Csr Abbott Usa 09
- 1. Redefining Responsibility 2009 Global Citizenship Report
- 2. About this Report Our Promise Welcome to Abbott’s 2009 Global Citizenship Summary We are here for the people we serve in their pursuit of healthy Report. This is an overview of Abbott’s citizenship lives. This has been the way of Abbott for more than a century – strategy, actions and performance in 2009. Our Web site, passionately and thoughtfully translating science into lasting www.abbott.com/citizenship, is the primary means of contributions to health. Our products encircle life, from newborns communication for our citizenship initiatives and houses to aging adults, from nutrition and diagnostics through medical our complete report. Our reporting is intended for a range care and pharmaceutical therapy. Caring is central to the work of audiences, including employees, investors, customers, we do and defines our responsibility to those we serve: suppliers, governments and regulators, and key opinion We advance leading-edge science and technologies that formers. We have referred to the Global Reporting Initiative hold the potential for significant improvements to health and to G3 Guidelines in compiling our report, and an index is the practice of health care. available on our site. We value our diversity – that of our products, technologies, markets and people – and believe that diverse perspectives About the Cover combined with shared goals inspire new ideas and better ways Abbott and Boston-based Partners In Health, a leading of addressing changing health needs. nonprofit organization, have worked together to address We focus on exceptional performance worldwide – health care and social challenges in Haiti for several years. demanding of ourselves and each other because our work Abbott nutrition scientists and engineers are working with impacts people’s lives. Haitian farmers and health care workers like those shown on We strive to earn the trust of those we serve by committing the cover to help create locally produced, sustainable sources to the highest standards of quality, excellence in personal of nutritious, affordable food products. This partnership is relationships and behavior characterized by honesty, fairness designed to support good health while creating jobs and and integrity. stimulating Haiti’s economy. For more information about We sustain success for our business and the people we Abbott’s work to help rebuild Haiti, turn to page 36. serve by staying true to key tenets upon which our company was founded over a century ago: innovative care and a desire to make a meaningful difference in all that we do. The promise of our company is in the promise that our work holds for health and life. Table of Contents 2 Message from the Chairman and CEO Performance and Operational Review 4 Our Business 30 Ethics and Compliance 6 Innovation to Impact: The Similac Story 31 Our People 8 Citizenship at Abbott 34 Supply Chain Management 9 Stakeholder Engagement 35 Global Community 36 Disaster Relief and Rebuilding in Haiti Our Strategic Priorities 37 Business Profile: Diagnostics 12 Innovating for the Future 40 External Commentary: Jane Nelson 16 Enhancing Access 41 Key Metrics Summary 20 Protecting Patients and Consumers 24 Safeguarding the Environment
- 3. 2009 GLOBAL CITIZENSHIP REPORT Abbott is redefining the concept of responsibility. Beyond philanthropy, we apply our science, expertise and technology to address critical health care needs through innovative collaborations and partnerships. We are working to build sustainable solutions to the world’s most pressing health care challenges, while reducing our impact on the environment – creating lasting value for both current and future stakeholders.
- 4. 2009 GLOBAL CITIZENSHIP REPORT To Our Stakeholders Welcome to Abbott’s 2009 In short, these are times that test a company’s commitment Global Citizenship Report to citizenship and operating responsibly and sustainably. We We appreciate your interest in our efforts to live out our believe that Abbott has met that challenge, across the spectrum company’s citizenship commitments while decreasing its impacts of its business activities. 2009 was another successful year on our planet – work that has never been more important for our company, with healthy growth across our range of than it is today. The environments in which we operate – businesses and around the world. This was due to both our natural, social, economic and political – are particularly sensitive continued introduction of advanced new products to improve and challenging, requiring companies like Abbott to be more health, and to our ongoing expansion in international markets. thoughtful than ever about the ways we operate our business. Approximately 54 percent of our sales in 2009 were outside 2009 was a year of extraordinary challenge on a number the United States, our highest percentage ever. We expect to of fronts, raising pressures on business and expectations for continue in this direction, and we took significant steps last year its contribution. First and foremost, of course, was the global to strengthen our position for global growth. These included economic situation, which has significantly affected trust in the construction of a state-of-the-art nutritional manufacturing business and prompted much discussion of change in certain facility in Singapore, our largest-ever capital investment in rules governing the economy. In the health care arena, budget Asia, as well as our acquisition of Solvay Pharmaceuticals, pressures are rising around the world, and the system reform which increases our presence around the world, particularly process in the United States bears significant implications for in emerging markets. We’re not only localizing our business the future of health care innovation and delivery. operations around the world – we’re doing the same with our citizenship efforts, enabling our international affiliates to adapt our approach to local stakeholders and their concerns. 2
- 5. 2009 GLOBAL CITIZENSHIP REPORT Innovating for the Future We also offer a wide range of educational Web sites to help We’re conducting this global expansion in accordance with our patients better understand their diseases and treatments, get values, as set out in the Abbott Brand Promise. Our Promise answers to their questions and connect with each other for pledges us to the welfare of “the people we serve in their information and support. pursuit of healthy lives.” The fundamental, and most important, way in which we do so is through our core purpose: developing Safeguarding the Environment new and better solutions to unmet health needs. We advanced Our commitment to improving lives extends, naturally, to this goal in a number of important ways in 2009. stewardship of our environment. By reducing the use of coal In addition to launching new technologies across our range and oil in our manufacturing operations, we’ve been able to of businesses, we increased our research capacity with new cut our CO2 emissions by 36 percent since 2006. We’re also laboratories in China and Singapore to help us focus on investing in renewable energy sources, such as solar power; varying regional requirements. Also with an installing energy-efficient equipment; and eye toward global needs, we’ve dedicated These are times that increasing our use of clean energy, such as an expert research team to a partnership we test a company’s co-generation technology that recycles waste formed with the Drugs for Neglected Diseases commitment to citizenship heat for electricity. Abbott is saving more than initiative (DNDi) to help advance potential and operating responsibly 1 billion gallons of water annually through and sustainably. We treatments for illnesses that disproportionately production process improvements. And we’re believe that Abbott has impact the developing world, such as malaria met that challenge. addressing the environmental impacts of our and tuberculosis. product packaging through more than 40 sustainable packaging initiatives. Enhancing Access As important as developing innovative treatments is ensuring Commitment that they get to the people who need them. We’re enhancing We understand well at Abbott that our citizenship work is access to our products through a multi-faceted approach. We never done. The essence of citizenship is that our commitment continue to engage with governments around the world to and vigilance are ongoing. Our inclusion on the Dow Jones offer our products at sustainable prices, appropriate to local Sustainability Index for the fifth consecutive year confirms that economic circumstances. We also offer our products for free we take our citizenship responsibilities very seriously. We or at deeply discounted prices through our Patient Assistance also were honored in early 2010 with recognition as the most Programs. And we donate them widely to address acute needs, admired pharmaceutical company on Fortune magazine’s such as the earthquake in Haiti. To help address needs on annual list, which is based on a rigorous survey of industry a sustainable, long-term basis, we also work in partnership executives, board members and financial analysts. with not-for-profit organizations and government agencies to We appreciate this affirmation that we’re moving in the right remove underlying barriers to access, such as lack of disease direction, as we will continue to do. We don’t have all the awareness or lack of infrastructure. In 2009, Abbott and the answers today. We will keep working to embed citizenship in Abbott Fund invested more than $580 million to support every aspect of our operations and to build a continually more capacity-building partnerships, provide patient assistance and sustainable business. We appreciate your interest in our efforts. donate cash and products to people in need. Sincerely, Protecting Patients and Consumers Because we develop products to improve human health, it’s particularly important that we ensure the safety of the ingredients and processes used in their production. We’re deeply committed to this goal at Abbott, and hold our more Miles D. White than 20,000 suppliers around the world to the same rigorous Chairman and Chief Executive Officer quality and safety standards we employ internally. April 2010 3
- 6. 2009 GLOBAL CITIZENSHIP REPORT Our Business About Abbott We have declared 345 consecutive quarterly dividends to We are a diverse, global health care company with scientific shareholders since 1924. Our return of cash to shareholders expertise and products that span the human life cycle. Our through dividends – combined with Abbott’s stock price products and services address the full range of health care appreciation – has generated a total shareholder return of needs from disease prevention and diagnosis to treatment and 20 percent over the past three years, compared to a decline of cure. Abbott discovers, develops, manufactures and markets 16 percent for the Standard & Poor’s Index over the same time leading pharmaceuticals, medical devices, diagnostics and period. Complete financial information about Abbott and a list nutrition products that are increasingly in demand in rapidly of our products are available at abbott.com. growing markets. In 2009, we took several strategic actions to augment and Global Presence reshape our portfolio for the long term. For example, with Solvay Abbott is headquartered in north suburban Chicago, Illinois. Pharmaceuticals, we are expanding our footprint in Eastern We serve customers worldwide with a staff of approximately Europe and emerging markets while adding approximately 83,000 employees at more than 100 research and development, $2.9 billion in annual sales of stable, global pharmaceutical brands. manufacturing and distribution locations. We also entered the vision care market through the acquisitions of Advanced Medical Optics – a global leader in cataract, refractive Improving Global Health and corneal products – and Visiogen, adding an entry point Our work targets diseases that pose the greatest burdens on into the key accommodating intraocular lens segment for the society, including cardiovascular disease, cancer, diabetes, treatment of presbyopia. In our vascular business, we entered infectious disease, autoimmune diseases, cognition disorders, the fast-paced sector of structural heart repair with Evalve, vision disorders and pain. Our diversity enables us to make a while in molecular diagnostics we added Ibis Biosciences. unique and powerful contribution to global health by combating diseases on many fronts. For example, scientists in our Global Growth pharmaceutical, medical devices and nutrition businesses are We deliver results for patients, health care professionals, collaborating on multiple approaches to prevent, diagnose, employees and shareholders. In 2009, we achieved record treat and manage heart disease. revenues of $30.8 billion. Abbott’s financial success enables us We constantly explore new ways of ensuring that patients to continually invest, innovate and introduce new products that have access to our products and benefit from our knowledge. are changing the practice of medicine. More than half our sales By partnering with others who share our commitment, we are come from international markets, with strong performance in finding better ways to help people in need overcome barriers to the emerging markets of Asia and Latin America. health care and live longer, more productive lives. Our success creates economic benefits for stakeholders around the world. We spent approximately $14 billion with READ MORE ONLINE suppliers during the year; paid $635 million in income taxes; paid ■ Citizenship Management and Governance our employees $7.7 billion; reinvested $2.7 billion in research and ■ GRI Index development; and returned $2.4 billion in dividends to investors. We also contributed more than $580 million in philanthropic www.abbott.com/citizenship/ourbusiness grants and product donations from Abbott and the Abbott Fund. 4
- 7. 2009 GLOBAL CITIZENSHIP REPORT 2009 Net Sales by Country Dollars in millions Total: $30,765 United States $14,453 The Netherlands $1,801 Japan $1,590 Germany $1,481 Italy $1,172 Spain $970 France $959 Canada $902 United Kingdom $779 All Other Countries $6,658 2009 Summary of Worldwide Sales Dollars in millions Total: $30,765 Pharmaceutical Sales $16,486 Nutritional Sales $5,284 Diagnostics Sales $3,578 Devices Sales $2,692 Other Sales $2,725 5
- 8. 2009 GLOBAL CITIZENSHIP REPORT Responding to the needs of moms and health care professionals, we developed Similac Advance EarlyShield to support a strong immune system, and designed the SimplePac container to enable easy, mess-free bottle preparation without adding stress to the environment. Innovation to Impact: The Similac Story 1 2 6 3 5 4 6
- 9. Citizenship and stakeholder engagement are integral to Abbott’s processes for developing, manufacturing, marketing and distributing our products. For example, we introduced in the United States Similac Advance EarlyShield, an innovative baby formula that Abbott scientists developed in consultation with physicians, hospitals and other infant nutrition experts. To meet the needs of today’s busy moms, we also designed a new, parent-friendly container, Similac SimplePac. 1 Consumer Input 4 Enhancing Hygiene and Convenience Seven out of 10 U.S. moms we surveyed told us that We redesigned our Similac packaging to improve both hygiene strengthening their babies’ immune systems is a top and ease of use. Our innovative SimplePac container, with priority. Abbott product development and packaging experts its unique scoop-in-the-lid design, eliminates the need for also gathered input from 1,600 moms to understand their caregivers to dig through the powdered formula by hand. concerns and frustrations with existing infant formula SimplePac is easy to grab and hold with one hand, and its products and packaging. wide rectangular shape and rounded corners make storage easier than ever. The Chicago Innovation Awards honored 2 Research and Development SimplePac with the organization’s first-ever People’s Choice Award in 2009. Abbott used cutting-edge science to create Similac Advance EarlyShield. Recognizing that 70 percent of the body’s immune system is located in the digestive tract, 5 Responsible Marketing we researched how creating a healthy digestive system Along with numerous health organizations worldwide, we can help support a strong immune system. We added believe breastfeeding is the optimal form of infant nutrition. prebiotics to promote digestive tract health, to complement We offer our infant nutrition products as high-quality alternatives nucleotides to support the immune system. Similac Advance for use when breastfeeding is either unavailable or not chosen. EarlyShield has been specially designed to help support We work diligently to educate consumers and health care the baby’s developing immune system. providers to help them make optimal nutrition choices for infants and toddlers, and we carefully audit our marketing and 3 Raw Materials Sourcing sales practices wherever Abbott nutrition products are sold to ensure compliance with relevant local codes and standards. The milk, ingredients, vitamins and minerals that go into Similac Advance EarlyShield come from only the highest-quality suppliers. As with all Abbott raw materials 6 Managing Environmental Impacts sourcing, we subject these ingredients to strict standards We subject certain Abbott product packaging to formal life for safety, purity and integrity. We also conduct rigorous cycle analysis so we can better understand its environmental tests to ensure the safety of our packaging materials. impacts – in our manufacturing and distribution as well as in consumers’ product usage and disposal. SimplePac packaging, for example, delivers improved hygiene and convenience with no increased environmental impact over conventional packaging. 7
- 10. Citizenship at Abbott We regard our strategies for business growth and profitability Embedding Citizenship Strategy as inseparable from our strategies for citizenship and with Business Strategy sustainability. We strive to simultaneously create both public A cross-functional Global Citizenship Working Group leads and private value – advancing science and helping to reduce the integration of citizenship strategy with core business the global disease burden, while rewarding our employees strategy, while driving transparency in the reporting of our and shareholders. economic, social and environmental performance. The team Given the growing complexities of our business, our includes representatives from Corporate Purchasing, Global markets and the world we inhabit, it is essential that we Environment, Health and Safety, the Office of Ethics and establish concrete goals, priorities and processes to guide our Compliance, Quality and Regulatory Affairs, Human Resources, citizenship strategies and measure our performance. Wherever Investor Relations, Government Affairs and Commercial possible, we develop metrics so that we and others may Operations. Many team members carry performance goals accurately assess our progress. and targets related to citizenship issues. The people of Abbott are committed to using our expertise Additionally, we conducted citizenship workshops with to improve global health care and advance sustainable senior managers and cross-functional teams from Brazil, development, while minimizing our impact on the environment. China, India, Ireland, Singapore and Vietnam in 2009 – enabling our local country operations to enhance their effectiveness Strategic Priorities in partnering with local stakeholders and addressing local A year ago, Abbott established four strategic priorities to align concerns. We will continue working throughout 2010 to further our citizenship activities with our business priorities. These embed our citizenship commitments and priorities across our are the areas where our core business can have the most diverse businesses, functions and geographies. significant impact on society and the environment. In the past year, we have worked diligently in pursuit of these priorities: • Innovating for the future – Using our core strengths as an innovator to make a difference to the health and well-being of people everywhere. • Enhancing access – Breaking down barriers that prevent many people worldwide from accessing the medicine and health care they need. Abbott was named to the Dow Jones Sustainability World Index for • Protecting patients and consumers – Protecting the health the fifth consecutive year. Abbott also was voted the most admired company in the pharmaceutical industry in Fortune magazine’s annual of those who use our products while promoting quality and list. Additionally, we ranked 6th on the 100 Best Corporate Citizens list safety in our operations and throughout our value chain. published by Corporate Responsibility magazine. • Safeguarding the environment – Playing our part in READ MORE ONLINE addressing the global challenges of climate change and ■ Citizenship Management and Governance water scarcity while minimizing the environmental impacts ■ GRI Index of our products. ■ Citizenship Awards and Recognition www.abbott.com/citizenship/ourbusiness 8
- 11. Stakeholder Engagement Engaging Our Stakeholders • Our employees offer important insights on the full range of Listening to our stakeholders and responding thoughtfully to company policies and procedures. We offer both formal and their concerns and ideas is vital to our progress as a global informal mechanisms to encourage employee feedback. citizen. Stakeholder insights help us develop • Our suppliers provide the raw materials new products to address unmet health The complex challenges we need to operate our businesses. care needs; educate patients, health care of advancing global Abbott maintains a Supplier Social professionals and others about emerging health care require Responsibility program (please see page extensive collaboration diseases and treatment options; and 34) that helps suppliers understand and partnership with a understand how and where our company wide range of groups. our citizenship expectations and can make a difference. requirements. We also work with them to improve both their processes and ours. Key Stakeholder Groups • Non-governmental organizations (NGOs) and How We Engage play a valuable role in alerting us to • Patients, consumers and customers emerging health care issues. We partner provide us with important insights about with numerous health care advocacy how we can improve our products and organizations around the world. develop new ones. We engage with • Local communities where we operate consumers through patient groups, have an important stake in how we do customer care lines, educational Web business and participate in community sites, surveys and market research. We affairs. In addition to our role as an also gain valuable feedback from retailers, employer and local taxpayer, we support employers, hospitals and clinics. educational efforts, civic, cultural and community health • Health care professionals help us stay abreast of challenges care organizations in our neighborhoods. on the front lines of health care; provide feedback on product quality, safety and efficacy; and conduct vital In 2010, Abbott will continue to expand our engagement research. We engage with health care professionals through with key stakeholders and further enhance our understanding educational programs, research partnerships and day-to-day of company impacts. We will conduct a survey of those who clinical practice. read this report, hold one-on-one meetings with key NGOs and • Governments and regulators help inform us about the needs expand our collaboration with certain patient organizations. and requirements of various communities, and we work with them both directly and through trade groups to advocate for advances in global health care. Abbott’s Code of Business READ MORE ONLINE Conduct guides all of our public policy engagements. ■ GRI Index ■ Our Partners www.abbott.com/citizenship/ourbusiness 9
- 12. 2009 GLOBAL CITIZENSHIP REPORT Our Strategic Priorities
- 13. Our society faces continuing economic challenges, coupled with rapid shifts in the global disease burden and rising concerns about the environment. We focus our global citizenship efforts in four key areas where we can best apply our science and expertise to address these complex, interrelated issues while earning stakeholder trust. Our strategic priorities remain: Innovating for the Future Enhancing Access Protecting Patients and Consumers Safeguarding the Environment
- 14. We are pleased to partner with Abbott to advance the treatment of cancer in Brazil. Our current efforts are focused around a clinical trial for an experimental medicine to treat non-small cell lung cancer. Supporting local studies not only helps develop new solutions for patients, but also helps to foster scientific and technological development in our country. Jose Rodrigues Pereira, M.D., professor, Irmandade da Santa Casa de Misericórdia de São Paulo
- 15. 2009 GLOBAL CITIZENSHIP REPORT INNOVATING FOR THE FUTURE Innovating for the Future Scientific discovery and innovation are the hallmarks of Abbott’s business – and the core of our commitment to advancing health and well-being. Our broad scientific expertise enables us to create new health care innovation and affordability with new approaches health care products, carry them through the critical stages that entail collaboration, coordination and partnership. of development and then deliver them to patients and health Abbott scientists regularly share knowledge and experience care providers around the world. Abbott’s diverse portfolio with scientists outside our company. For example, Abbott of pharmaceuticals, nutritionals and medical and diagnostic nutritionists and endocrinologists are assisting health care devices share a common framework of excellence in science, professionals in China in establishing new guidelines for research, development and engineering. appropriate treatment of malnutrition and diabetes. In the In 2009, Abbott invested $2.7 billion in research and United States and Europe, our scientists participate in development. Our ongoing investment in R&D enables us to numerous public-private technical advisory committees. address the ever-changing global disease burden and to Abbott works to ensure sufficient diversity in our product foster new, improved solutions for emerging pipeline so as to minimize risks while still health care challenges. pursuing exciting new discoveries. We $ 2.7 As health care systems become balance our R&D investments in high-risk, increasingly integrated, health care payers, potentially breakthrough products with governments, patients and others are billion investments in less risky, yet still vitally demanding that health care companies Abbott’s research and important refinements and improvements demonstrate clinical comparative development investment to existing products. effectiveness, and these stakeholders in 2009. Both types of investment deliver real increasingly negotiate for value-based value for patients and for our company. pricing. Abbott understands these Achieving the right balance is crucial expectations and works to meet the changing needs to Abbott’s continued economic sustainability – and to the of our customers and stakeholders. sustainability of our investment in potential new cures. A Collaborative Approach Health care research and development is costly and time-consuming. Only about one in 1,000 pharmaceutical (Top left) Abbott has engaged in decades of research to understand compounds progresses from the laboratory into a clinical the progression of cancer and identify new ways of treating it. Our trial, and only one in 12 compounds entering clinical trials oncology research is focused on the discovery and development of more targeted, less toxic therapies than are currently available – ultimately will reach patients. The development cycle for enabling patients to live longer, healthier lives. medical devices is also long and complex, with products being (Bottom left) A diseased non-small cell lung carcinoma is depicted tested in large clinical trials throughout the world. We believe among healthy cells. This cancer is the focus of the clinical trial under Abbott and our industry must respond to the challenges of way at Irmandade da Santa Casa de Misericórdia de São Paulo. 13
- 16. 2009 GLOBAL CITIZENSHIP REPORT INNOVATING FOR THE FUTURE Breakthrough Discoveries System, which has the potential to identify virtually all bacteria, Many of Abbott’s pharmaceutical R&D initiatives focus on viruses, fungi and other microorganisms. Our recent acquisition discoveries with the potential to unlock solutions to an of Solvay Group’s pharmaceuticals division positions us to important spectrum of medical needs. For example, Abbott introduce our innovative health care products and services into has spent more than 14 years researching our investigational Eastern Europe and emerging markets. Bcl-2 family protein inhibitor known as ABT-263, which may correct defects in cancer cells that allow them to survive Innovation Outside the Laboratory chemotherapy and other existing cancer treatments. Our Our commitment to innovation extends beyond the laboratory research – the subject of a 2005 cover story in the leading into everything we do – not only the creation, development and scientific journal Nature – aided in the development of a distribution of important health care products, but also to the potential new product for Abbott. Moreover, the findings work we do to help more people throughout the world gain are continuing to help answer fundamental access to appropriate health care products questions about the progress of cancer itself, and services. Abbott’s new PlexID as well as the progress of new therapies. We look for innovative ways to operate High-Throughput Similarly, Abbott is conducting innovative Bioidentification in developing and emerging countries – research in neuroscience – developing System was named pioneering new products to address unmet compounds that target receptors in the brain one of 2009’s top and rapidly growing health care needs. For that help regulate mood, memory and other innovations by The example, we are developing a mid-price neurological functions. These innovations are Scientist magazine, nutritional formula for infants and toddlers and also won The designed to address such conditions as in India, where much of the population is Wall Street Journal’s Alzheimer’s disease and schizophrenia. annual Technology anemic but cannot afford the premium Additionally, our experience in bringing Innovation Award. formulas that are common in developed to market the anti-TNF biologic HUMIRA countries. These innovative products meet provides a strong foundation for developing all international standards for quality and additional therapies designed to inhibit on ingredient composition but are offered in a selective basis the proteins responsible more affordable forms and package sizes. for inflammation. Along with innovative new products, we Responsible Research work tirelessly to enhance existing products. Biomedical research often raises ethical In 2009, for example, we launched our questions about the conduct of clinical trials, next-generation drug eluting stent, the the use of new technologies, animal welfare XIENCE PRIME Everolimus Eluting Coronary practices and other matters. Stent System, in Europe and select countries Abbott is committed to the highest throughout the Asia-Pacific and Latin standards of clinical practice in all of our American regions. XIENCE PRIME capitalizes on the proven research, including areas of bioethics bearing upon the clinical benefits of the market-leading XIENCE V stent, and is complex interaction of human life, science and technology. designed to provide outstanding flexibility and deliverability. In Our biomedical principles, embodied in our corporate addition, Abbott is studying new alloys and thinner struts for policies, focus on safeguarding the volunteers and patients improving ease of use, and we are leading the way in studying who participate in clinical trials. We take numerous steps – a fully bioresorbable device. Abbott’s rich vascular pipeline often going above and beyond what is legally required – to demonstrates our commitment to continuous innovation in uphold our high standards of quality, safety and transparency advancing treatment for coronary artery disease. at all stages and in all countries where we conduct trials. Abbott also invests in science that has originated outside our We also are committed to transparency in the clinical trials company where we see opportunities to apply our expertise we sponsor, recognizing the important public health benefits in developing and distributing new products. For example, we that can ensue. Abbott registers and discloses all applicable recently acquired the PlexID High-Throughput Bioidentification clinical studies, regardless of outcome, in a publicly accessible 14
- 17. Addressing Neglected Diseases Along with our research targeting some of the world’s most “Science has the power to change lives. pressing health care concerns, Abbott is committed to Today’s health care challenges faced by addressing neglected diseases – including tropical diseases that are especially endemic in low-income regions of Africa, low-income countries require innovative Asia and South America. In 2008, we formed a Neglected and collaborative approaches. Drugs for Disease Council to align and coordinate previously disparate Neglected Diseases initiative is proud to scientific discovery efforts. In 2009, we partnered with the partner with Abbott and other leading Drugs for Neglected Diseases initiative (DNDi) to identify existing molecular compounds in Abbott’s research library that scientific institutions to explore potential may offer promise in addressing neglected diseases. Using solutions to some of the world’s most computer modeling techniques, we have identified about pressing health care needs.” 70,000 compounds from a library of more than 1 million. For Shing Chang, Ph.D., R&D Director, DNDi example, one class of Abbott compounds shows potential impact on parasitic diseases. We are working with DNDi (Top left) Shing Chang, Ph.D., Research and Development Director on a plan to further investigate this discovery. Abbott also at the Geneva-based Drugs for Neglected Diseases initiative (DNDi), encourages its scientists to provide pro bono consulting to is working to develop new treatments for conditions such as malaria, sleeping sickness and Chagas’ disease. Abbott is partnering with DNDi on projects of mutual interest. DNDi to identify existing compounds in our library that may offer promise in addressing these and other illnesses. clinical trials registry (www.ClinicalTrials.gov). We voluntarily READ MORE ONLINE abide by the PhRMA Principles on Conduct of Clinical Trials ■ R&D Productivity and Capacity and Communication of Clinical Trial results and comply with ■ Product Highlights all relevant state and federal laws. For further details, please ■ Clinical Trial Registrations and Results Disclosures ■ Animal Welfare visit abbott.com. www.abbott.com/citizenship/innovation 15
- 18. We’ve taken on the major health problems of the poorest – tuberculosis, maternal mortality, AIDS, malaria. We’ve scored some victories in the sense that we’ve cured or treated thousands and changed the discourse about what is possible. Paul Farmer, United Nations Deputy Special Envoy for Haiti, Co-Founder, Partners In Health
- 19. 2009 GLOBAL CITIZENSHIP REPORT ENHANCING ACCESS Enhancing Access Abbott’s expertise, resources and global presence help to bridge gaps in health care access. We tailor our approach to specific patient needs in specific regions of the world. Expanding access to care requires addressing a complex We work to reach patients and health care professionals in array of challenges. Affordability is only one of many factors different economic strata through a variety of methods including that may be a barrier to access. The proportion of health education, infrastructure improvements and making specific care costs borne by individuals, governments and employers products more available and affordable. In 2009, we: varies significantly around the world. Lack • Trained more than 1,000 community of awareness about health care issues Affordability is only one of physicians in 10 Chinese cities in diabetes and treatments, inadequate health care many factors that play a prevention, treatment and management infrastructure and social stigmas also can role in increasing access. techniques. make it difficult for patients to get the • Trained more than 600 Chinese health medicines they need. Abbott works to address care professionals from 17 provinces in awareness pediatric nutrition management through these and other obstacles as part of our core business strategy and as part of our the Abbott Fund Institute of Nutrition infrastructure affordability commitment to enhancing global health and Science – helping to achieve a 27 percent well-being. reduction in malnutrition among pediatric overcoming The recent economic recession has education hospital patients in Shanghai. stigma heightened concerns about health care access • Reduced malnutrition by 42 percent among issues. For example, in the United States, 46 children under age 5 participating in a million patients do not have health insurance – school-based pediatric nutrition program and nearly a quarter of uninsured adults report in Vietnam. forgoing needed care. In 2009, Abbott worked with partners • Partnered with Drug Resource Enhancement against AIDS to advocate for U.S. health care reform legislation that would and Malnutrition (DREAM) to provide high-quality molecular expand health care coverage while fostering medical advances. testing systems to measure HIV viral load in patients in Mozambique and Malawi. Changing Disease Burden Abbott’s areas of expertise address many of the world’s most serious health care challenges – including cardiovascular (Top left) Paul Farmer, M.D., formed Partners In Health (PIH) to serve patients in Haiti more than 20 years ago. Abbott supports PIH projects disease, cancer, HIV/AIDS, malnutrition and diabetes. In including health clinics, training programs and mobile health screening keeping with our expertise, our approach to expanding health units in 12 countries. care access is designed not only to address acute health care (Bottom left) Abbott experts are helping the Haiti PIH team expand dilemmas in the developing world, but also to help governments its nutrition production and quality while supporting local economic development. We are committed to advancing this work in 2010 and health care professionals in emerging markets manage and beyond as part of Haiti’s rebuilding process (for further details, the growing impact of chronic conditions. please see page 36). 17
- 20. 2009 GLOBAL CITIZENSHIP REPORT ENHANCING ACCESS We also introduced a number of country-specific innovations Intellectual Property to address local health care challenges. In Tanzania, Abbott Intellectual property protection and the patent system and the Abbott Fund are working with the government to are necessary mechanisms to promote the research and implement a comprehensive, multi-year strengthening of the development of new, life-improving products. Strong country’s health care system, with special focus on treating intellectual property systems foster an innovative culture, Tanzanians living with HIV and other chronic diseases. In in which local innovators can develop new products and December 2009, we helped open the nation’s first emergency technologies knowing that their inventions and creativity are medical department at Muhimbili National Hospital in Dar es secure. Policies and mechanisms that undermine intellectual Salaam. The department treated more than 500 patients – and property protections impede the development of, and access helped to stabilize more than 100 patients who were critically to, new medicines over the longer term because they lead ill – during its first week of operation. Going forward, a new, to an unsustainable environment for the introduction of new Abbott-sponsored educational effort housed at Muhimbili is medicines for that environment. helping to train health care providers throughout the country in Abbott evaluates intellectual property protection on emergency medicine. a case-by-case basis, taking into consideration the drivers 42% We also donate rapid diagnostic tests for promoting innovation and investment, to relief partners in developing countries, as well as public health needs. For our focusing on preventing mother-to-child HIV products, Abbott does not have HIV transmission. Since 2002, Abbott has patent protection in any least developed donated 15 million rapid HIV tests through country or all of Africa, with the exception this program – helping to avert more than of South Africa. In conjunction with 125,300 potential infections of infants. this, Abbott offers a comprehensive In addition, Abbott has developed an reduction in malnutrition program to expand access and HIV-1 dried blood spot protocol that allows among children under age affordability to its HIV medicines in the patient samples from remote areas of the 5 participating in a school- developing world. We are committed based pediatric nutrition world to be transported and tested while to broad registration of our products program in Vietnam. maintaining high-quality results. We have with regulatory agencies in nations where studied performance of the methodology in those products are needed. countries such as Tanzania, Cameroon and South Africa, and the technology is gaining greater acceptance in remote health Broad Registration care settings that are far from centralized laboratory services. As an example of our approach to broadening health care access, Kaletra (lopinavir/ritonavir), marketed as Aluvia in Sustainable Pricing developing countries, is Abbott’s leading HIV protease inhibitor Abbott works with governments and other health care and is the most widely registered HIV medicine in the world, purchasers to make our products available at prices according to the World Health Organization. Kaletra/Aluvia appropriate to each marketplace. Abbott’s preferential pricing tablets are available, filed or approved in 170 countries where system is designed to increase access to our HIV medicines 98 percent of the developing world’s HIV-positive population while balancing affordability based on disease burden and lives. Additionally, our lower-strength tablet for pediatric governments’ ability to finance health systems. In 2002, Abbott use has been approved, available or filed in 124 countries. made a commitment to sell its HIV medicines at $500 per Abbott plans an equally broad and rapid registration process patient per year in Africa and the United Nations-defined least for our new heat-stable Norvir tablet. developed countries (69 countries). Abbott then expanded its preferential pricing program to create a new tier for low- Effective Philanthropy and low-middle-income countries. A further price reduction Whenever possible, we work to facilitate access by occurred in 2009. In less developed countries, Abbott has expanding our core businesses in pharmaceuticals, nutritionals, consistently provided branded antiretroviral medicines at prices medical devices and diagnostics – developing new products competitive with those of generic alternatives. and services to address unmet health care needs. We favor 18
- 21. Increasing Access Through Our Diversified Expertise Abbott is a worldwide leader in helping physicians treat patients with heart disease. For example, Abbott’s market- leading XIENCE V stent is used to prop open a narrowed or blocked artery and release the drug everolimus in a controlled manner to prevent the artery from becoming blocked again following a stent procedure. XIENCE V – which is available in the United States, as well as key countries in Europe, Asia and Latin America – is built upon Abbott’s market-leading bare metal stent, the MULTI- LINK VISION Coronary Stent System. The VISION platform is designed to facilitate ease of delivery, making it easier for physicians to maneuver the stent and treat the diseased portion of the artery. Abbott sponsors numerous education and training programs to provide physicians with the latest information on interventional cardiology procedures. For example, we support the Crossroads Institutes in Tokyo and Brussels in providing advanced training and peer-to-peer education for physicians from around the world. “I couldn’t believe I was having a heart attack. At first, doctors planned to perform a triple bypass. But my partner asked if stents were an option – and we were relieved to discover that I was a good candidate. I got to go home two days after the procedure – and two days after that, I was back to work and feeling great.” Terry Ortega, patient, San Diego, California market-based solutions to health care challenges because (Top left) Terry Ortega suffered a heart attack during a family camping trip. His partner, Danell Templeton, an Abbott Vascular employee, requested they tend to be more sustainable over the long term. that he be treated with a XIENCE V stent. Following his stent procedure, However, when market-based solutions are impractical Terry was able to rapidly return to his active lifestyle – including camping, or insufficient, we facilitate access through our philanthropy – bowling and riding motorcycles. by donating health care products and services, by (Bottom left) Abbott’s XIENCE V stent is just one example of the facilitating patient assistance and by supporting a wide company’s focus on advancing the treatment of vascular disease by combining the latest medical device innovations with world- array of civil society organizations engaged in the direct class pharmaceuticals, investing in research and development and delivery of health care services. Often, we find we can most supporting training and education. effectively promote health care access by collaborating READ MORE ONLINE in public-private partnerships with governments, universities, ■ Patient Assistance Programs research centers, health care companies, hospitals, clinics ■ Product Donations and related institutions. ■ Global AIDS Care ■ Our Commitment to Child Nutrition www.abbott.com/citizenship/access 19
- 22. I am proud to work at Abbott, a company that takes good care of patients with rheumatoid arthritis, especially those like my mom. Working in Abbott’s immunology business, I have the privilege of helping patients with serious diseases manage their symptoms and live normal lives. Elaine Wagner, Executive Assistant, Abbott Brazil
- 23. 2009 GLOBAL CITIZENSHIP REPORT PROTECTING PATIENTS AND CONSUMERS Protecting Patients and Consumers Our ability to create life-enhancing and life-preserving products is constantly bolstered by scientific advances, but delivering on our commitment to patients and consumers goes well beyond the laboratory. To deliver safe and effective products – at the right time and that health care professionals are fully informed of the in the right dosage or formulation – we must work to educate benefits and risks of our products. patients and health care professionals about potential risks • Directly and indirectly educating patients about proper and side effects. All medicines, health care interventions, storage, use and disposal of their medicines and health care procedures and products carry some degree of risk – which products under appropriate medical supervision. must be balanced against the often greater risks associated with the illnesses or injuries these interventions are designed Our commitment to patient and consumer protection is to treat. Fully educating and informing our stakeholders shared by all 83,000 Abbott employees and by the even about these risks and benefits is paramount. greater numbers of people working on behalf of our partners Along with educating patients and health care and suppliers. We hold all of them to high ethical and professionals about the safe use of our performance standards and maintain 83,000 products, we work hard to ensure safety effective management systems to review and consumer protection throughout the and audit them. manufacturing and distribution process. The safety of many product ingredients Ensuring Quality is generating significant debate throughout Abbott employees in Manufacturing are committed to patient and the health care industry. At the same time, Abbott employs and adheres to robust consumer protection. the broad geographic dispersion of our manufacturing quality standards for all of supply chain requires increased oversight our products at every manufacturing site and auditing. around the world. We use a common quality management Our obligation to protect patients and consumers goes system throughout the company, and when we acquire new beyond simply complying with regulatory requirements. businesses, we use due diligence and rigorous analysis to It means earning and keeping the trust of all those who ensure that their quality standards and practices quickly depend on Abbott products by: conform to ours. • Ensuring quality, safety and product integrity across the full spectrum of research, development and manufacturing (Top left) Elaine Wagner, Executive Assistant in Abbott Brazil’s of our products and packaging. immunology department, has seen firsthand how HUMIRA helps • Ensuring the quality, safety and authenticity of each her mother, Neusa Maria Franco Wagner, manage the challenges of rheumatoid arthritis. product that bears the Abbott name through our distribution channels. (Bottom left) Muriel Huf was born prematurely, with a congenital heart defect. Abbott’s Synagis helped protect her lungs, and her parents • Effectively identifying and targeting the most appropriate used Abbott Brazil’s educational Web site about caring for premature patient profiles for each Abbott product and ensuring babies to help her grow and thrive. 21
- 24. 2009 GLOBAL CITIZENSHIP REPORT PROTECTING PATIENTS AND CONSUMERS All our processes and products are regularly monitored the senses to make disease prevention information more and assessed against approved specifications before accessible – is currently touring throughout Europe. distribution. We conduct a rolling two-year program of audits Web sites are another important educational tool. Abbott’s covering all of our 60 manufacturing sites, assessing each public education Web sites include: site against our quality standards. The Abbott Quality and • www.knowyourhdl.com and www.knowyourtrigs.com, two Regulatory organization is itself routinely challenged and Web sites introduced in 2009 that help people understand assessed through internal audits and regulatory inspections. the importance of HDL and triglyceride levels in managing the risk of heart disease. Addressing Concerns • Our Diabetes Control for Life program Abbott takes potential safety concerns about our products (www.diabetescontrolforlife.net), a free, 24-week Web- very seriously, and we work quickly to address questions based diabetes management program available in multiple from customers, patients, consumers and the general public. languages for people with Type 2 diabetes. We investigate each instance and communicate findings and • www.prematuro.com.br, a Brazilian Web site providing lessons learned to the appropriate regulators, to others in our education about the unique health care needs of premature industry, if appropriate, and across all our business divisions. infants following hospital discharge. For example, in early 2009, we acted voluntarily to withdraw • www.iLASIK.com, a Web site educating patients about laser several varieties of ZonePerfect nutrition bars vision correction that is available in seven in 13 countries because they contained peanut Educational Web sites languages and offers an automated physician ingredients sourced from a supplier that had help patients and finder feature to help facilitate patient been implicated in a salmonella contamination families more effectively interactions with potential surgeons. incident. Although none of our nutrition bars manage their ongoing health care needs. was shown to be contaminated, we withdrew Responsible Sales and them from the market in order to ensure Marketing Practices consumer confidence in our brand. Abbott recognizes public concerns about the interaction between health care sales Patient Education and marketing personnel and health care Education efforts aimed directly at patients providers, and we work to ensure that our and consumers are another important tool for employees and partners stay focused at ensuring appropriate use of medicines and all times on our commitment to protecting health care products, and for helping families patients and consumers. An important more effectively manage their ongoing health part of that commitment is an obligation to care needs. Abbott supports a wide range of educational communicate responsibly with patients, consumers and health programs and activities, ranging from health screenings care professionals about diseases, our health care products to education about the signs and symptoms of chronic and the alternatives available to them. diseases to partnerships with health care professionals Abbott’s sales force and distribution network are trained and nonprofit organizations. to educate health care professionals on disease prevention For example, in the United States, Abbott partners with the and treatment options, and we adhere to a rigorous set of American Diabetes Association to support Safe at School, standards and codes governing advertising, payments to an educational program that helps families learn more about doctors and other health care professionals, as well as other appropriate diabetes care in school settings. In 2009, we sales and marketing practices. For details of these codes and joined forces with several Danish patient associations and guidelines, please turn to page 30 or visit abbott.com. health care providers to sponsor an interactive exhibit called LEARN (Living the Experience of Autoimmune ReactioNs). Appropriate Patient Treatment This exhibit enables visitors to literally walk through a model of Providing health care professionals with detailed data and the immune system to understand how autoimmune diseases information to help them properly use and prescribe our cause the body to attack itself. The exhibition – which engages products is another essential element of patient protection. 22
- 25. “Abbott’s commitment to vision care Darlene Schweitzer discovered she had fast-growing cataracts during a routine eye exam. Her doctor recommended surgery to restore her has allowed me to offer a safe, state- vision and help her maintain her active lifestyle. of-the-art, effective option for cataract patients. It allows them to achieve more functional vision after the surgery than was possible before.” Coleman R. Kraff, M.D., surgeon, Chicago, Illinois Improved science, epidemiologic data and increasingly clinical evidence; and that Abbott sales representatives are customized diagnostic technologies provide us with better accountable for serving the interests of patients and consumers. understanding of which medical interventions are most appropriate for a given patient. For instance, Abbott’s research Product Protection and investment in the rapidly expanding field of molecular Today’s health care marketplace also faces a growing black testing are enhancing health care professionals’ ability to market for counterfeit medicines, as well as stolen or illegally customize treatment methodologies for each patient. Our diverted products. Counterfeit or diverted products pose risks scientists are working to integrate findings from our ongoing to patients because they may contain incorrect ingredients or study of molecular diagnostics into our discovery and be subject to tampering. Our Global Product Protection Team development programs for new treatments. constantly analyzes our global supply chain – looking for weak We continually improve and update the training we provide points and identifying opportunities to reduce the possibility of for our sales representatives so that they can help physicians product interference. better understand how to prescribe the right medicine or medical device for the right patient in the right dosage at the READ MORE ONLINE right time. In 2009, Abbott continued to establish mechanisms ■ Ensuring Quality in Manufacturing to enhance the quality of our interactions with health care ■ Serious Adverse Events Consortium professionals. These metrics help ensure that sales initiatives ■ Sales, Marketing and Advertising Standards and Codes ■ Protecting Patients from Counterfeiting and Theft for a given product reach only the appropriate physicians; that www.abbott.com/citizenship/patients those physicians are well apprised of the most up-to-date 23
- 26. Abbott strives to be a good steward of the earth and its resources – with every product we make and in every market we serve. We are committed to reducing our carbon intensity and water use, while revising product features and design to minimize environmental impact. Corlis Murray, Vice President, Global Engineering Services
- 27. 2009 GLOBAL CITIZENSHIP REPORT SAFEGUARDING THE ENVIRONMENT Safeguarding the Environment Abbott has long been committed to minimizing our global environmental impacts – in the sourcing of raw materials, in the manufacture and distribution of our products and in the use and disposal of our products by consumers and health care practitioners. Our environmental stewardship initiatives help protect to working with governments and non-governmental the planet while improving efficiency, reducing costs and organizations to achieve further progress toward climate preserving our ability to do business in the future. Our three policy goals. environmental priorities are climate change, water usage and product stewardship. Abbott’s Carbon Footprint Our direct emissions come from the carbon emitted at our Addressing Global manufacturing sites, in our offices and by Climate Change Abbott’s Carbon Footprint our vehicle fleets. We have been measuring Climate change is a serious issue that has ■ Manufacturing these emissions for many years, and we the potential to affect businesses in many Fuel Combustion have established a policy and targets to help ways. For example, extreme weather patterns 656,000 metric tons CO2e us achieve continuous improvements. and water scarcity may pose challenges for ■ Electricity Use sourcing, operations and distribution, while 764,000 metric tons CO2e Adapting Our Energy Practice a more carbon-constrained world could ■ Global Sales Fleet for a Low Carbon Economy add to energy costs. Additionally, climate 171,000 metric tons CO2e High energy prices and price volatility, change could lead to significant changes in coupled with the impact on the environment the global disease burden. 764k of greenhouse gases generated from our 656k Action is required on our part – not only use of energy, make energy management an to mitigate these risks, but also to help important issue for Abbott. The greenhouse society adapt to the climatic changes already gases we emit from fuel combustion at under way. As a global health care company, 171k our manufacturing sites make up a significant we are committed to helping address this proportion of our direct impacts. Includes Greenhouse Gas Protocol issue – by reducing our direct impacts through Scopes 1 and 2 By the end of 2009, we had reduced manufacturing and by making a positive carbon dioxide equivalent (CO2e) emissions difference through indirect impacts across our value chain. from manufacturing by 36 percent compared with 2006 Government and policymakers also have an important (Top left) Corlis Murray, Vice President, Global Engineering Services, role to play in addressing global climate change. Through leads a team of more than 1,000 professionals in advancing Abbott’s our membership in the U.S. Environmental Protection operational excellence while ensuring environment, health and safety leadership. Agency’s Climate Leaders Program and the Business Roundtable Climate Resolve Initiative, Abbott has joined (Bottom left) Through an educational partnership with Project WET, volunteers from Abbott’s manufacturing plant in Casa Grande, Arizona, other businesses in advocating public-private partnerships are helping local businesses, schools and families save more than to ensure responsible climate policy. Abbott is committed 2.3 million gallons of water. 25
- 28. 2009 GLOBAL CITIZENSHIP REPORT SAFEGUARDING THE ENVIRONMENT levels, normalized by sales, thereby exceeding our target company is committed to managing its water use in an of a 30 percent reduction by 2011. efficient, sustainable manner and to improving people’s We are achieving these reductions primarily by access to clean water in the communities where we improving energy efficiency and switching to low carbon work and live. fuels and renewable energy. We generate electricity through In 2004, we set a global target to reduce total water co-generation at five of our manufacturing sites, which intake by 40 percent (normalized by sales), and we exceeded increases plant efficiency while reducing both energy costs that target by the end of 2009. Each of our 15 most and environmental impact. In 2009, we achieved a 32 percent water-stressed manufacturing sites is preparing local water reduction in electricity purchased, exceeding our 2011 target management plans to help them better understand, anticipate of a 12 percent reduction (on a 2006 baseline, indexed to and plan for water restrictions that could have an impact sales). We also have committed to investing in renewable on business operations. We also are partnering with Project energy projects at each of our major manufacturing locations WET – a leading water advocacy organization – to educate by the end of 2011. To date, we have launched renewable communities about steps that homes, schools and businesses energy programs at three of our 10 largest facilities. can take to conserve water and reduce waste. In 2009, To challenge ourselves further, we have set an absolute more than 2,600 grade school students and dozens of reduction target for CO2e for 2015 (see box on page 28). businesses in neighborhoods surrounding our Casa Grande, Between 2005 and 2009, we reduced our absolute CO2e Arizona, manufacturing plant participated in Project WET by 10.7 percent, which represents good progress toward training and conservation measures – saving more than this new target. 2.3 million gallons of water. In addition, Abbott is incorporating water-saving design Our Wider Footprint and technology into our facilities in less stressed areas. Carbon emissions also are associated with the sourcing, For example, our manufacturing plant in Altavista, Virginia, distribution and consumer use of our products. conducted a utilities optimization study during 2009 that We are starting to estimate this wider carbon footprint, helped us reduce usage by 12 million gallons of water annually. focusing first on indirect emissions arising in the supply chain and distribution. Our initial analysis suggests that our supply Product Stewardship chain footprint is on the order of six to 14 times that of our Abbott is partnering with a wide range of stakeholders direct emissions. Emissions from agriculture, particularly linked to ensure appropriate stewardship of our products across to the nutrition business, represent the single largest source our value chain. We are revising product features as well of emissions. as design to enhance the safety and effectiveness of our We are using this analysis of our wider footprint to help products, while minimizing their environmental impact across shape our future priorities. For example, waste management the life cycle of manufacturing, storage, distribution, use emerged as one of the top five sources of emissions – and and disposal. we are responding by putting in place a phased waste management strategy that covers both our own manufacturing Sustainable Packaging operations and, ultimately, those of our suppliers. Abbott’s primary concern in designing packaging for our Additionally, we are working to assess other sources health care products is to ensure their quality, safety and of indirect emissions, such as consumer use and disposal of integrity. Packaging protects our products from damage and our products. contamination, communicates essential product information and often makes the product easier to use. A Comprehensive Water Approach Nonetheless, we recognize that our packaging has impacts Clean water is a critical and finite resource, essential to on the environment through the energy and materials required sustaining human health, economic growth and the in its manufacture and in its ultimate disposal. We are environment. Access to water is essential to our manufacturing implementing strategies to reduce those impacts throughout operations and to all those who use our products. Our our company. 26
- 29. (Left) A worker at Abbott’s manufacturing plant in Casa Grande, Arizona, prepares corrugate trays for loading into a wrapping machine. “Working on the new wastewater Abbott is working to reduce our environmental impact through treatment facility at our nutrition investments in sustainable packaging. manufacturing plant helps me know (Right) Abbott’s manufacturing plant in Sturgis, Michigan, operates a state-of-the-art wastewater treatment facility and has installed I’m playing a role in building a sustainable efficient water scrubbers to control dust emissions – resulting in the saving of 8.5 million gallons of water per year. future for our community.” James Hoffman, Industrial Treatment Operator, Sturgis, Michigan Our sustainable packaging initiatives are aimed at reducing reduces polystyrene use by 15.8 percent, or 12,750 the amount of packaging materials used through better design; pounds per year. substituting more environmentally friendly packaging materials • A new pilot program for shipping physician samples that wherever we can; reducing the use of materials of particular require refrigeration. In place of our old, single-use shipping environmental concern; and working with others in our industry box, the program introduces a new, smaller box that can to promote best practices. be reused more than 100 times. It is made of recyclable, To date, Abbott has launched more than 40 sustainable 100 percent organic-based materials. packaging initiatives aimed at achieving a 5 percent reduction in the amount of packaging we use by 2013. Recent Since 2007, we have eliminated nearly 4.5 million pounds sustainable packaging initiatives include: of packaging annually. In addition, we have adopted new • A 15 percent reduction in the amount of plastic used in packaging guidelines to help our packaging and purchasing infant formula containers. professionals make more sustainable choices. • An 8.3 percent reduction in the amount of polypropylene Abbott also works with several industry and independent plastic used in Abbott Nutrition’s eight-ounce reclosable advisory groups, including the Sustainable Packaging bottles. By reducing the use of polypropylene by 2.7 million Coalition and the European Organization for Packaging and pounds annually, we also help save some 436,000 gallons the Environment (EUROPEN). We also partner with suppliers of gasoline needed to transport our products. who have achieved or are pursuing certification with respected • A new, smaller package for Abbott Vascular’s Emboshield sustainability organizations, such as the Program for the filters weighing 30 percent less. Endorsement of Forest Certification, the Forest Stewardship • Redesigned clamshell packaging for Lupron, used in treating Council and the Sustainable Forestry Initiative. prostate cancer and endometriosis. The new package 27
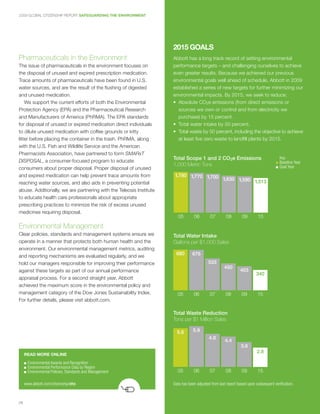
- 30. 2009 GLOBAL CITIZENSHIP REPORT SAFEGUARDING THE ENVIRONMENT 2015 GOALS Pharmaceuticals in the Environment Abbott has a long track record of setting environmental The issue of pharmaceuticals in the environment focuses on performance targets – and challenging ourselves to achieve the disposal of unused and expired prescription medication. even greater results. Because we achieved our previous Trace amounts of pharmaceuticals have been found in U.S. environmental goals well ahead of schedule, Abbott in 2009 water sources, and are the result of the flushing of digested established a series of new targets for further minimizing our and unused medication. environmental impacts. By 2015, we seek to reduce: We support the current efforts of both the Environmental • Absolute CO2e emissions (from direct emissions or Protection Agency (EPA) and the Pharmaceutical Research sources we own or control and from electricity we and Manufacturers of America (PhRMA). The EPA standards purchase) by 15 percent. for disposal of unused or expired medication direct individuals • Total water intake by 50 percent. to dilute unused medication with coffee grounds or kitty • Total waste by 50 percent, including the objective to achieve litter before placing the container in the trash. PhRMA, along at least five zero waste to landfill plants by 2015. with the U.S. Fish and Wildlife Service and the American Pharmacists Association, have partnered to form SMARxT DISPOSAL, a consumer-focused program to educate Total Scope 1 and 2 CO2e Emissions Key ■ Baseline Year 1,000 Metric Tons ■ Goal Year consumers about proper disposal. Proper disposal of unused and expired medication can help prevent trace amounts from 1,780 1,770 1,700 1,630 1,590 1,513 reaching water sources, and also aids in preventing potential abuse. Additionally, we are partnering with the Teleosis Institute to educate health care professionals about appropriate prescribing practices to minimize the risk of excess unused medicines requiring disposal. 05 06 07 08 09 15 Environmental Management Clear policies, standards and management systems ensure we Total Water Intake operate in a manner that protects both human health and the Gallons per $1,000 Sales environment. Our environmental management metrics, auditing and reporting mechanisms are evaluated regularly, and we 680 676 hold our managers responsible for improving their performance 533 450 against these targets as part of our annual performance 403 340 appraisal process. For a second straight year, Abbott achieved the maximum score in the environmental policy and management category of the Dow Jones Sustainability Index. 05 06 07 08 09 15 For further details, please visit abbott.com. Total Waste Reduction Tons per $1 Million Sales 5.6 5.9 4.8 4.4 3.6 2.8 READ MORE ONLINE ■ Environmental Awards and Recognition ■ Environmental Performance Data by Region ■ Environmental Policies, Standards and Management 05 06 07 08 09 15 www.abbott.com/citizenship/ehs Data has been adjusted from last report based upon subsequent verification. 28
- 31. Performance and Operational Review Ethics and Compliance Our People Supply Chain Management Global Community Disaster Relief and Rebuilding in Haiti