Unit 1 Biosynthetic Pathways Pharmacognosy and Phytochemistry II.pdf
- 1. 1 Pharmacognosy & Phytochemistry II B. Pharm. Vth Semester Metabolic pathways in higher plants & their determination Unit I Dr. Amit Gangwal HoD, Pharmacognosy I/C: Publicity Wing Criterion 7 I/C, NAAC
- 2. Content has been taken from various sources using Google as search engine. I don’t have any copyright on these content/material/images etc. I am thankful to original creators/right holders. 2
- 3. 3
- 4. 4 CO Code Course Outcomes CO504.1 Understand basic metabolic pathways and formation of different primary & secondary metabolites through different pathways besides learning how these pathways are traced. CO504.2 Explain basics related to chemistry, extraction, classification and various properties pf varied secondary metabolites like alkaloids, glycosides, phenolics and others. CO504.3 Explain basics related to chemistry, extraction, classification and various properties pf varied secondary metabolites like iridoids, resins, volatile oils etc. and explain and perform isolation, identification, analysis of various individual secondary metabolites from plants. CO504.4 To summarize the modern extraction techniques and examine applications of latest techniques in the isolation, purification and identification of crude drugs.
- 6. 6
- 7. 7
- 8. What is Biosynthesis? Process of forming large molecules from smaller subunits within living organism, done by mainly enzymes. Also known as anabolism- simple molecules join to form macromolecules. ◦ E. g. formation of carbohydrates through photosynthesis in chloroplast through conversion of light energy into chemical energy. (synthesis of glucose from H2O+CO2) 8
- 9. 9
- 10. 10 • Gush • Hour • Roguish • Rough • Rush • Shrug • Sigh
- 11. 11
- 12. 12
- 13. 13
- 14. Metabolism What is Metabolism? Metabole: Greek word: Means Change Metabolic pathway: is a series of steps in biochemical reactions that help to convert molecules or substrates like sugar into different usable materials. All these reactions take place inside cell, enzymes which are protein molecules, break down or build up molecules. Enzymes are catalyst to these metabolic reactions. 14
- 15. 15 Anabolism: synthesis of larger/complex molecules – requires energy (Glucose → glycogen) Catabolism: Met. Reac. in cell that degrade substrate into smaller/simpler products- releases energy (Glucose to smaller molecules) Anabolic: Small molecules join into larger ones. Hence Energy is required + Energy Catabolic: Large Molecules are broken down into the smaller one and hence energy is released + Energy
- 16. Metabolic pathways Two types: Anabolic Pathway: Utilization of energy for biosynthetic reactions. Utilizes NADH, ATP etc. Metabolites: intermediates, small mol. products of metabolism Primary metabolites- responsible for primary function for plants’ own growth and development) Catabolic Pathway: release energy by breaking down molecules into simpler molecules. Ex. Cellular respiration: Sugar is taken in by cells and broken down to release energy. 16
- 17. 17
- 18. Metabolic pathways Primary metabolites: carbohydrates, lipids, proteins, amino acids, nucleic acids, cellulose etc. Not responsible for therapeutic activity Primary metabolic pathway: pathways followed for production of primary metabolites Secondary metabolites: not responsible for growth of plants Secondary metabolites: synthesized for adaptation by plants under stress conditions, toxic subs, for attracting pollination, for defense etc. They are therapeutically active. Ex: Alkaloids, glycosides, tannins, terpenoids etc. They are the derivatives of primary metabolites. Synthesis of secondary metabolites: pathway known Secondary metabolic pathway. 18
- 19. Metabolic pathways Primary Metabolic pathways: 1. Glycolysis 2. TCA/ Kreb’s / Citrate pathway/Citric acid cycle (Tricarboxylic Acid Cycle) 3. Pentose phosphate pathway/ Hexose monophosphate pathway Primary metabolic pathway of carbon 1st step is photosynthesis 19 Energy Water CO2 Sugar O2 6CO2 + 6H2O C6H12O6 +6O2 Photosynthesis
- 20. 20
- 21. 21
- 22. 22
- 23. 23
- 24. Metabolic pathways Sugar or glucose enters Glycolysis cycle & finally converted to 2 pyruvate. After glycolysis, pyruvate in presence of O2 (aerobic) is converted to Acetyl CoA. In TCA cycle - Oxidation of Acetyl CoA takes place. 24
- 25. 25 Glycolysis Glucose Hexose Kinase ATP ADP Glucose 6 Phosphate Phospho glucose isomerase Fructose 6 Phosphate Phospho Fructose kinase ATP ADP Fructose 1, 6 Bis Phosphate GLAP DHAP Trios PO4 isomerase 1,3 Bisphospho Glycerate 2 NAD+ + 2Pi 2 NADH 3 Phosphate Glycerate Phospho glycerate kinase 2 ATP 2 ADP 2 Phosphate Glycerate Phospho glycerate mutase Phospho enol pyruvate enolase Pyruvate Pyruvate kinase 2 ATP 2 ADP aldolase GLAP hydrogenase DHAP: di hydroxy acetone phosphate GLAP: Glyceraldehyde 3 Phospahte
- 26. Citric acid Cycle CITRIC ACID CYCLE 26
- 27. Metabolic pathways Sugar or glucose alternatively enters pentose phosphate pathway (HMP) It helps in formation of NADPH from fatty acids, synthesis of ribose for nucleotide & nucleic acid (oxidative pathway) Helps in formation of erythrose 4 phosphate, used in synthesis of aromatic amino acids through shikimic acid pathway. (non oxidative) ↓ precursor for lignin, & other biosynthetic pathways. 27 HMP SHUNT PATHWAY Glucose 6 Phosphate NADP+ NADPH + H+ Mg+2 Glucose 6 P dehydrogenase 6 Phosphoglucanolactone Glucanolactone hydrolase 6 Phosphogluconate NADP+ Mg+2 CO2, NADPH + H+ Phosphogluconate dehydrogenase Ribulose 5 phosphate
- 29. 29
- 30. 30
- 31. SHIKIMIC ACID PATHWAY It is an intermediate from carbohydrate for biosynthesis of phenyl propane derivative (C6-C3 unit) ex. Tyrosine & phenylalanine Precursor for synthesis of shikimic acid is phospho enol pyruvate- (from Glycolysis) & erythrose-4-phosphate an intermediate of PPP/ HMP The N required in pathway is obtained from – other amino acids- Glutamine, glycine, serine. 31
- 32. Role of Shikimic acid Precursor for biosynthesis of some phenolic, phenyl propane derivatives- flavonoid, coumarin, tannins, lignin Precursor for indole, indole derivative & many alkaloids & other aromatic metabolites Gallic acid biosynthesis from 3 de- hydroshikimate (intermediate) 32 In the genesis of aromatic building blocks of lignin & in formation of few tannins, vanillin, phenylpropane units of flavones & coumarins and also in the formation of alkaloids.
- 33. SHIKIMIC ACID PATHWAY 33 Phosphoenol pyruvate (Glycolysis) + erythrose-4-PO4 (PPP) 2-keto, 3-deoxy-7-phospho-D-gluco heptanoic acid DAHP synthase 3-dehydro- quinic acid (DHQ) DHQ synthase 3-dehydro-shikimic acid 3-dehydro quinate dehdratase dehydration Shikimate dehydrogenase Reduction Shikimic acid Shikimic acid-3-PO4 Shikimate kinase Catalyses ATP dep. phosphorylation 5-enol-pyruvylshikimate-3-PO4 Coupling, loss of H2O EPSP synthase Phosphoenol pyruvate Chorismic acid Chorismate synthase Claisen rearrangment Chorismate mutase Prephenic acid p-hydroxy phenyl pyruvate Oxidative decarboxylation Retention of -OH Prephenate dehydrogenase Tyrosine & α- ketoglutarate Transamination Glutamate as N2 source Phenyl pyruvic acid Tryptophan Aldol condensation Anthranilic acid Phenylalanine
- 34. 34
- 35. 35
- 36. 36
- 37. 37
- 39. Acetate Pathway Acetate required for synthesis of various secondary metabolites in plants. Starting material is acetate, utilized as acetyl Co-A (active form of acetate) Various straight chain or aromatic compounds are synthesized by acetate pathway. Acetate Mevalonate Pathway (MAP) contributes to 1/3rd of secondary metabolites. 39
- 40. Acetate Pathway Glycolysis Pyruvate End product Acetyl Co-A TCA Acetate mevalonate pathway Acetate melonate pathway- mevalonic acid Malonyl Co-A Higher terpenoids, rubber etc. Long chain fatty acids & polyketides Isoprenoids Squalene Steroids Lipids, fats , waxes
- 41. 41
- 42. 42 Acetate-Mevalonate Pathway • The role of acetic acid in biogenetic pathways was discovered in 1950 after the discovery of acetyl co -enzyme A ( active acetate ). • Later, it was discovered that mevalonic acid is associated with acetate. • Isopentenyl Pyrophosphate (IPP) and its isomer Dimethylallyl Pyrophosphate (DMAPP) are produced by mevalonic acid. • IPP and DMAPP are the two chief intermediates that set the ‘active isoprene’ unit as the basic building block of isoprenoid compounds. • Both of the units produce geranyl pyrophosphate (C10- monoterpenes), which further associates with IPP to yield farnesyl pyrophosphate (C15-sesquiterpenes).
- 43. Acetate Mevalonate Pathway Isopentenyl pyrophosphate & its isomer that is dimethyl allyl pyrophosphate are universal precursors for synthesis of isoprenoids/terpenoids/volatile oils/cholesterol/saponins/steroids. Isoprene unit (C5H8) - responsible along with other pathways for biogenesis of anthraquinone, naphthaquinone, terpenoids etc. Isoprene units (C5H8) 43 CH3 CH2 C CH CH2 C CH3 CH2 C H CH2 1 2 3 4 5
- 44. Terpenoids are derived from isoprene units (C5H8) which are joined in a head-to-tail or head-to-head fashion. C5 C10 C15 C20 C25 C30 C40 hemiterpenes monoterpenes sesquiterpenes diterpenes sesterterpenes triterpenes tetraterpenes CH3 CH2 C CH CH2
- 45. 45
- 46. 46 Iso Pentenyl Pyrophosphate + Di Methyl Allyl Pyro Phosphate (active isoprene units) CONDENSE Geranyl pyropyrophosphate C10 Monoterpenes e. g. Menthol, limonene, geraniol, linalool, citral Farnesyl PP C15 Sesquiterpenes e.g. termerone, zingiberene, abscisic acid, santalone IPP C30 Triterpenoids Squalene Steroids, cholesterol Solanine, diosgenin IPP Diterpenes Taxol Polyterpenes Rubber C40 Tetraterpenes Carotenoids, lycopene, β carotene
- 48. 48
- 49. 49
- 50. 50
- 51. 51 • Farnesyl pyrophosphate produces geranyl-geranyl pyrophosphate (C20-diterpenes) by combining with another unit of IPP. • Farnesyl pyrophosphate multiplies with its own unit to form squalene and its successive cyclisation produces cyclopentanoperhydrophenanthrene skeleton that contains steroidal compounds (like cholesterol and triterpenoids). • Thus, two different skeleton containing compounds, i.e., steroids and triterpenoids are produced by the acetate mevalonate pathway working through IPP and DMAPP via squalene. • A huge array of monoterpenoids, sesquiterpenoids, diterpenoids, carotenoids, polyprenols, glycosides, and alkaloids are also produced in association with other pathway.
- 52. 52
- 53. 53
- 54. 54 The sapogenins occurs in glycosidic form of saponins. The neutral saponins are steroidal derivatives possessing spiroketal side chain and acid saponins have triterpenoid structure. The pathway is similar for the biosynthesis of sapogenins. The triterpenoid hydrocarbon squalene is formed after cyclisation of triterpenoids in one direction and spiroketal steroids in other direction. The squalene, cholesterol and other steroidal compounds including aglycone are formed in the following manner.
- 55. 55
- 56. 56 Acetate-Malonate Pathway • With the involvement of Acyl Carrier Protein (ACP), the acetate pathway works functionally to produce fatty acyl thioesters of ACP. • These acyl thioesters build the important intermediates in fatty acid synthesis. • In a later stage, even numbers of fatty acids from n - tetraenoic (butyric) to n -eicosanoic (arachidic acid) are produced by these C 2 acetyl CoA units. • Subsequent direct dehydrogenation of saturated fatty acids produces unsaturated fatty acids. • Enzymes play a crucial role in directing the position of newly introduced double bond in the fatty acids.
- 57. 57 • The acetate pathway is important in the formation of various important phytoconstituents like fatty acids, polyketides, prostaglandins, aflatoxin, tetracycline and other various important phytoconstituents. • For the biosynthesis of fatty acid, the acetyl CoA carboxylated to form malonyl CoA by the presence of enzyme named acetyl CoA carboxylase. • The energy requisite for this carboxylation is supplied by ATP and loss of CO2 occurs. • After this step reduction, dehydration and again reduction will occur. • During both reductions process the electron is provided by NADH+ and H+ and the formation of butyryl ALP will occur. • The coupling between malonyl ALP and butyryl ALP will occur and their reduction is repeated again for whole chain. • Malonyl CoA bind again with the fatty acid residue by increase the chain with two carbon unit. • The first end product is palmitic acid which has 16 carbon atoms. • The chain is further elongated by various mechanisms.
- 58. 58
- 59. Amino acid pathway 59 • Plants and bacteria can synthesize all 20 of the amino acids. Whereas humans cannot synthesize 9 of them. • These 9 amino acids must come from our diets and therefore they are called essential amino acids. • The essential amino acids are Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, and Valine. • The 11 amino acids are called non-essential amino acids like Alanine, Arginine, Aspargine, Aspartate, Cysteine, Glutamate, Glutamine, Glycine, Proline, Serine and Tyrosine. • The non-essential amino acids are synthesized by simple pathways, whereas biosynthesis of the essential amino acids are complex. All 3 aromatic amino acids are derived from shikimate pathway.
- 60. 60
- 61. 61
- 62. 62
- 63. 63
- 64. 64
- 65. 65
- 66. 66
- 67. 67
- 68. 68 • The carbon skeletons of amino acids are derived from different intermediates of the central carbon metabolism (boxed in blue). • According to their respective precursors, the amino acids are grouped into five families derived from glutamate, serine, pyruvate, aspartate, or chorismate. • The nine amino acids that cannot be synthesized in animals are shown in dark-green boxes, while those that can be synthesized but additionally need to be taken up with the diet are in brighter boxes. • Proteinogenic amino acids that can be sufficiently synthesized in animals are in pale green boxes and non-proteinogenic amino acids and other important intermediates are boxed in white. DAHP, 3-deoxy- D-arabinoheptulosonate-7-phosphate.
- 69. Biosynthesis of Glycosides 69 • The metabolic process of glycoside formation occurs in two steps: • In first step various types of aglycone are formed by biosynthetic reactions whereas in second step coupling of aglycone with sugar moiety occurs. • In different types of glycosides interaction of nucleotide glycoside occurs between UDP-glucose with alcoholic or phenolic group of secondary compound aglycone (called O-glycosides), through linkage with carbon (C- glycosides), nitrogen (N-glycosides) or sulphur (S- glycosides).
- 70. 70 The following two steps are involved in this process: 1. In first step, the uridine triphosphate (UTP) transferred an uridylyl group to sugar-1-phosphate and forms UDP-sugar and inorganic pyrophosphate. The enzyme which catalyzes this reaction is uridylyl transferases. UTP + Sugar-1-PO4 UDP- sugar + PP1 2. In second step, transfer of the sugar moiety from UDP to a suitable acceptor (aglycone) occurs. This reaction is mediated by enzyme glycosyl transferases and forms glycoside. UDP-sugar + Acceptor (aglycone) Acceptor-sugar (glycoside) + UDP
- 71. 71 • The sugars of glycosides are monosaccharides (like Rhamnose, Glucose, Frucose or deoxy sugars i.e. digitoxose or cymarose). • The aglycone moieties of cardiac glycosides are steroidal in nature. • These are derivatives of cyclopentenophenanthrene ring which contains unsaturated lactone ring attached with C17, a 14-alpha hydroxyl group and a cisjucture of ring C and D. • The anthraquinone glycosides are biosynthesized from shikimic acid pathway in Rubiaceae family. • The alizarin biosynthesis shows ring A is derived from shikimic acid whereas mevalonic acid is included in ring C.
- 72. 72 It is very important to understand the biosynthesis of flavonoids due to their diversity. These flavonoidal molecules are biosynthesized by their precursor which is three molecules of acetic acid and phenyl propane moiety. It mainly involves the interaction of five different pathways which are named as: 1. The Glycolytic pathway. 2. The Pentose phosphate pathway. 3. The Shikimate pathway that synthesizes phenylalanine (An amino acid). 4. The phenylpropanoid metabolism that produces activated cinnamic acid derivatives i.e. 4-coumaroyl-CoA and lignin (also the plant structural component). 5. The diverse specific flavonoidal pathway.
- 73. 73 • The flavonoids are biosynthesized via condensation of the shikimic acid and acylpolymalonate pathways. • The phenyl propane (cinnamic acid derivative) synthesized from shikimic acid which acts as a precursor in a polyketide synthesis. • In this scheme additional three acetate residues are incorporated into the structure and followed by ring closure. • The plants biosynthesizes different classes of flavonoids like flavonols, flavanones, flavones, flavanols or catechins, iso-flavones, dihydro-flavonols, anthocyanidins, and chalcones through subsequent hydroxylation and reduction.
- 74. 74
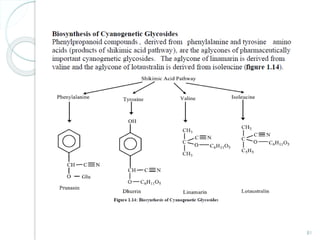
- 75. 75 • In the biosynthesis of Cyanogenetic or Cyanophoric glycosides (e.g. Prunasin) the amino acid phenylalanine acts as a precursor. • In this biosynthetic pathway an aldoxime, a nitrile and a cyanohydrin are involved as intermediate. • The chiral centre in the mandelonitrile provides the opportunity for 2 β–glucosides to form. • D-mandelonitrile glucoside is formed in Prunus serotina (wild cherry) whereas L-mandelonitrile glucoside (isomeric samburgrin) is found in Sambucus nigra. • These compounds are not found in same species.
- 76. 76
- 77. 77
- 78. 78
- 79. 79
- 80. 80
- 81. 81
- 82. 82
- 83. Alkaloids 83
- 84. 84
- 85. 85
- 86. 86
- 87. 87
- 88. 88 Summary
- 89. 89 What do you understand by study of basic metabolic pathways and formation of different secondary metabolites? 2. How secondary metabolites are produced from biosynthetic pathways? 3. What is Shikimic acid pathway? Draw its pathway. 4. How glycosides are biosynthesized? Draw its pathway. 5. Draw biosynthetic pathway of Flavonoids. 6. Draw biosynthetic pathway of Isothiocyanate aglycones. 7. Draw biosynthetic pathway of Cyanogenetic glycosides. 8. How secondary metabolites are obtained by Cholesterol metabolism? 9. Draw Acetate pathway. 10. Draw Amino acid pathway. 11. Draw general scheme for amino acid production through pathway. 12. Write a detailed note on utilization of radioactive isotopes in the investigation of biogenetic studies. 13. What is the role of radioactive tracers? How they are detected? 14. What is autoradiography? Explain precursor product sequence. 15. What is competitive feeding? How precursors are administered in any pathway? 16. What is sequential analysis? Describe briefly.
- 90. Thanks 90