Study design in research
- 1. STUDY DESIGN IN RESEARCH * Few jewels from ocean Dr. Kusum Gaur Professor, PSM WHO Fellow IEC
- 2. Definition of Research “Research is a systematized effort to gain new knowledge”. 12/08/2012 Dr. Kusum Gaur 2
- 3. Steps in Research (Holy 11) 1. Collect review of literature/Situation Analysis 2. Identify and prioritize health problems 3. Decide aims & objectives 4. Planning Methodology including study design 5. Execution 6. Compilation, Classification & Presentation of data 7. Analysis 8. Test of Significance/Test of Hypothesis 9. Inferences 10. Report Writing 11. Dissemination of Report 12/08/2012 Dr. Kusum Gaur 3
- 4. Study Design A study design is a specific plan or protocol for conducting the study, which allows the investigator to translate the conceptual hypothesis into an operational one. 12/08/2012 Dr. Kusum Gaur 4
- 5. Direction of Study Backward Forward Cross -sectional Retrospective Prospective 3 4. Ambidirectional 12/08/2012 Dr. Kusum Gaur 5
- 6. Decision Tree Intervention Done No Yes Observational Study Experimental Study Comparison Group Randomization No Yes No Yes Descriptive Study Analytic Study NRCT Study RCT Study Direction of Study E O E O Cohort Study E = O Case-Control Study Cross-Sectional Study 12/08/2012 Dr. Kusum Gaur 6
- 7. Epidemiological Study Design Observational Studies Descriptive Studies Analytic Cross-Sectional Case-Control Cohort Experimental / Interventional studies As per Control: RCT/NRCT As per Blinding: Single /Double Blind As per Design: Simple/Cross-over As per Area: Field/Clinical/Lab 12/08/2012 Dr. Kusum Gaur 7
- 8. Descriptive Studies • Case reports • Case series • Population studies 12/08/2012 Dr. Kusum Gaur 8
- 9. Descriptive Studies: Uses • Hypothesis generating • Suggesting associations 12/08/2012 Dr. Kusum Gaur 9
- 10. Descriptive Type of Observational Study • Other Name Case-Series/Population • Unit of Study Case/Individuals • Study Question What is happening • Direction Of Inquiry • Study Design ☻☻☻☻☻☻ desired information ☻☻☻☻☻☻ about cases/individuals is collected 12/08/2012 Dr. Kusum Gaur 10
- 11. Case-Series ……. Advantages • Easy to do • Excellent at identifying unusual situation • Good for generating hypotheses Disadvantages • Generally short-term • Investigators self-select (bias!) • no controls 09/03/2010 Dr. Kusum Gaur 11
- 12. Analytical Observational Studies • Cross-sectional • Case-control • Cohort 12/08/2012 Dr. Kusum Gaur 12
- 13. Cross-sectional Study • Data collected at a single point in time • Describes associations • Prevalence A “Snapshot” 12/08/2012 Dr. Kusum Gaur 13
- 14. Cross-Sectional Study • Other Name Prevalence Study • Unit of Study Individual • Study Question What is happening • Direction of Inquiry • Study Design Exposed to Factor Not Exposed Diseased to Factor Population Exposed to Factor Non- Disease Not Exposed to 12/08/2012 Dr. Kusum Gaur Factor 14
- 15. Objectives of a Cross-Sectional Study To find out association 12/08/2012 Dr. Kusum Gaur 15
- 16. Cross-sectional Study Sample of Population Defined Population Regular Not doing meditation Meditation Prevalence of Prevalence of DM DM Time Frame = Present 12/08/2012 Dr. Kusum Gaur 16
- 17. Cross-sectional Study E.G. Out of 1000 population if 100 were doing meditation regularly & out of that only 2 were having DM. Remaining 900 were not doing meditation at all, out of that 220 were having DM. + DM - 2 98 Meditation + - 220 680 12/08/2012 Dr. Kusum Gaur 17
- 18. Cross-Sectional Study • Strengths – Quick – Cheap • Weaknesses – Cannot establish cause-effect 09/03/2010 Dr. Kusum Gaur 18
- 19. Case-Control Studies Start with people who have disease(Cases) Match them with controls that do not have disease (Match Confounding) Look back and assess exposures 12/08/2012 Dr. Kusum Gaur 19
- 20. Controls A control is a standard of comparison (confounded with variability but without effect) for • Effects • Variability 12/08/2012 Dr. Kusum Gaur 20
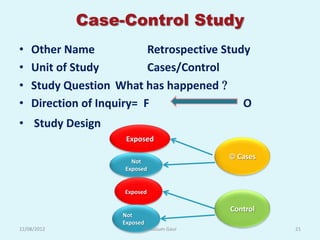
- 21. Case-Control Study • Other Name Retrospective Study • Unit of Study Cases/Control • Study Question What has happened • Direction of Inquiry= F O • Study Design Exposed Cases Not Exposed Exposed Control Not Exposed 12/08/2012 Dr. Kusum Gaur 21
- 22. Objective of a Case-Control Study To find out association To assess Risk Ratio 12/08/2012 Dr. Kusum Gaur 22
- 23. Case-Control Study Cases Regular Meditation Patients with DM No Meditation Controls Regular Meditation Persons w/o DM No Meditation Past Present 12/08/2012 Dr. Kusum Gaur 23
- 24. The logic of Case-Control Studies Cases differ from controls only in having the disease If exposure does not predispose to having the disease, then exposure should be equally distributed between the cases and controls. The extent of greater previous exposure among the cases reflects the increased risk that exposure confers 12/08/2012 Dr. Kusum Gaur 24
- 25. Case-Control Studies: Strengths • Good for rare outcomes: cancer • Can examine relation of exposures to disease • Useful to generate hypothesis • Fast • Cheap • Provides Odds Ratio 09/03/2010 Dr. Kusum Gaur 25
- 26. Case-Control Studies: Weaknesses • Cannot measure – Incidence – Prevalence – Relative Risk • Can only study one outcome • High susceptibility to bias 09/03/2010 Dr. Kusum Gaur 26
- 27. Cohort Study • Begin with disease-free individuals • Classify patients as exposed/unexposed • Record outcomes in both groups • Compare outcomes using relative risk 12/08/2012 Dr. Kusum Gaur 27
- 28. Cohort Study • Other Name Prospective Study / Follow-up Study/Incidence Study • Unit of Study Individual • Study Question What is happening • Direction of InquiryF O Diseased • Study Design • Exposed to Factor Not Non Diseased Cohort Cohort Diseased Not Exposed to Factor Non-Diseased 12/08/2012 Dr. Kusum Gaur 28
- 29. Logic of Cohort Study Cohort is a group of persons sharing a common characteristics Differences in the rate at which exposed and control subjects contract a disease is due to the differences in exposure, since others are known and similar. 12/08/2012 Dr. Kusum Gaur 29
- 30. Cohort Study Prospective (usually) Controlled Can determine causes and incidence of diseases as well as identify risk factors Generally expensive, time consuming and difficult to carry out 12/08/2012 Dr. Kusum Gaur 30
- 31. Steps for Cohort Study Identify geographically defined group Identify exposed subjects and not exposed subjects Follow over a specific time Record the fraction in each group who develop the condition of interest Compare these fractions using RR, AR or OR 12/08/2012 Dr. Kusum Gaur 31
- 32. Objectives of a Cohort Study To find out association To assess Risk Ratio To find out Relative Risk To find out Attributed Risk 12/08/2012 Dr. Kusum Gaur 32
- 33. Prospective Cohort Study DM No Meditation No DM Cohort DM Regular Meditation No DM Present Future 12/08/2012 Dr. Kusum Gaur 33
- 34. Cohort Study: Strengths • Can measure multiple outcomes • Can adjust for confounding variables • Can calculate Attributed Risk 09/03/2010 Dr. Kusum Gaur 34
- 35. Cohort Study: Weaknesses • Expensive • Time consuming • Cannot study rare outcomes • Confounding variables 09/03/2010 Dr. Kusum Gaur 35
- 36. Measurements of Association Cohort Study Case Control Study •Significance Test •Significance Test •Relative Risk •OR •Attributable Risk •OR 12/08/2012 Dr. Kusum Gaur 36
- 37. Measures of Association Significance Test – to test significance of difference in exposure between control and Cases Odds ratio - ratio of the odds of contracting disease in given exposure Relative Risk – Ratio between incidence among exposed and incidence among non-exposed Attributed Risk – percentage of difference between incidence among exposed and non-exposed with incidence among exposed RR or OR of 1 indicate no effect of exposure (equal odds) 12/08/2012 Dr. Kusum Gaur 37
- 38. ‘Z’ Score of Exposure Rates Cases control Exposed a b a x 1oo Exposure Rates = in Cases Non- c d exposed (P2) a+c b x 1oo Exposure Rates = in Controls P2 – P1 (P1) b+d Z Score = SEDP P1 Q 1 P 2 Q 2 SEDP = ------------- + -------- 09/03/2010 Dr. Kusum Gaur 38 N1 N2
- 39. ad ODD’s Ratio = Times bc Incidence among Exposed RR = Times Incidence among Non-Exposed a/a+b a (c+d) = = c/c+d c (a+b) 09/03/2010 Dr. Kusum Gaur 39
- 40. Attributed Risk (Incidence among Exposed - Incidence among Non-Exposed) AR = x 100 Incidence among Exposed a Incidence among Exposed= x 100 a+b c Incidence among Non-Exposed= x 100 c+d 09/03/2010 Dr. Kusum Gaur 40
- 41. Experimental Studies Clinical trials provide the “gold standard” of determining the relationship between factor and the event 12/08/2012 Dr. Kusum Gaur 41
- 42. Types of Experimental Study As per Randomization: • Randomized Control Trials (RCT) • Concurrent Parallel Design (RCT) • Sequential RCT Design • RCT with External Control • Non – Randomized Trials 12/08/2012 Dr. Kusum Gaur 42
- 43. Types of Experimental Study…. As per Construction: • Simple • Cross-Over Study Design As per Study Area: • Field Trials • Clinical Trials • Lab. Trials 12/08/2012 Dr. Kusum Gaur 43
- 44. Quality of Experimental Study • Randomization • Blinding • Control • Cross-Over 12/08/2012 Dr. Kusum Gaur 44
- 45. Controls in Clinical Trials A clinical trial is a comparative, prospective experiment conducted in human subjects • Historical controls are better than no controls • Patients can serve as own controls - This is usually beneficial as the comparison removes patient differences 12/08/2012 Dr. Kusum Gaur 45
- 46. Blinding Good practice: factors that can affect the evaluation of outcome should not be permitted to influence the evaluation process Single-blind Patient or evaluator (either of one) is blinded as to intervention Double-blind design Neither patient nor outcome evaluator knows Rx to which patient was assigned 12/08/2012 Dr. Kusum Gaur 46
- 47. Randomized Control Trials (RCT) • Before and After Comparison • Comparison with Placebo • Comparison Of two medicine/procedure/tests • Comparison Of > two medicine/procedure/tests 12/08/2012 Dr. Kusum Gaur 47
- 48. Experimental Study • Other Name Intervention Study • Objective To know the effect of intervention • Unit of Study Individual meeting entry criteria • Study Question What is happening after intervention in both groups • Direction of Inquiry I E • Study Design 1(Intervention with Placebo) Positive Outcome Group 1/cases Intervention Negative Outcome Positive Outcome Group Placebo 2/control Negative Outcome 12/08/2012 Dr. Kusum Gaur 48
- 49. Clinical Trial R Treatment a Outcomes Group n d Study o Population m i z Outcomes e Control Group 12/08/2012 Dr. Kusum Gaur 49
- 50. Intervention Study - Design 2 (Comparison of Effect of Two Interventions) Cases Meeting Entry criteria Group - 1 Group -2 Intervention -1 Intervention Intervention - 2 Positive Negative Positive Outcome Negative Outcome Outcome Outcome 12/08/2012 Dr. Kusum Gaur 50
- 51. Cross Over Design Group -1 Cases Group-2 Meeting Entry criteria Intervention - 2 Intervention - 1 Positive Negative Positive Negative Outcome Outcome Outcome Outcome Group -1 Group -2 Crossover Intervention -2 Intervention -1 Positive Negative Positive Negative Outcome Outcome Outcome Outcome 12/08/2012 Dr. Kusum Gaur 51
- 52. Other Types of Experimental Study • Quincy Experimental Study • Block Experimental Study 12/08/2012 Dr. Kusum Gaur 52
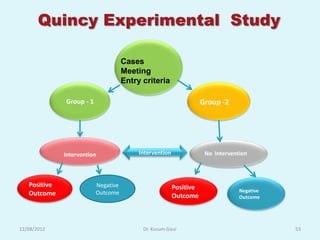
- 53. Quincy Experimental Study Cases Meeting Entry criteria Group - 1 Group -2 Intervention Intervention No Intervention Positive Negative Positive Outcome Negative Outcome Outcome Outcome 12/08/2012 Dr. Kusum Gaur 53
- 54. Block Experimental Study Cases Meeting Entry criteria Group -3 Group - 1 Group -2 Intervention Intervention-3 Intervention -1 Intervention Intervention-2 Positive Positive Negative Negative Outcome Outcome Outcome Outcome Positive Negative Outcome Outcome 12/08/2012 Dr. Kusum Gaur 54
- 55. Steps of Experimental Study Drawing up a Protocol Reference Population Sample Population Exclusions Randomization Experimental Group Control Group Manipulation/Intervention Follow - up 12/08/2012 Assessment of Outcome Dr. Kusum Gaur 55
- 56. Ideal Study Design for established causality Ethical Issues
- 57. STUDY QUESTIONS AND APPROPRIATE DESIGNS Type of Question Appropriate Study Design Burden of illness Field Surveys - Prevalence Cross Sectional Survey - Incidence Longitudinal survey Causation, Risk & Prognosis Case Control Study, Cohort study, RCT Treatment Efficacy Randomized Controlled study Diagnostic Test Evaluation Randomized Controlled study Cost Effectiveness Randomized Controlled study 12/08/2012 Dr. Kusum Gaur 57
- 58. Hierarchy of Epidemiological Study Design Establish Causality RCT Cohort Case Control Cross-Sectional Case Series Generate Hypothesis Case Report 12/08/2012 Dr. Kusum Gaur 58
- 59. 12/08/2012 Dr. Kusum Gaur 59