Chemistry(1)
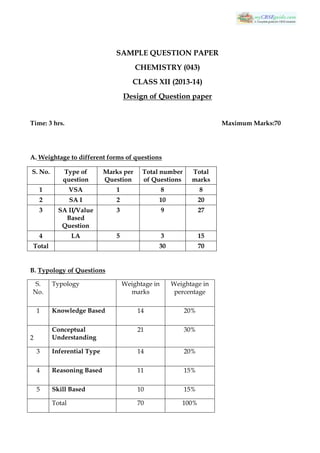
- 1. SAMPLE QUESTION PAPER CHEMISTRY (043) CLASS XII (2013-14) Design of Question paper Time: 3 hrs. Maximum Marks:70 A. Weightage to different forms of questions S. No. Type of question Marks per Question Total number of Questions Total marks 1 VSA 1 8 8 2 SA I 2 10 20 3 SA II/Value Based Question 3 9 27 4 LA 5 3 15 Total 30 70 B. Typology of Questions S. No. Typology Weightage in marks Weightage in percentage 1 Knowledge Based 14 20% 2 Conceptual Understanding 21 30% 3 Inferential Type 14 20% 4 Reasoning Based 11 15% 5 Skill Based 10 15% Total 70 100%
- 2. C. Scheme of options There will be no overall choice. However, internal choice in any one question of two marks, any one question of three marks and all the three questions of five marks weightage has been provided. D. Difficulty level of questions S. No. Estimated difficulty level Percentage of marks 1 Easy 15 2 Average 70 3 Difficult 15
- 3. SAMPLE QUESTION PAPER CHEMISTRY (043) CLASS-XII – (2013-14) BLUE PRINT Time Allowed: 3 Hrs. Maximum Marks:70 S.No. UNIT VSA (1 Mark) SAI (2 Marks) SAII(3)/ Value based question (3marks) LA (5 marks) TOTAL 1. Solid State 4(2) 4 (2) 2. Solutions 5 (1) 5 (1) 3. Electrochemistry 2(1) *3(1) 5(2) 4. Chemical Kinetics 1(1) 4(2) 5(3) 5. Surface Chemistry 1(1) 3(1) 4(2) 6. General Principles and Processes 3(1) 3(1) of Isolation of Elements 7. p-block Elements 1(2) 3(2) 8(4) 8. d & f- Block Elements 5(1) 5(1) 9. Co-ordination Compounds 1(1) 2(1) 3(2) 10. Haloalkanes and Haloarenes 2(2) 4(2) 11. Alcohols, Phenols & Ethers 1(1) 3(1) 4(2) 12. Aldehydes, Ketones & Carboxylic 1(1) 5(1) 6(2) Acids 13. Organic Compounds Containing 4(2) 4(2) Nitrogen 14. Biomolecules 1(1) 3(1) 4(2) 15. Polymers 3(1) 3(1) 16. Chemistry in Everyday Life 3(1) 3(1) Total: 8(8) 20(10) 27(9) 15(3) 70(30) Key: Total marks (no. of questions) * Value Based Question NOTE : - Value Based Questions may be asked from any unit / chapter / topic. - It will carry 3-5 marks.
- 4. SAMPLE QUESTION PAPER CHEMISTRY (043) CLASS-XII – (2013-14) Time Allotted: 3 Hrs Maximum Marks: 70 General Instructions: 1. All questions are compulsory. 2. Question No. 1-8 are very short answer questions and carry 1 mark each. 3. Question No. 9-18 are short answer questions and carry 2 marks each. 4. Question No. 19-27 are also short answer questions and carry 3 marks each. 5. Question No. 28-30 are long answer questions and carry 5 marks each. 6. Use log tables if necessary, use of calculators is not allowed. Q.1 Bond enthalpy of fluorine is lower than that of chlorine, why? Q.2 Give the IUPAC name of the following compound: (CH3)3-C-C-COOH ║ O Q.3 On increasing temperature, activation energy of a reaction decreases, why? Q 4 Which of the following is most effective electrolyte in the coagulative of AgI/Ag+sol? K2SO4, MgCl2, K4[Fe(CN)6] Q.5 Write the reaction when glucose is heated with excess of HI. Q.6 Which Xe compound has distorted octahedral shape? Q.7 What is the denticity of co-ordination compound used for the treatment of lead Poisoning? Q.8 An alkoxide is a stronger base than hydroxide ion. Justify. Q.9 (a) State the law which helps to determine the limiting molar conductivity of weak electrolyte. (b) Calculate limiting molar conductivity of CaSO4 (limiting molar conductivity of calcium and sulphate ions are 119.0 and160.0 S cm 2 mol -1 respectively). Q.10 Rate constant K for first order reaction has been found to be 2.54 x 10 -3 sec -1 . Calculate its three-fourth life. OR A first order gas reaction A2(g)B2(g) → 2A(g) + 2B(g) at the temperature 400 0 C has the rate Constant K =2.0x10 -4 sec -1 . What percentage of A2B2 is decomposed on heating for 900 seconds.
- 5. Q.11 Do the following conversions: (i) Methyl bromide to acetone. (ii) Benzyl chloride to 2-phenyl acetic acid. Q.12 How will you distinguish between the following pairs of compounds: (i) Chloroform and carbon tetra chloride. (ii) Benzyl alcohol and chlorobenzene. Q.13 For a chemical reaction variation in rate with conc. is shown below: Rate Conc.—→ What is the order of the reaction? What are the units of rate constant K for the reaction? Q.14 Give the electronic configuration of d-orbitals of K3 [Fe(CN6)] and K3 [FeF6] and explain why these complexes give different colour with same solution. (At. No. Of Fe=26u) Q.15 Give reason for the following: (i) O-Toludine is more basic than aniline. (ii) Tertiary amines do not undergo acetylation reaction. Q.16 Write the following name reaction: (i) Gabriel phthalimide reaction. (ii) Hoffman bromamide reaction. Q.17 Silver metal crystallises with a face centred cubic lattice. The length of unit cell is found to be 4.077x10 -8 cm. Calculate atomic radius and density of silver. (atomic mass of Ag = 108u, NA = 6.02x10 23 mol -1 ) Q.18 Calculate packing efficiency in ccp structure.
- 6. Q.19 Manu and his father went to a shop to purchase a battery for their inverter. Shopkeeper showed them two types of batteries, one with lead plates and the other with cadmium plates. The battery with cadmium plates was more expensive than the lead battery. They decided to purchase lead battery as it was cheaper. After reading the above passage, answer the following questions: a) As a student of chemistry, why would you suggest to Manu and his father to buy the expensive cadmium plate battery. Give two reasons. b) Which two values will you be promoting through your suggestions? Q.20 Give a reason for the following: (i) Rough surface of catalyst is more effective than smooth surface. (ii) Smoke passed through charged plates before allowing it to come out of chimneys in factories. (iii) Ne gets easily absorbed over charcoal than He. Q.21 (a) Give one example of each of the following: (i) Acidic flux (ii) Basic flux (b) What happens when: (i) Cu2O undergoes self reduction in a silica line converter. (ii) Haematite oxidises carbon to carbon monoxide. OR (a) What role does cryolite play in Hall Haraoult process? (b) How can alumina be separated from silica in a bauxite ore associated with silica? Give equations also. Q.22 Write balanced chemical equations for the following reactions. (a) Hypophosphorous acid is added to AgNO3 solution. (b) Chlorine gas is passed through hot and concentrated solution of sodium hydroxide. (c) XeF2 undergoes hydrolysis.
- 7. Q.23 (i) Draw the structure of sulphuric acid. (ii) A sparkless current is passed through oxygen to prepare ozone. why? (iii) Bleaching action of sulphur is a temporary action .Comment. Q.24 (i) Give one structural difference between amylose and amylopectin (ii) Name the protein and its shape present in oxygen carrier in human body. (iii) Name two fat storing tissues in human body. Q.25 Define the following by giving one example of each: (i) Antiseptics (ii) Antioxidants (iii) Narcotic analgesics Q.26 (a) Write the names of the monomers of polymer used for making unbreakable crockery. . (b) write the reaction of preparation of neoprene. (c) Arrange the following polymers in decreasing order of intermolecular forces. PVC, Nylon 66, Natural rubber. Q.27 Write the mechanism for preparation of ethanol from ethene. Q.28 (a) What is the freezing point of 0.4 molal solution of acetic acid in benzene in which it dimerises to the extent of 85%. Freezing point of benzene is 278.4k and its molar heat of fusion is 10.042kj mol -1 . (b) Explain the following: (i) Solution of chloroform and acetone is an example of maximum boiling azeotrope. (ii) A doctor advised a person suffering from high blood pressure to take less quantity of common salt. OR (a) Calculate the boiling point of a solution containing 0.61g of benzoic acid in 5 g of CS2 .Assuming 84% dimerisation of acid. The boiling point and Kb of CS2 are 46.2 0 C and 2.3 K Kg mol -1 respectively. (b) State Raoult's law for the solution containing non-volatile solute. Give its mathematical expression also. Q.29 Account for the following :
- 8. (i) Transition elements show highest oxidation state in their oxides than fluorides. (ii) Cu has positive electrode potential in the first transition series. (iii) Ionisation enthalpy of lanthanides is higher than actinides. (iv) Potassium dichromate is a good oxidising agent in acidic medium. (v) Actinides show more number of oxidation states than lanthanides. OR (a) Compare non transition and transition elements on the basis of their (i) Variability of oxidation states (ii) stability of oxidation states. (b) Give chemical reactions for the following observations: (i) Potassium permanganate is a good oxidising agent in basic medium. (ii) Inter convertibility of chromate ion and dichromate ion in aqueous solution depends Upon pH of the solution. (iii) Potassium permanganate is thermally unstable at 513K. Q.30. (a) Give names of the reagents to bring about the following transformations: i) Ethanoic acid to ethanol ii) Propane-1-ol to propanal iii) Pent-3-en-2-ol to pent-3-en-2-one iv) Sodium benzoate to benzene (b) Arrange the following in the uncreasing order of: i) Methanal, Propanal, Butanone, Ethanal, Propanone (nucleophilic addition reaction) ii) Formaldehyde, Acetone, Acetaldehyde (reactivity towards HCN) iii) Acetophenone, p-tolualdehyde, p-nitrobenzaldehyde, Benzaldehyde (nucleophilic addition reaction) OR (a) Bring out the following conversions:. (i) 4-nitrotoluene to 2-bromobenzoic acid. (ii) Ethylcyanide to 1- phenyl propanone. (c) Give a reason for the following : (i) Chloroacetic acid is more acidic than acetic acid. (ii) Carboxylic acids have higher boiling point than alcohols. (iii) 4-nitrobenzoic acid is more acidic than 4-methoxy benzoic acid.
- 9. SAMPLE QUESTION PAPER CHEMISTRY(043) MARKING SCHEME TIME ALLOTED :3 Hrs MAXIMUM MARKS;70 1. Relatively large electron-electron repulsion among the lone pairs of F2 molecule but they are much closer to each other in Cl2 molecule. 1 2. 3,3-Di methyl-2-oxobutanoic acid. 1 3. Temperature and activation energy are inversely proportional to each other. 1 4. K4[Fe(CN)6] 1 ∆ 5. CHO-(CHOH)4CH2OH + HI (excess) –———→ n-hexane (C6H14) 6. XeF6 1 7. Name is Ethylenediamine tetraacetate and denticity is 6. 1 8. Due to the presence of an alkyl group higher electron density is found on alkoxide ion. 1 9. (a)Kohlrausch law of independent migration of ions: The limiting molar conductivity of an electrolyte can be represented as the sum of the individual contribution of the anions and cations of the electrolyte. 1 (b) Λ 0 m (CaSO4) = λ 0 Ca2 + + λ 0 SO4 2- = 119.0 S cm 2 mol -1 + 106.0 S cm 2 mol -1 ½ = 225.0 S cm 2 mol -1 . ½ 10. 2.303 a K = —— log —— 1/2 t a-x 2.303 a t = —— log —— k a-x [a = 1, x = ¾] 2.303 1 t3/4 = —— log —— 1/2 2.54 x 10 3 1-3/4 1 t3/4 = 0.9066 x 10 3 x 0.6021 176
- 10. t3/4 = 5.46 x 10 2 sec OR 2.303 a K = —— log —— 1/2 t a-x 2.303 a 2.0 x10 -4 (sec -1 ) = —— log —— 1/2 900 a-x . 2.0x10 -4 x 900 a log —— = ———————— = 0.0781 1/2 a-x 2.303 a taking antilog —— = antilog (0.0781) =1.197 a-x a = 1.197a - 1.197x 0.197 x = ——— a = 0.1645a 1/2 1.197 Where a = 100, then x = 0.1645 x 100 = 16.45 i.e 16.45 % of initial concentration has changed into products. OMgBr dry ether | H3O + Cu 11.(i) CH3Br + Mg ——— —→ CH3MgBr + CH3CHO — — →C— H— →CH3CHOH——→ | | 573K . CH3 CH3 1 (CH3)2CO H3O + . (ii) C6H5CH2OH +SOCl2 —→ C6H5CH2Cl + KCN—→ C6H5CH2CN—→ C6H5CH2COOH 1 12. (a) On heating chloroform and carbon tetrachloride with aniline and ethanolic potassium hydroxide separately chloroform forms pungent smelling isocyanide but carbon tetra chloride does not form this compound. 1 (b) On adding sodium hydroxide and silver nitrate to both the compounds benzyl chloride 1 forms white precipitate but chlorobenzene does not form white precipitate. 13. (i) Order of reaction is zero. 1 (ii) units of rate constant is mol L -1 s- 1 1
- 11. 14. Oxidation state of Fe in K3[Fe(CN)6] is +3 Configuration of Fe 3+ is [Ar]3d 5 3d 4s 4p ↿ ↿ ↿ ↿ ↿ ↿⇃ 3d 4s 4p ↿⇃ ↿⇃ ↿ it has 5 unpaired electrons in 3d orbital which get paired leaving behind one unpaired electron only. In K3[FeF6] oxidation state of Fe is +3 and 5 unpaired electrons are there in 3d orbitals. 3 d 4s 4p ↿ ↿ ↿ ↿ ↿ Because of the presence of different no. of unpaired electrons these impart different colour of same solution. 1 15.(i) Ortho toludine is more basic than aniline due to the presence of electron releasing methyl group (+I effect) electron density at nitrogen of NH2 in o-toluidine increases. 1 (ii) Due to the absence of replaceable hydrogen atom at nitrogen. 1 16.(i) Gabriel phathalimide reaction 1 ( b) Hoffmann bromamide degradation reaction R-C-NH2 + Br2 + 4NaOH → R-NH2 + Na2CO3 + 2NaBr + 2H2O 1 ║ O 17. Given =a = 4.077 x 10 -8 cm, Z = 4 , M = 108 g mol -1 NA =6.022 x 10 23 Z x M = ————— 1/2 a 3 x N A
- 12. 19. a) -Harmful effects of lead on the human being 2 - as well as on the environment It is wise to choose sustained long term benefit than short term gain with regard to health/money Knowledge of chemistry and its relation to environment b) (i) Environmental protection (ii) Health concerns 1 20. (i) Rough surface of a catalyst provides more surface area for adsorption. 1 (ii) So that unburnt charged carbon particles get settled between the charged plate leaving behind air free from pollutants. 1 (iii) Ne has higher critical temperature i.e stronger vander waal, s forces therefore easily adsorbed. 1
- 13. 21 (a) Acidic flux is SiO2 1/2 Basic flux is CaO 1/2 (b) (i) Cu2O undergoes self reduction to form blister copper as 2Cu2O + Cu2S ——→ 6Cu + SO2 1 (ii) Fe2O3 + 3C ——→ 3CO + 2Fe 1 OR (a) Cryolite reduces melting point of alumina . 1 (b) Concentration of ore is carried out by digesting the powdered ore with a concentrated solution of NaOH at 473-523 K and 35 -36 bar pressure. Al2O3 is leached out as sodium meta aluminate and SiO2 as sodium silicate leaving behind impurities. Al2O3 (s) + 2 NaOH(aq) + 3H2O → 2Na[Al(OH)4] (aq) 1 The resulting solution is filtered, cooled and neutralised by passing CO2 gas through it. Then hydrated Al2O3 gets precipitated leaving sodium silicate in the solution. 2Na[Al(OH)4] (aq) + CO2(g) — →Al2O3.xH2O(s) + 2NaHCO3(aq) 1/2 Sodium silicates remain in the solution and hydrated alumina is filtered , washed, dried and heated to get pure alumina (Al2O3). 1470K Al2O3 .x H2O(s) ——— → Al2O3(s) + x H2O(g) 1/2 22.(i) H3PO3 + 2AgNO3 + H2O —— → 2Ag + HNO 3+ 2 H3PO4 1 (ii) 3Cl2 +6 NaOH (conc.) —— → 5NaCl + NaClO3 +3 H2O 1 (iii) 2XeF2 + 2H2O —— → 2Xe + O2 +4 H+ + 4F- 1
- 14. 23(i) 1 (ii) Since the formation of ozone from oxygen is endothermic reaction silent electric discharge prevents its decomposition. 1 (iii) Bleaching by sulphur dioxide is temporary because of its reducing nature. 1 24 (i) Amylose is a long unbranched chain polymer -D(+) glucose. 1/2 Amylopectin is a branched chain polymer of -D glucose. 1/2 (ii) Globular protein and its shape is spherical. ½ +1/2 (iii) Liver and adipose tissue. ½ +1/2 25. (i) Antiseptics are the chemicals applied to the living tissues either to kill or prevent the growth of microorganisms .e.g dettol. ½ +1/2 (ii) Antioxidants are the compounds which retard the action of oxygen on food and reduces its rate of decomposition by oxidation.e.g BHA ½+1/2
- 15. (iii) Narcotic analgesics are the chemicals used for the relief of post operative pain.e.g morphine.1 26. (i) Monomers of the polymer are melamine and formaldehyde. ½+1/2 polymerisation (ii) n CH2=C-CH=CH2————— —→ —[ —CH2-C =CH-CH2 —]n— 1 | | Cl Cl (iii) Nylon66 > Natural rubber > PVC 27. 1 1 28 : : Given, m= 0.4, Tf 0 = 278.4, ∆Hfus = 10.042 KJ mol-1 RTf 2 M 8.34 x 103 x (278.4)2 x 78 Kf = ——— = ——————————— = 5.0 K Kg mol-1 ½ + ½
- 16. 1000 x∆Hfus 1000 x 10.042 2CH3COOH————→ (CH3COOH)2 i-1 i-1 α = ——— = ——— 1/2 1/n -1 ½ i-1 = ——— = 0.85 1/2 -1 = i-1 = -0.425 1/2 i = 0.575 ∆Tf = I Kf m = 0.575 x 5 x 0.4 = 1.15 Tf = Tf 0 - ∆Tf = 278.4 – 1.15 = 277.25 K ½ + 1/2 (b) i) This solution has lesser vapour pressure due to stronger interactions between chloroform and acetone molecules. 1 ii) Because higher quantity of NaCl will increase no. of sodium and chloride ions in the body fluid which can increase the osmotic pressure of body fluid i.e blood pressure of a person. 1 OR 2C6H5COOH ———→ (C6H5COOH)2 1
- 17. i -1 i -1 α = —————— = 0.84 = ————— ½ +1/2 1/n – 1 ½ -1 0.84 i = 1 - —————— = 1 -0.42 =0.58 2 Normal molar mass of benzoic acid =122g mol-1 ∆Tb =i.Kb . m = 0.58 x 2.3 x0.1 = 0.13340 C ½+1/2 Tb = Tb0 + ΔTb = 46.2 + 0.1334 = 46.3340 C (b)Raoult,s law: The relative lowering of vapour pressure of the solvent over a solution is equal to the mol fraction of non-volatile solute present in the solution. 1+1 P0 1 - P1 Mathematical expression χ B = —————— P0 1 29.(i) Because oxygen forms multiple bonds with transition metals but fluorine does not form multiple bonds. 1 (ii) Cu has lower hydration enthalpy which is unable to compensate sum of first and second ionisation enthalpy. 1 (iii) 4f electrons of lanthanides is less effectively shielded by nuclear charge than 5f electrons of actinides. 1 (iv) Because dichromate ions get reduced to chromium ions in acidic medium. 1 (v) The actinides show more number of oxidation states than lanthanides because in actinides 5f,6d and7s levels have comparable energies. 1 OR
- 18. (i) Oxidation states of transition elements differ from each other by unity. In non transition elements Oxidation states normally differ by a unit of two. ½+1/2 (ii) In transition elements higher oxidation states are favoured by heavier elements whereas in non transition elements lower oxidation state is favoured by transition elements. ½+1/2 (b) i) 2MnO4 - + H2O + I- ——→ 2MnO2 +2OH- + IO3 - 1 Acid (pH less than 7) (ii) 2CrO4 2- + 2H+ —————————→ Cr2O7 2- + H2O 1 ←————————— Alkali(pH more than 7) Δ 513K (iii)2 KMnO4 ———→ K2MnO4 + MnO2 + O2 1 30. (a) (i) Li AlH4/H3O+ 1/2 (ii) PCC 1/2 (iii) PCC 1/2 (iv) sodalime 1/2 (b) (i) Butanone < Propanone < Propanal < Ethanal < Methanal. 1 (ii) Acetone< Acetaldehyde< Formaldehyde. 1 (iii) Acetophenone< p- Tolualdehyde< Benzaldehyde<p-Nitro benzaldehyde. 1 OR (a) (i)
- 19. 1 NMgBr C6H5 Ether ║ H3O+ | (ii) CH3—CH2—CN + C6H5MgBr ———→ CH3CH2—C—C6H5 ——→C2H5—C=O 1 (b) (i) Because chloroacetate ion is more resonance stabilised than acetate ion due to the presence of electron withdrawing chlorine atom (+I effect). 1 (ii) Due to the presence of extensive intermolecular hydrogen bonding in carboxylic acids than in alcohols molecules. 1 (iii) Because of –I effect of nitro group stability of benzoate ion increases in 4-nitro benzoic acid. But methoxy (+Ieffect) group decreases the stability of benzoate ion. 1