA/G = 1–9 molar ratio, t = 0.25–2 h). The results showed the formation of mono- (MAG), di- (DAG) and tri- (TAG) acetylglycerols, liquid products with multiple applications in the chemical industry. From these results, a kinetic model based on the power law was implemented, which could acceptably estimate the experimental concentrations with an average relative error of 14.9%. Then, crude glycerol samples from different biodiesel industries were characterized by identifying and quantifying the impurities present in them (H2O, CH3OH, NaOH, NaCOOH, MONG and NaCl), and employed as reactants in the reaction tests. Given the significant differences observed in the glycerol conversion values compared to those obtained with analytical glycerol, further reaction tests were conducted to elucidate the effect of each impurity over the glycerol conversion. In these tests, the different impurities were added individually, maintaining the same concentration range as that of the crude glycerol samples. From the results obtained, global activity factors were introduced, which allowed us to modify the kinetic model to estimate glycerol conversions in the crude glycerol samples with an average relative error of 7%. It is hoped that this kinetic model will be a powerful tool useful for designing reactors on an industrial scale.
Full article

Figure 1
Open AccessArticle
Using Phosphogypsum as a Source of Calcium Sulfate When Synthesizing Calcium Molybdate Nanoparticles
by
Youssef Belaoufi, Meryem Bensemlali, Badreddine Hatimi, Halima Mortadi, Najoua Labjar, Jean-Michel Nunzi, Mohammed El Idrissi, Abdellatif Aarfane, Mina Bakasse and Hamid Nasrellah
Reactions 2024, 5(3), 462-471; https://doi.org/10.3390/reactions5030024 - 7 Aug 2024
Abstract
Calcium molybdate (CaMoO4) is of significant interest due to its unique properties and numerous industrial applications, such as catalysis, electrochemistry, and optoelectronics. In this study, we developed an economical and environmentally friendly method to synthesize calcium molybdate from Moroccan phosphogypsum (PG)
[...] Read more.
Calcium molybdate (CaMoO4) is of significant interest due to its unique properties and numerous industrial applications, such as catalysis, electrochemistry, and optoelectronics. In this study, we developed an economical and environmentally friendly method to synthesize calcium molybdate from Moroccan phosphogypsum (PG) industrial waste and sodium molybdate, all at room temperature. Comprehensive analysis through X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), Raman vibrational spectroscopy, and scanning electron microscopy (SEM) revealed the high purity of the synthesized calcium molybdate, with particle sizes of only 12 nm. Additionally, optical characteristics were studied using ultraviolet-visible spectroscopy (UV-vis), which showed an optical band gap of Egap = 3.96 eV for CaMoO4. These results confirm the successful synthesis of calcium molybdate nanoparticles from Moroccan phosphogypsum, demonstrating an effective pathway to valorize this industrial waste into a valuable material. This approach contributes to environmental sustainability by reducing dependence on rare chemicals while offering innovative solutions for the industry’s sustainable development.
Full article
(This article belongs to the Special Issue Nanoparticles: Synthesis, Properties, and Applications)
►▼
Show Figures

Figure 1
Open AccessCommunication
A New Nonaqueous Flow Battery with Extended Cycling
by
Diqing Yue, Weilin Zhang, Ivy Zhao, Xiaoting Fang, Yuyue Zhao, Jenny Li, Feng Zhao and Xiaoliang Wei
Reactions 2024, 5(3), 452-461; https://doi.org/10.3390/reactions5030023 - 28 Jul 2024
Abstract
►▼
Show Figures
Nonaqueous flow batteries hold promise given their high cell voltage and energy density, but their performance is often plagued by the crossover of redox compounds. In this study, we used permselective lithium superionic conducting (LiSICON) ceramic membranes to enable reliable long-term use of
[...] Read more.
Nonaqueous flow batteries hold promise given their high cell voltage and energy density, but their performance is often plagued by the crossover of redox compounds. In this study, we used permselective lithium superionic conducting (LiSICON) ceramic membranes to enable reliable long-term use of organic redox molecules in nonaqueous flow cells. With different solvents on each side, enhanced cell voltages were obtained for a flow battery using viologen-based negolyte and TEMPO-based posolyte molecules. The thermoplastic assembly of the LiSICON membrane realized leakless cell sealing, thus overcoming the mechanical brittleness challenge. As a result, stable cycling was achieved in the flow cells, which showed good capacity retention over an extended test time.
Full article

Figure 1
Open AccessReview
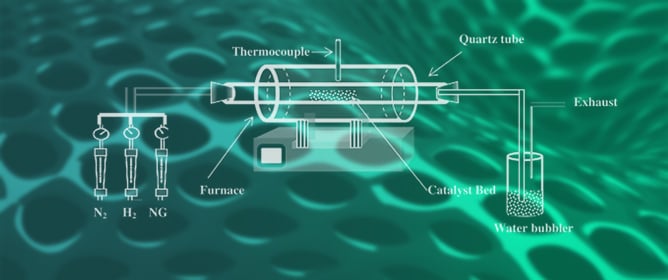
Carbon Nanotubes: A Review of Synthesis Methods and Applications
by
Arash Yahyazadeh, Sonil Nanda and Ajay K. Dalai
Reactions 2024, 5(3), 429-451; https://doi.org/10.3390/reactions5030022 - 4 Jul 2024
Cited by 2
Abstract
Carbon nanotubes (CNTs) are cylindrical-shaped materials composed of hexagonally arranged hybridized carbon atoms with versatility in synthesis methods and diverse applications. This review is focused on the fabrication, physicochemical and spectroscopic characterization, and industrial applications of CNTs. This review discusses some promising synthesis
[...] Read more.
Carbon nanotubes (CNTs) are cylindrical-shaped materials composed of hexagonally arranged hybridized carbon atoms with versatility in synthesis methods and diverse applications. This review is focused on the fabrication, physicochemical and spectroscopic characterization, and industrial applications of CNTs. This review discusses some promising synthesis methods for the preparation of CNTs such as catalytic chemical vapor deposition, arc discharge, and laser ablation. A comparative discussion is made between these synthesis methods in terms of strengths, opportunities and challenges. Furthermore, functionalization and purification of CNTs’ surface leading to improved functionality has also been highlighted in this article. Finally, the analytical techniques employed to shed light on the physicochemical and morphological properties of CNTs are described.
Full article
(This article belongs to the Special Issue Nanoparticles: Synthesis, Properties, and Applications)
►▼
Show Figures

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
Reactions
Cycloaddition Reactions at the Beginning of the Third Millennium
Guest Editors: Donatella Giomi, Alberto Brandi, Fabrizio MachettiDeadline: 30 November 2024
Special Issue in
Reactions
High Temperature Corrosion
Guest Editor: César Augusto Correia de SequeiraDeadline: 31 December 2024
Special Issue in
Reactions
Feature Papers in Reactions in 2024
Guest Editor: Dmitry Yu. MurzinDeadline: 31 December 2024
Special Issue in
Reactions
Electrocatalysis Technologies for Organic Synthesis
Guest Editor: César Augusto Correia de SequeiraDeadline: 31 December 2024