Metals
- 1. * Grade 10 IGCSE Chem
- 2. * Properties of metals * Distinguish between metals and non-metals by * Deduce an order of reactivity from a given set their general physical and chemical properties. of experimental results. * Identify and interpret diagrams that represent Extraction of metals the structure of an alloy. * Describe the use of carbon in the extraction of * Explain why metals are often used in the form of some metals from their ores. alloys. * Describe the essential reactions in the Reactivity series extraction of iron in the blast furnace. * Place in order of reactivity: potassium, sodium, * Relate the method of extraction of a metal calcium, magnesium, zinc, iron, hydrogen and from its ore to its position in the reactivity copper, by reference to the reactions, if any, of series. the elements with Uses of metals * water or steam, * Explain the use of aluminium in aircraft * dilute hydrochloric acid (except for alkali manufacture in terms of the properties of the metals). metal and alloys made from it. * Compare the reactivity series to the tendency of * Explain the use of zinc for galvanising steel, a metal to form its positive ion, illustrated by its and for sacrificial protection. reaction, if any, with: * Explain the use of aluminium in food containers * the aqueous ions of other listed metals, because of its resistance to corrosion. * the oxides of the other listed metals.
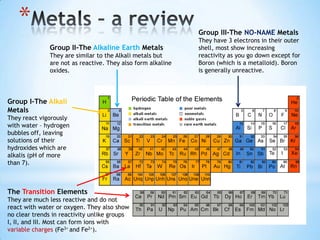
- 3. * Group III-The NO-NAME Metals They have 3 electrons in their outer Group II-The Alkaline Earth Metals shell, most show increasing They are similar to the Alkali metals but reactivity as you go down except for are not as reactive. They also form alkaline Boron (which is a metalloid). Boron oxides. is generally unreactive. Group I-The Alkali Metals They react vigorously with water – hydrogen bubbles off, leaving solutions of their hydroxides which are alkalis (pH of more than 7). The Transition Elements They are much less reactive and do not react with water or oxygen. They also show no clear trends in reactivity unlike groups I, II, and III. Most can form ions with variable charges (Fe3+ and Fe2+).
- 4. * Which of these properties belong to metals? shiny low density weak malleable sonorous high melting and ductile conductor boiling points brittle dull insulator strong low melting and boiling thin high density points Remember metals are on the LEFT side. Over 80% of the elements are metals
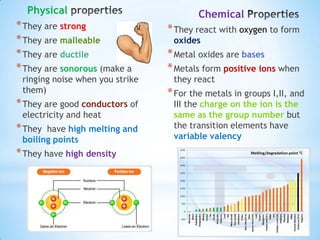
- 5. Physical Chemical * They are strong * They react with oxygen to form * They are malleable oxides * They are ductile * Metal oxides are bases * They are sonorous (make a * Metals form positive ions when ringing noise when you strike they react them) * For the metals in groups I,II, and * They are good conductors of III the charge on the ion is the electricity and heat same as the group number but * They have high melting and the transition elements have boiling points variable valency * They have high density
- 6. * An alloy is a mixture of metals that has different (improved) properties from the metal elements that make it. The pure metal is usually too soft to be useful. Often an alloy is composed mainly of one metal (the parent metal) with small amounts of other metals added. The other metals replace positions of the parent metal in the metal structure, or sometimes fit into the spaces in the metal structure. The red balls are the parent metal. The blue and yellow balls are other metals. The blue lines represent free electrons. The other metals in the structure can change the properties of the alloy by preventing the metal ions from sliding over each other. This can make the alloy tougher and stronger than the parent metal.
- 7. Which one of these diagrams represents a pure metal? Diagram b is a pure metal – the atoms are arranged in a regular lattice. When pressure is applied (by hammering the metal) the layers can slide over each other – this is why metals are malleable and ductile. But when the metal is an alloy (diagrams a, c, and d) new atoms enter the lattice and the layers can no longer slide easily. So the alloy is stronger than the original metal.
- 8. Some examples of alloys: (remember this is a small list- there are thousands of them Alloy Made from Special properties Uses 70% Cu Harder than copper, Musical instruments, ornaments, Brass 30% Zn does not corrode door knobs, etc… 90.25% Al Aluminium 6% Zn Light but very strong, Aircraft alloy 2.5% Mg does not corrode 1.25% Cu Stainless Cookware, cutlery, hardware, steel (there are 80% Fe Strong, does not surgical instruments, major many types of 12% Cr corrode appliances, industrial equipment, stainless steel – 8% Ni this is one) and construction A very old video from 1938 about alloys- start at 3:40
- 9. * What does reactive mean? A reactive element has a strong drive to become a compound, so that its atoms GAIN stable outer shells. So the metal reacts readily with other elements and compounds. Potassium (K) Please Sodium (Na) Send How did we determine which metal is Cats Calcium (Ca) more reactive than another? R Monkeys Magnesium (Mg) Well- by using experimental Aluminium (Al) E And observations of course! (Carbon) A Coloured Zinc (Zn) C Zebras The elements that are bolded are Iron (Fe) T In the ones that YOU will have to place Lead (Pb) I Large in order of reactivity by reference to (Hydrogen) V Hired the reactions with water, steam, or Copper (Cu) I Cages dilute hydrochloric acid. Mercury (Hg) T Make Silver (Ag) Y Sure Gold (Au) Guarded Platinum (Pt) Properly
- 10. * WATER Some metals react chemically with cold water metal + water → metal hydroxide + hydrogen Metal hydroxide is Hydrogen gas is formed liberated Example: Potassium with cold water YOU WRITE THE EQUATION 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g) This is typically the apparatus that is used
- 11. This is a violent reaction! The 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g) hydrogen gas produced usually explodes because of the heat released! Aqueous potassium hydroxide is produced – it is an alkaline solution. The test for an alkaline solution: Red litmus paper will turn blue in the solution The test for hydrogen gas: A lighted splint goes off with a “pop” sound
- 12. Reaction of magnesium with cold water Reaction is much slower! YOU WRITE THE EQUATION 2Mg(s) + 2H2O(l) → 2MgOH(aq) + H2(g) Reaction of zinc with cold water YOU WRITE THE EQUATION but does it happen?? Zn(s) + H2O(l) → No reaction
- 13. Reactions of metals with cold water Metal Observations/Equations • Reacts very violently; explodes with cold water • Enough heat is produced to ignite the hydrogen Potassium gas produced • Hydrogen gas burns in air 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g) • Reacts violently Sodium • Hydrogen gas formed may catch fire and explode 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) Reactivity of metals • Reacts readily increase Calcium • Hydrogen gas formed 2Ca(s) + 2H2O(l) → Ca(OH)2(aq) + H2(g) up the series • Reacts very slowly with cold water Magnesium • A few bubbles of hydrogen gas produced only Zinc Iron Lead • No reaction occurs Copper Silver
- 14. * STEAM Some metals react chemically with hot water – aka STEAM metal + steam → metal oxide + hydrogen Metal oxide is formed Hydrogen gas is liberated Metals like zinc and iron do not react with cold water but do react with steam Example: Magnesium with steam YOU WRITE THE EQUATION This is typically the Mg(s) + H2O(g) → MgO(s) + H2(g) apparatus that is used
- 15. Reaction Reaction of metals with steam is Mg(s) + H2O(g) → MgO(s) + H2(g) vigorous! Metal Observations/Equations • Reacts violently with steam • Magnesium oxide (white power) and hydrogen Magnesium produced • Bright white glow produced Mg(s) + H2O(g) → MgO(s) + H2(g) • Burns in steam • Zinc oxide (yellow when hot and white when cold) Zinc and hydrogen produced Zn(s) + H2O(g) → ZnO(s) + H2(g) • Reacts slowly with steam • Iron(IV)oxide and hydrogen produced Reactivity Iron • Reaction is easily reversible; so iron needs to be of metals heated constantly for reaction to take place increase 2Fe(s) + 3H2O(g) → Fe2O3(s) + 3H2(g) up the Lead series Copper • No reaction occurs Silver
- 16. * dilute acid Some metals react chemically with dilute hydrochloric acid (HCl) metal + dilute acid → salt + hydrogen Metal chloride is formed (salt) Hydrogen gas is liberated You are given 3 unknown metals X, Y and Z and each is added to a test tube filled with 1/3 dilute hydrochloric acid. What do you observe? 1. Arrange the reactivity of the metals (starting with the most reactive) with reference to your observations. 2. Using the following table predict a suitable identity for metals X, Y, and Z
- 17. Reactions of metals with dilute hydrochloric acid Metal Observations/Equations • Explosive reaction- should not be carried out in Potassium school lab. Sodium 2K(s) + 2HCl(aq) → 2KCl(aq) + H2(g) 2Na(s) + 2HCl(aq) → 2NaCl(aq) + H2(g) • Reacts violently to give hydrogen and calcium Calcium chloride 2Ca(s) + 2HCl(aq) → 2CaCl2(aq) + H2(g) • Reacts rapidly to give hydrogen and magnesium Reactivity Magnesium chloride of metals 2Mg(s) + 2HCl(aq) → 2MgCl2(aq) + H2(g) increase • Reacts moderately fast to give hydrogen and zinc up the Zinc chloride series 2Zn(s) + 2HCl(aq) → 2ZnCl2(aq) + H2(g) • Reacts very slowly to give hydrogen and iron(II) Iron chloride – and only if acid is concentrated Fe(s) + 2HCl(aq) → FeCl2(aq) + H2(g) Lead Copper • No reaction occurs Silver
- 18. You are given 3 unknown metals X, Y and Z and each is added to a test tube filled with 1/3 dilute hydrochloric acid. What do you observe? 1. Arrange the reactivity of the metals (starting with the most reactive) with reference to your observations. 2. Predict a suitable identity for metals X, Y, and Z How do you know the gas produced is hydrogen? A lighted splint goes “pop” Magnesium Copper Iron
- 19. Summary of observations of the reactivity of the metals from experimental observations Remember the ones Reaction with in bold you have to Reaction with dilute Metal water or know! acids steam Potassium Explosive reaction Reactive React with Sodium metals Calcium cold water Violent reaction Fairly Magnesium Moderately fast reaction React with reactive Zinc steam Slow reaction metals Iron Lead No reaction Unreactive Copper No reaction with either Silver metals Displacement of hydrogen is a REDOX • Hydrogen gas is produced in all three reactions reaction! H+ gained e- • Reactive metals tend to react with non-metals to form compounds • Unreactive metals have a greater tendency to remain uncombined
- 20. * carbon • The reaction with metal oxides and carbon can be used to compare the reactivity of metals. In other words when metals compete with each other and with carbon to form a compound what will happen? • The more reactive a metal is the more difficult it is to split its oxide up. We can compare these two reactions: Magnesium oxide was not reduced MgO(s) + C(s) → no reaction 2CuO(s) + C(s) → 2Cu(s) + CO2(g) Copper oxide reduced to copper Can be tested using limewater Magnesium being a more reactive metal than copper in the reactivity series did not decompose when heated K > Na > Ca > Mg > Zn > Fe > Pb > Cu > Ag
- 21. Why is this important? • Metals below aluminum in the reactivity series can be reduced by carbon • Metals can be extracted from their ores by reduction with carbon • This includes metals from zinc to gold in the reactivity series • Oxides of metals above zinc in the reactivity series Potassium (K) cannot be reduced by carbon Sodium (Na) • These metal oxides are very stable Calcium (Ca) • They can be reduced by electrolysis Magnesium (Mg) Aluminium (Al) (Carbon) Zinc (Zn) Iron (Fe) Lead (Pb) (Hydrogen) Copper (Cu) Mercury (Hg) This is typically Silver (Ag) the apparatus Gold (Au) that is used Platinum (Pt)
- 22. * • The reaction with metals, a metal oxide, and oxygen can also be used to compare the reactivity of metals. In other words when metals compete with each other and with oxygen to form a compound what will happen? • A metal will reduce the oxide of a less reactive metal. Powdered iron is heated with copper(II)oxide. Can the iron grab the oxygen from the copper(II)oxide? This reaction gives out heat and Fe(s) + CuO(s) → FeO(s) + Cu(s) starts to glow. The iron has “won”. Iron removes the oxygen from the oxide of the metal below it. It is a more reactive metal than copper in the reactivity series. K > Na > Ca > Mg > Zn > Fe > Pb > Cu > Ag
- 23. * Reactivity of a metal depends on the ease with which it forms metal ions. Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s) Zinc “wins” against copper. It displaces the copper from the copper(II)sulfate solution All of these are REDOX reactions – we can also call them SINGLE DISPLACEMENT reactions because zinc and copper displaced each other. A metal that is higher up in the reactivity series has a higher tendency to form its positive ion. K > Na > Ca > Mg > Zn > Fe > Pb > Cu > Ag
- 24. * Remember the ones in bold you have to MOST REACTIVE Potassium (K) know! Sodium (Na) Calcium (Ca) Magnesium (Mg) Metals above blue line: carbon Aluminium (Al) can’t reduce their oxides (Carbon) Zinc (Zn) Iron (Fe) Metals above red line: they displace Lead (Pb) hydrogen from acids LEAST REACTIVE (Hydrogen) Copper (Cu) Mercury (Hg) Silver (Ag) Non-metals carbon and Gold (Au) hydrogen are included Platinum (Pt) here for reference only!
- 25. * 1. The reactivity series is a list of the metals in order of their drive to form positive ions (therefore a stable outer shell). The more easily K > Na > Ca > Mg > Zn > Fe > Pb > Cu > Ag its atoms give up electrons, the more reactive the metal is. 2. A metal will react with a compound of a less reactive metal (an oxide, an aqueous salt) by pushing the less reactive metal out of the compound and taking it’s place. 3. The more reactive the metal, the more stable its compounds are. They do not break down. 4. The more reactive the metal, the more difficult it is to extract from its ores (they are stable). For the most reactive metals you need the toughest method of extraction – electrolysis! 5. The less reactive the metal, the less it likes to form compounds. This is why copper, silver, and gold are found as elements and not potassium, sodium, or calcium. These elements are always found as compounds.
- 26. * How do you extract a metal from it’s ore? * The most unreactive metals (Ag and Au) occur in their ores as elements. All you need to do is separate the Potassium (K) metal from sand and other impurities Sodium (Na) – it does NOT involve chemical Calcium (Ca) reactions. Magnesium (Mg) electrolysis Aluminium (Al) * The ores of all other metals are (Carbon) compounds - they have to be reduced Zinc (Zn) Heating with reducing to get the metal. Iron (Fe) agent – ex Carbon * The compounds of more reactive Lead (Pb) metals are very stable so electrolysis Silver (Ag) Occur naturally as Gold (Au) is needed. elements – no Platinum (Pt) chemical reaction * The compounds of less reactive needed metals are less stable so a reducing Which method is agent can be used. more expensive?
- 27. * The blast furnace! The charge contains three things: A mixture called the charge, containing Iron iron ore, is added through the top of the ore, limestone, and furnace. coke Hot air is added through the bottom. Stage 1: The coke burns, giving off heat Iron forms and trickles down Stage 2: Carbon monoxide is made Carbon monoxide forms and rises Stage 3: The iron(III) oxide is reduced Carbon dioxide After a series of reactions, liquid iron forms and rises collects at the bottom of the furnace.
- 28. * Metal Used for… Properties that make it suitable Overhead electricity A good conductor of electricity (not as good cables (with a steel as copper, but cheaper and much lighter); core for strength) ductile, resists corrosion Aluminium Cooking foil and food Non-toxic, resistant to corrosion, can be cartons rolled into thin sheets Drink cans Light, non-toxic, resistant to corrosion Can be deposited as a thin film, shiny surface Coating CDs and DVDs reflects a laser beam Protecting steel from Offers sacrificial protection to the iron in Zinc rusting steel Coating or galvanising Resists corrosion, but offers sacrificial iron and steel protection if coating cracks For torch (flashlight) Gives a current when connected to a carbon batteries pole, packed into a paste of electrolyte
- 29. * Galvanization is the process of applying a protective zinc coating to steel or iron, in order to prevent corrosion (rusting). Sacrificial zinc anodes are used to protect steel tanks, rails and ship hulls from rusting. Since zinc oxidizes more easily than iron (it is higher in the reactivity series), it corrodes first. When the anode is mostly consumed, it can simply be replaced. Any time you have two different metals that are physically or electrically connected and immersed in seawater, they become a battery. The electrons that make up that current are supplied by one of the metals giving up bits of itself - in the form of metal ions - to the seawater. This is called galvanic corrosion and, left unchecked, it quickly destroys underwater metals. The way to counteract galvanic corrosion is to add a third metal into the circuit, one that is quicker than the other two to give up its electrons. This piece of metal is called a sacrificial anode, and most often it is zinc.
- 30. * Aluminum is one of the most widely used metals in making planes. Why? -high strength/weight ratio -corrosion-resisting qualities -comparative ease of fabrication -nonmagnetic -an excellent conductor of electricity Aluminium-magnesium alloys are both lighter than other aluminium alloys and much less flammable than alloys that contain a very high percentage of magnesium. Check this blog out about Aluminium alloys in planes: http://blog.aluminium- messe.com/aluminium-remains-the-number- one-material-in-aircraft-construction/182/