3.2 Solubility
- 1. Chemistry Chapter 3 - Section 2 Solubility
- 2. Water – The Universal Solvent •Many solutions have water as the solvent. •These solutions are called an aqueous solution. •Water dissolves many solutes and so scientists call it the universal solvent. •Why is water such a great solvent? Let’s find out.
- 3. Dissolving an Ionic Compound
- 4. Dissolving an Ionic Compound example: NaCl
- 5. Dissolving an Ionic Compound example: NaCl
- 6. The positive area of the water molecules surround the negative Chloride ions. 6
- 7. The negative area of the water molecules surround the positive sodium ions.
- 8. As the attractions from the water molecules and their motion pulls the ions apart, the sodium chloride crystal dissolves.
- 9. Dissolving a Molecular Compound
- 10. Dissolving a Molecular Compound example: sugar
- 11. Water molecules arrange themselves around the sucrose molecules according to opposite polar areas. 11
- 12. The attraction of the water molecules and their motion overcome the attraction between sucrose molecules. 12
- 13. The sucrose molecules dissolve as they are separated from the other molecules and mix into the water. 13
- 14. The sucrose molecules dissolve as they are separated from the other molecules and mix into the water.
- 15. What will dissolve? The spoon or the sugar??
- 16. Like Dissolves Like •Polar solvents will dissolve polar solutes. •Nonpolar solvents will dissolve nonpolar solutes. •Polar will not dissolves nonpolar and vice versa. •Think about oil and water. Is the oil polar or nonpolar?
- 17. How Much Will Dissolve?
- 18. How Much Will Dissolve? •If you tried to dissolve a lot of sugar in a small glass of water, not all of the sugar would dissolve. •Solubility is the measurement of how much solute dissolves in a given amount of solvent. •Solubility is usually describes as how much can dissolve in 100g of solvent at a given temperature.
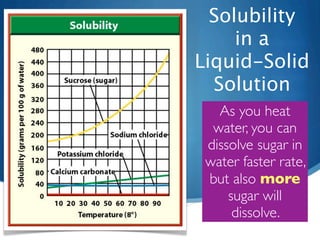
- 19. Solubility in a Liquid-Solid Solution Temperature is included here because the solubility of a solute changes as the temperature of the solvent changes.
- 20. Solubility in a Liquid-Solid Solution As you heat water, you can dissolve sugar in water faster rate, but also more sugar will dissolve.
- 21. Solubility in a Liquid-Gas Solution In a liquid-gas solution, the opposite is true. As the temperature increases, the solubility decreases.
- 22. Solubility in a Liquid-Gas Solution This is because the water molecules will be moving faster at higher temperatures. This movement will bump the gas out of the solution, therefore decreases solubility.
- 23. Saturated Solutions •If you add calcium carbonate to 100g of water at 25˚C, only 0.0014g will dissolve. No more can dissolve. •This solution is called a saturated solution. •If the solution was a liquid- solid solution, the extra solid would settle on the bottom of the container.
- 24. Saturated Solutions •The solubility of sugar in 100g of water at 25˚C is 204g. •If you had a solution with 50g of sugar in 100g of water, the solution is called unsaturated. More solute could dissolve.
- 25. Saturated Solutions •A hot solvent can usually hold more solute then a cooler one. •If a saturated solution cools, some of the solute can fall out of the solution.
- 27. Saturated Solutions •A hot solvent can usually hold more solute then a cooler one. •If a saturated solution cools, some of the solute can fall out of the solution. • But if the solution is cooled slowly, sometimes the excess solute remains dissolved for a period of time. This solution is called supersaturated.
- 28. Rate of Dissolving •Solubility does not tell you how fast a solute will dissolve. •Some solutes will dissolve quickly, others take a long time to dissolve. •A solution dissolves faster when: ✴it is stirred ✴it is shaken ✴the temperature is increased •How exactly do these factors help? ✴They increase the rate at which the surfaces of the solute come into contact with the solvent. ✴Increasing the area of contact between the solute and the solvent can also increase the rate of dissolving
- 29. Concentrated! •The concentration of a solution tells you how much solute is present compared to the amount of solvent. •When you are comparing the concentrations of two solutions with the same type of solute and solvent, you can use a simple description such as: dilute or concentrated.
- 30. Concentrated! •The Dead Sea is 5+ times saltier than Earth's oceans. As water evaporates, salt is left behind. When the saturation point is reached, the salt forms these pillars.
- 31. Measuring Concentrations •One way to give the concentration is by using the percentage of the volume of the solution that is made up of the solute.
- 32. Effects on Solute Particles •Solute particles affect the physical properties of the solvent, such as the boiling point or the freezing point.
- 33. Effects on Solute Particles •Adding a solute, such as NaCl, changes the way the water molecules can arrange themselves. The solute gets in the way.
- 34. Effects on Solute Particles •To overcome this interference of the solute, a lower temperature is needed for the solvent to freeze.
- 35. Effects on Solute Particles •When a solvent begins to boil, the solvent molecules are gaining enough energy to move from the liquid state to the gaseous state.
- 36. Effects on Solute Particles •When a solute is introduced, its particles interfere with the evaporation of the solvent particles.
- 37. Effects on Solute Particles •For water and NaCl, the NaCl will block the H2O from escaping the liquid and becoming a gas.
- 38. Effects on Solute Particles •More energy is needed for the solvent particles to escape from the liquid, and the boiling point of the solution will be higher.
Editor's Notes
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n