States of matter
- 2. Physical STATES OF MATTER The 5 States of Matter are: ◦ Solid ◦ Liquid ◦ Gas * Plasma * Bose-Einstein Condensates
- 3. States of Matter 1. The molecules of solid are locked in a rigid structure and can only vibrate. (Add thermal energy and the vibrations increase.) Some solids are crystalline, like table salt, in which the atoms are arranged in a repeating pattern. Some solids are amorphous, like glass, in which the atoms have no orderly arrangement. Either way, a solid has definite volume and shape.
- 4. 2. A liquid is virtually incompressible and has definite volume but no definite shape, if you pour a liter of juice into several glasses, the shape of the juice has changed but the total volume hasn’t. 3. A gas is easily compressed. It has neither definite shape nor definite volume, If a container of CO2 is opened, it will diffuse throughout the room.
- 5. *A plasma is an ionized gas and is the most common form of matter in the universe, since the insides of stars are plasmas.
- 6. KINETIC THEORY Also known as “The Kinetic (Particle) Model of Matter” Kinetic Theory states that the tiny particles in all forms of matter are in constant motion. 3 Principles of Kinetic Theory: • All matter is made of tiny particles. • These particles are in constant motion • When particles collide with each other or the container, the collisions are perfectly elastic.
- 7. * BOSE-EINSTEIN CONDENSATE Particles extremely close together Particles barely move Only found at extremely cold temperatures Basically Bose-Einstein is a cold solid Lowest energy of the 5 states/phases of matter
- 9. Fluids The term fluid refers to gases and liquids. Gases and liquids have more in common with each other than they do with solids, since gases and liquids both have particles that are free to move around. They are not locked in place as they are in a solid. The hotter the fluid, the faster its molecules move, and the more space the fluid will occupy. Also, unlike solids, fluids can flow.
- 10. Does Heat Affect Matter?
- 11. Phase Changes Evaporation: Liquid Gas Condensation: Gas Liquid Melting: Solid Liquid Freezing: Liquid Solid *Sublimation: Solid Gas *Examples of sublimation: • Dry ice (frozen CO2) goes directly from the solid to the gaseous state (it sublimates). This creates an eerie. • Comets are very small objects containing frozen gases that sublimate when the comet get close enough to the sun. This creates the characteristic tail the can be millions of miles long.
- 13. Fast Facts about Heat The boiling point is the temperature at which a substance changes from a liquid to a gas. How does the movement of the particles change as it nears the boiling point? The particle move faster and faster until they escape from the liquid.
- 14. Fast Facts about Heat The freezing point is the temperature at which a substance changes from a liquid to a solid. How does this happen? The particles move slower until they are closer together.
- 15. Pressure & Freezing For most liquids the freezing point increases if its pressure is increased, i.e., it’s easier to freeze most liquids if they’re subjected to high pressures. In order to turn a liquids into a solid, the molecules typically must get close enough together to form a crystal. Low temps mean slow moving molecules that are closer together, but high pressure can squeeze the molecules closer together, even if they’re not moving very slowly. Water is an exception to this because, due to its molecular shape, it expands upon freezing. So, squeezing water makes freezing it harder. The pressure on ice due to a passing skater can actually melt a small amount of the ice.
- 16. Pressure & Boiling The lower the pressure on a liquid, the easier it is to make it boil, i.e., as pressure increases, so does the boiling pt. This is because in order for a liquid to boil, molecules need enough kinetic energy to break free from the attraction of the molecules around it. It’s harder for a liquid to vaporize when subjected to high pressure, since gases take up more space than liquids. Water, for example, boils at temps below 100 ºC up in the mountains where the air pressure is lower. It takes longer to cook food in boiling water at high altitudes because the boiling water isn’t as hot. In a vacuum water will boil at any temp, since there is no pressure at the surface to prevent the water from vaporizing. At high pressure water boils at a high temp. In a pressure cooker water can remain liquid up to 120 ºC, and the hotter water can cook food faster.
- 17. Freezing of Solutions The freezing point of a solution, such as salt water, is lower than the freezing point for the solvent by itself, e.g., pure water. The higher the concentration of the solute, e.g. salt, the more the freezing point is lowered. The reason it is more difficult to freeze a liquid when a substance is dissolved in it is because the “foreign” molecules or atoms of a solute interfere with the molecules of the solvent as they’re trying to form a crystalline structure.
- 18. boiling and evaporation Evaporation takes place only at the surface of a liquid or solid, while boiling takes place throughout the body of a liquid. Boiling occurs at the boiling temperature. Particles that have “higher kinetic energy” escape and become vapor (gas).
- 20. Factors affecting the rate of evaporation:
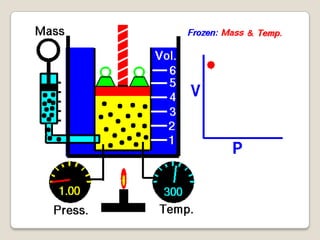
- 21. Boyle’s Law: Robert Boyle in 1662 discovered that “as pressure on a gas was increased, its volume decreased”. Boyle’s Law states that “if the temperature of an ideal gas is held constant, the pressure and volume of a given amount of an ideal gas are inversely proportional” (a given amount of gas means a fixed mass or number of molecules).
- 24. Boyle's Law Formula is expressed symbolically as Another way to express Boyle's Law Formula is or Where: P is pressure of the gas V is volume of the gas k is a constant, and has units of force times distance
- 25. Boyle's Law Problems Question 1: A sample of gaseous nitrogen in a 65.0 L automobile air bag has a pressure of 700 mm Hg. If this sample is transferred to a 25.0 L bag at the same temperature. what is the pressure of the gas in the 25.0 L bag? Question 2: A sample of neon (Ne) occupies 4.00L at a pressure of 5.00 × 104 Pa and a temperature of 273K. Determine the volume of the sample at 100KPa? Answers: (1) P1 = 1820 mm Hg, (2) V2 = 2 L