0% found this document useful (0 votes)
34 views14 pagesMangrove Biomass and Carbon in Cameroon
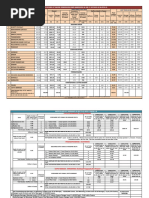
This study investigates the distribution of aboveground biomass and soil organic carbon in a degraded mangrove ecosystem within Douala-Edéa National Park, Cameroon. It highlights the significant relationships between mangrove stand structure and soil organic carbon at various depths, revealing a negative association at deeper soil levels. The findings emphasize the importance of mangrove ecosystems in carbon sequestration and the impact of human activities on their health and sustainability.
Uploaded by
Haruhi333Copyright
© © All Rights Reserved
We take content rights seriously. If you suspect this is your content, claim it here.
Available Formats
Download as PDF, TXT or read online on Scribd
0% found this document useful (0 votes)
34 views14 pagesMangrove Biomass and Carbon in Cameroon
This study investigates the distribution of aboveground biomass and soil organic carbon in a degraded mangrove ecosystem within Douala-Edéa National Park, Cameroon. It highlights the significant relationships between mangrove stand structure and soil organic carbon at various depths, revealing a negative association at deeper soil levels. The findings emphasize the importance of mangrove ecosystems in carbon sequestration and the impact of human activities on their health and sustainability.
Uploaded by
Haruhi333Copyright
© © All Rights Reserved
We take content rights seriously. If you suspect this is your content, claim it here.
Available Formats
Download as PDF, TXT or read online on Scribd